Start New Query
Effect of autologous mesenchymal stem cells induced by low level laser therapy on cardiogenesis in the infarcted area following myocardial infarction in rats
Hana Tuby1, Tali Yaakobi1, Lidya Maltz1, Yaakov Delarea2, Orit Sagi-Assif2, Uri Oron1* - (Publication) 4467
This study showed rats that were give a heart attack and then treated with the laser on their shins saw a 55% reduction in infarction size in the heart showing that the stem cells released from the bone migrated to the heart.
View Resource
1Department of Zoology, The George S. Wise Faculty of Life Sciences, Tel-Aviv University, Tel-Aviv, Israel
2Department of Cell Biology and Immunology, The George S. Wise Faculty of Life Sciences, Tel-Aviv University, Tel-Aviv, Israel
Email: *oronu@post.tau.ac.il
Received 27 May 2013; revised 29 June 2013; accepted 16 July 2013
ABSTRACT
In this study, we investigated the hypothesis that photo- biostimulation by low-energy laser therapy (LLLT) applied to the bone marrow (BM) of myocardial in- farcted rats may attenuate the scarring processes that follow myocardial infarction (MI). Wistar rats under- went experimental MI. LLLT (Ga-Al-As diode laser) was applied to the BM of the exposed tibia at differ- ent time intervals post-MI (4 hrs, 48 hrs and 5 days). Sham-operated infarcted rats served as control. In- farct size was significantly reduced (55%) in the la- ser-treated rats as compared to the control non-treat- ed rats, at 2 weeks post-MI. A significant 3-fold in- crease was observed in the density of desmin immu- nopositive stained cells 14 days post-MI in the infarc- ted area of the laser-treated rats as compared to the non-laser-treated controls. The electron microscopy from the control infarcted rat hearts revealed a typi- cal interphase area between the intact myocardium and the infarcted area, with conspicuous fibroblasts with collagen deposition dispersed among them. In rats that were laser treated (to BM), the interphase zone demonstrated cells with different intracellular struc- tures. There was also a significant increase in the per- centage of c-kit positive cells and macrophages in the circulating blood of the laser treated rats as compar- ed to control non treated ones. In the majority of the cells clusters of myofibrils anchored to well-developed Z-lines and structures resembling the morphological characteristics of mature intact cardiomyocytes were evident. In conclusion, LLLT to the BM of rats post- MI induces cardiogenesis mainly at the borders of the infarcted area in the heart.
Keywords: Low-Level Laser Therapy; Myocardial Infarction; Macrophage; Desmin; Ultrastructure; c-Kit Positive Cells
1. INTRODUCTION
Regenerative capacity and mitotic activity in the heart are confined mainly to the lower vertebrates [1]. Amputation of ~20% of the zebrafish’s ventricular myocardium re- sulted in full regeneration without scarring [2]. In am- phibians, heart injury was associated with increased cell proliferation of myocytes and enhanced regeneration [3]. The adult mammalian heart was traditionally considered to be a post-mitotic organ with terminally differentiated cardiac myocytes. However, this dogma has recently been challenged by several studies and reviews [4-8]. These studies have suggested that cardiac myocytes are replaced throughout the lifespan even in the human heart, and that myocytes can regenerate from resident cardiac progenitor cells (CPC) as well as from bone marrow (BM). Studies in human infarcted hearts have shown evidence of cytoki- nesis of cells in the heart and evidence of cardiac stem cells that are activated in response to ischemic injury. This growth response is attenuated in chronic heart fail- ure [9]. Some studies have reported that cardiac myocyt- es can be derived from BM; specifically, side population precursor cells following induction of myocardial infarc- tion (MI) by left anterior descending artery (LAD) liga- tion [10-12]. Contradicting these findings, other laborato- ries using genetic markers have reported that lineage ne- gative, c-kit+ BM cells did not differentiate into cardio- myocytes [13]. It was also suggested that BM-derived stem cells may stimulate the small population of stem cells in the ischemic heart to proliferate and differentiate to enhance cardiac repair post-MI [14]. In a recent study transient regenerative potential in the mouse heart was demonstrated during the neonatal period [15].
Low-level laser therapy (LLLT) has been found to modulate various biological processes [16,17], such as increasing mitochondrial respiration and ATP synthesis [18], facilitating wound healing and promoting the proc- ess of skeletal muscle regeneration and angiogenesis [19- 21]. In an experimental model of the infarcted heart in rats and dogs, it was demonstrated that LLLT application directly to the infarcted area in the heart at optimal power parameters significantly reduced scar tissue formation [22-24]. This phenomenon was partially attributed to a significant elevation in ATP content, heat shock proteins, vascular endothelial growth factor (VEGF), inducible ni- tric oxide (NO) synthase, and angiogenesis in the ischemic zone of the laser-irradiated rats, as compared to non- irradiated rats [25].
The effect of photobiostimulation on stem cells or pro- genitor cells has not been extensively studied. LLLT ap- plication to normal human neural progenitor cells signi- ficantly increases ATP production in these cells [26]. LLLT delivery to MSCs and cardiac stem cells in vitro caused a significant enhancement in their proliferation rate [27,28]. LLLT has also been shown to increase the proliferation rate of adipose-derived stem cells in vitro [29]. Recently, we demonstrated that LLLT application to autologous BM could induce mesenchymal stem cells (MSCs) in the BM to proliferate and cause their recruit- ment and specific homing in on the infarcted rat heart and not on other organs [30,31]. The laser treatment to the BM also caused a marked and statistically significant reduction of 79% in the scarring and ventricular dilata- tion followed MI as compared to infarcted non-laser- treated rats. The aim of the present study was to investi- gate the possibility that induction of stem cells in the BM of rats by LLLT could also affect cardiogenesis in the in- farcted rat heart.
2. MATERIALS AND METHODS
2.1. Experimental Procedures
A total of 21 Wistar male rats, weighing 200 - 250 gr, that underwent ligation of the LAD artery to induce MI, were used as described by us previously [23]. All the ex- perimental procedures were approved by the animal care committee of Tel-Aviv University. Briefly, rats were anes- thetized with Avertin (1 ml/100 g body weight I.P.) and the lungs were ventilated. Thoractomy was performed by invasion of the intercostals muscles between the 5th and 6th rib to expose the heart. The LAD artery was occluded 2 mm from the origin with 5-0 polypropylene thread (Ethicon Inc., Cincinnati, OH). Following LAD artery occlusion the chest muscles and skin were sutured and the rats were ventilated until they woke up. The infarcted rats were divided randomly into two groups. In one group LLLT was applied directly to the BM 4 hrs, 48 hrs and 5 days post-MI (see below). The second group was non-laser-treated (the rat’s bone was exposed for the same duration as the laser-treated group but the laser was not turned on). Food and water were supplied ad libitum. Rats were sacrificed 14 days post-MI.
2.2. Laser Application
After induction of MI rats were randomly assigned to a laser-treated or control non-laser-treated group. A diode (Ga-Al-As) laser, wavelength 804 nm with a tunable po- wer output of maximum of 400 mW (Lasotronic Inc., Zug, Switzerland) for application to the BM was used. The laser device was equipped with a metal-backed glass fiber optic (1.5 mm diameter). An infrared viewer (Laso- tronic Inc. Zug, Switzerland) and infrared-sensitive de-tecting card (Newport, Inc., Irvine, CA) were used to de- termine the infrared irradiation area. Laser application was done by a 10 mm longitudinal cut in the skin above the medial aspect, and further delicate cleaning of the bone surface was carried out. The tip of the fiber optic (1.5 mm diameter) was placed perpendicularly to the center of the exposed medial aspect of the tibia and power den- sity of 10 mW/cm2 was applied to the BM. The laser was applied for a duration of 100 sec (energy density 1.0 J/cm2). Left or right exposed tibias were chosen at random for LLLT application. In sham-operated infarcted rats that served as control the tibias were exposed and the fi- ber optic was placed as described above but the laser beam was not turned on.
2.3. Histology and Electron Microscopy
A defined cross-section sample (2 mm thick) from the central part of the infarcted area was taken from all hearts for histology. Eight micron paraffin sections were pre- pared from the tissue samples of each heart. Infarct size was determined using Masson’s trichrome staining as described by us previously [23]. Three observers, blinded to control or laser-treated rats, analyzed infarct size. Six microscopic slides from the infarcted area of each heart were chosen at random for determination of infarct size. Infarct size was expressed as the percentage of the total infarcted area relative to the total area of the left ventri- cle (LV) in each section, using image analysis software Sigma Scan Pro (Sigma, St. Louis, MO).
For electron microscopy three tissue samples from each of the control and laser-irradiated rat hearts were taken from the interphase zone between the infarcted and non-infarcted tissue by macroscopic examination. Fixa- tion was performed in 3.5% glutaraldehyde in 0.1 M ca- codylate buffer for 24 hrs followed by embedment in Epon-812. Semi-thin sections (1 micron) were prepared in order to localize the interphase zone. Thin sections were then prepared and stained with uranyl acetate and lead citrate followed by examination with a Jeol electron microscope.
2.4. Immunohistochemistry
The total number of cells immunostained for desmin (bone marrow cells or newly formed) in the infarcted area were determined using a desmin kit (Zytomed Laboratory, Ber- lin, Germany). The procedure was performed at room temperature with anti-mouse (dilution 1:25 - 1:50) primary antibody for 60 min. Following washing, slides were in- cubated with HRP secondary antibody for mouse for 30 min followed by DAB Chromogen system (Covance Inc., Dedham). Slides were rinsed again in wash buffer, stain- ed in Hematoxylin for nuclei detection, mounted and viewed using a Zeiss microscope equipped with a camera and video screen. The total number of desmin immuno- stained cells within the infarcted area was counted and their density expressed as the percentage of the total area of the infarct using SigmaPro software.
2.5. Flow Cytometry Analysis
Blood samples were taken 2 and 7 days post-IR injury for fluorescence-activated cell sorting (FACS) analysis. 100 μl of blood were mixed with different antibodies: anti-mouse CD117 (c-kit) PE (eBioscience San Diego, USA) and rat IgG2b isotype control PE (eBioscience San Diego, USA) and anti-rat macrophage marker PE (eBio- science San Diego, USA) and mouse IgG2a K isotype control PE (eBioscience San Diego, USA), were used for the FACS analysis according to the manufacturer’s guide- lines. Forty five min post incubation of the whole fresh blood with the relevant antibodies, 2 ml of Fix/Lyse so- lution (eBioscience, San Diego, USA) was added. After mixture the suspended cells were left for 60 min in the dark at room temperature. Centrifugation was performed for 10 min, supernatant was removed and washing of the pellet was performed with 2 ml of Flow Cytometry Stain- ing Buffer Solution (eBioscience, San Diego, California, USA). After another centrifugation for 10 minutes the supernatant was decanted. The pellet containing mono- nucleated cells was resuspended in 200 μl of flow stain buffer for FACS analysis.
2.6. Statistical Analysis
The SigmaStat 2.0 (Sigma, St. Luis, USA) software was used for statistical analysis. Tests were performed first for normality distribution, followed by parametric (stu- dent’s t-test) test.
3. RESULTS
Application of LLLT to the infarcted heart caused a sig- nificant (p = 0.049) reduction of 55% in infarct size as compared to control. The present of macrophages and c- kit positive cells in the blood was determined by FACS analysis (Figure 1). It was found that at 5 days post MI there was a statistical significant 2-fold higher concentra- tion of macrophages and significant 1.4-fold higher c-kit positive cells (mesenchymal cells) in the laser treated rats as compared to the infarcted non laser treated rats. Des- min immunostaining of histological sections of the in- farcted zone from laser-treated rats demonstrated a higher density of positively stained cells than in the non laser-treated ones (Figures 2-4). In the interphase zone, cells extending from the myocardium towards the in 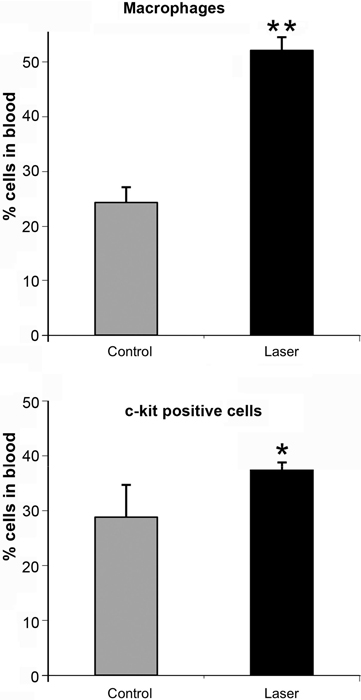
Figure 1. Percent (out of total mononucleated cells) of macro- phages and c-kit positive cells in blood of control and laser treated rats (to the bone marrow) 5 days post MI as revealed by FACS analysis. The results are mean ± S.E.M of 15 rats at each group. Statistical significance *p < 0.05; **p < 0.01.
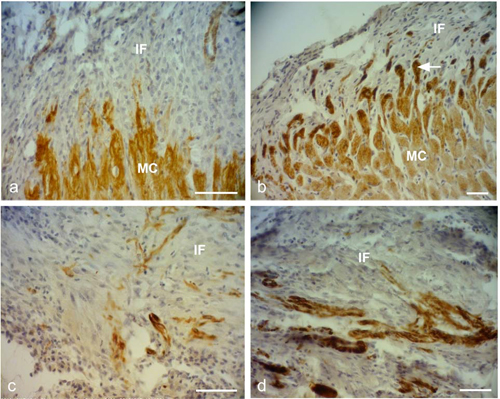
Figure 2. Representative desmin immunostained light micro- graphs of the infarcted zone of non-laser-treated rats (a, c) and laser-treated rats (to the bone marrow at 4 and 48 hrs and 5 days) (b, d) taken 2 weeks post-MI. Note that the zone in the control non-laser-treated rats contains mainly collageneous mate- rial with a few desmin immunopositive cells in the infarcted area (a, c); while in the laser-treated rats the zone displays posi- tive desmin staining in extended outgrowths (arrow) from the myocardium (MC) in (b), and in the cytoplasm of many cells in the infarcted area in (d). IF, Infarcted area. Bar = 50 μm.
farcted area showed higher immunostaining for desmin in the laser-treated rat hearts as compared to the control non-treated ones (Figure 2). The cell density of desmin immune-positive cells was also determined quantitatively in histological sections of both the infarcted laser-treated rats and infarcted non-laser-treated rats. The cell density was significantly (p < 0.01) 3-fold higher in the infarcted area of the laser-treated rats as compared to the non-la- ser-treated controls (Figure 4).
The electron micrographs of all samples taken from the control non-laser-treated infarcted rat hearts revealed a typical interphase area between intact and infarcted heart (Figure 5(a)). Adjacent to the non-ischemic intact myocardium there were conspicuous fibroblasts with col- lagen deposition dispersed among them (Figure 5(a)). In all samples taken from the laser-irradiated hearts the in- terphase zone between intact and infarcted area demon- strated different characteristics to those of the non-laser- treated infarcted rat hearts. Cells with newly-formed or- ganized contractile myofilaments dispersed in the cyto- plasm were detected in groups of several cells (Figure 5(b)). In these cells numerous mitochondria, clusters of ribosomes, and conspicuous clusters of contractile pro- teins were evident in the cytoplasm (Figures 6-8). Some cells contained dispersed contractile myofilaments in the cytoplasm that were still in an early stage of organization (Figure 6). The organization of newly-formed contractile myofilaments in the cytoplasm was observed in various
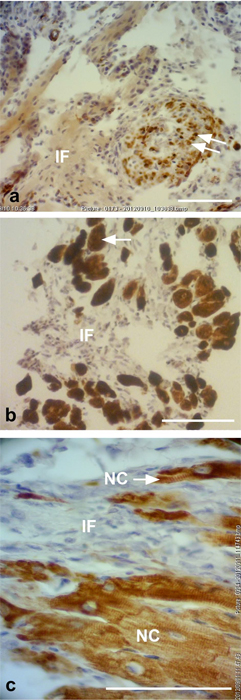
Figure 3. Representative desmin immunostained light micro- graphs of the interphase of the infarcted zone of laser-treated rats. Note that desmin positively stained cross-sections of myo- fibers (arrows) intermingled in the infarcted zone in (a). In (b) immunopositively stained cross-sections of myofibers (arrow) are visible in the infarcted area (IF). In (c) newly-formed car- diomyocytes (NC) are seen, with the desmin immunostaining mainly confined to the Z-line. Bar = 50 μm.
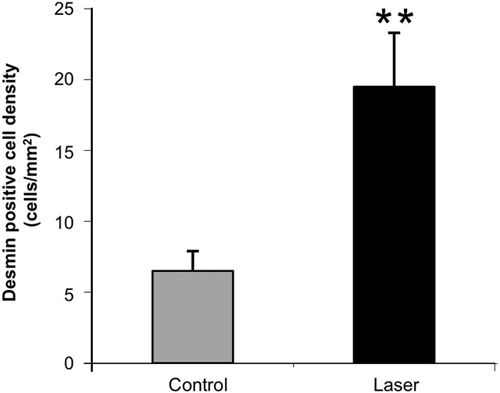
Figure 4. Density of desmin positively stained area (relative to total area) in the infarcted areas of control (non-laser-treated) and laser-treated (to the bone marrow) rats at 14 days post-MI. Results are mean+ S.E.M from 6 - 8 rats in each group. **p < 0.01.
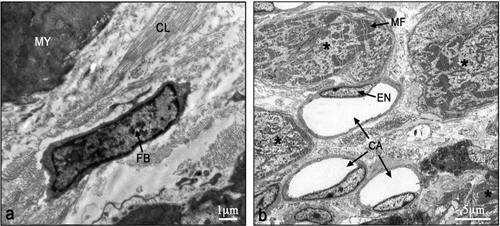
Figure 5. Electron micrographs of typical interphase zone be- tween myocardium and infarcted area of control non-laser- treated (a) and laser-treated (b) to bone marrow rats. Note intact myocardium (MY) and adjacent fibroblast (FB) in the infarcted area surrounded by collagen (CL) deposition in (a). In (b) sev- eral newly-formed cardiomyocytes (marked with asterix) with conspicuous well-organized myofilaments (MF) in their cyto- plasm are evident adjacent to blood capillaries (CA). EN, En- dothelial cell.
degrees of maturation in those cells. In some cells the myofilaments were dispersed in the cytoplasm and in others they were organized in clusters anchored to well- developed Z-lines (Figure 7(a)). In certain cells the myo- filaments were organized parallel to the longitudinal di- rection of the cells, resembling the morphological char- acteristics of mature intact cardiomyocytes (Figure 7(b)). Some of the cells were also seen in a process of forma- tion of typical intercalated disc between them (Figure 9).
4. DISCUSSION AND CONCLUSION
The most significant outcome of this study was the ap- pearance of newly-formed cardiomyocytes following laser treatment to the BM, as indicated by light and electron microscopy. There was a 3-fold increase in the density of
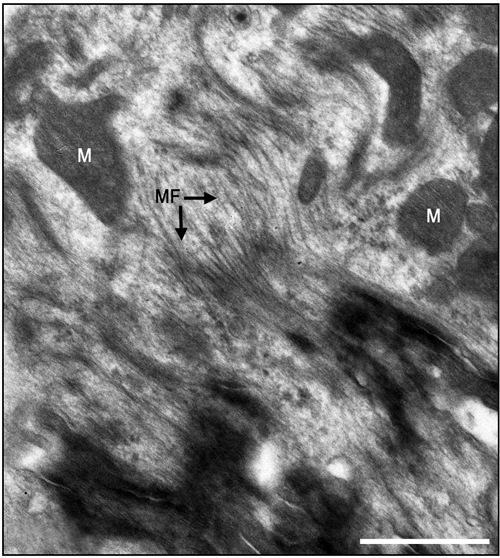
Figure 6. Electron micrographs of most probably newly-formed cardiomyocytes at an early stage of organization of contractile myofilaments. Note myofilaments (MF) in the cytoplasm. M, Mitochondrion. Bar = 1 μm.
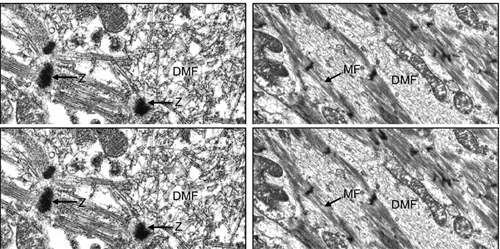
Figure 7. Electron micrographs of most probably newly-formed cardiomyocytes with early (a) and late (b) stages of the organi- zation of the contractile myofilaments in the cytoplasm. Note contractile myofilaments that are dispersed (DMF) in the cyto- plasm with a few organized in clusters anchored to Z-lines (Z) in (a). In (b) myofilaments (MF) are organized in parallel to the longitudinal axis of the cardiomyocyte, resembling their orga- nization in mature cardiomyocyte. N, Nucleus. Bar = 1 μm.
desmin immunostained cells in the infarcted rat hearts that had been laser treated. Desmin is a protein found in the cytoplasm of developing myocytes and cardiomyo- cytes [32]. The significantly higher occurrence of des- min-positive cells in the infarcted area of the laser- treated hearts may indicate the synthesis of new contrac- tile proteins in the developing new cardiomyocytes, re- sembling the process that takes place during embryonic development. The ultrastructural features of the cells in the interphase between the intact myocardium and the
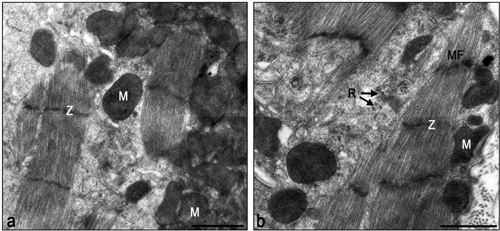
Figure 8. Electron micrographs of typical interphase zone be- tween myocardium and infarcted area of laser-treated infarcted rat heart. Note numerous mitochondria (M) in the cytoplasm of the cardiomyocytes in (a) and (b). Also note organized contrac- tile myofilament with well-developed Z-lines (Z), some dis- persed myofilaments and clusters of ribosomes (R). Bar = 1 μm.
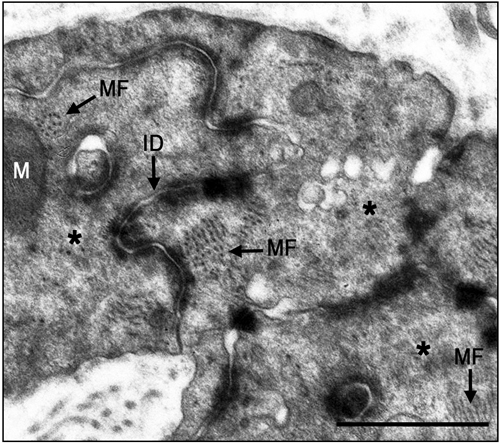
Figure 9. Electron micrographs of typical intercalated disk formation in the interphase region of the infarcted heart of la- ser-treated rats. Formation of intercalated disks (ID) between cells (marked with asterix) is evident. Note that the most proba- bly newly-formed cardiomyocytes contain clusters of myofila- ments (MF) in the cytoplasm that are conspicuous in their obli- que or cross-sections (arrows). Bar = 1 μm.
infarcted myocardium of the laser-treated rats, as shown in this study, clearly resemble the characteristics of car- diomyocytes during embryonic development of the heart [33]. Furthermore, the clusters of ribosomes and the nu- merous clusters of mitochondria in the cytoplasm of these cells may characterize cells that are active in the synthe- sis of proteins. It was previously demonstrated that direct LLLT to the infarcted hearts of rats, dogs and pigs caus- ed a significant reduction of scarring post-MI [23,24]. It was suggested that part of this reduction could be ex- plained by the regenerative response that takes place in the interphase zone [24].
The results of the present study indicate that the LLLT
applied to autologous BM attenuates the concentration of macrophages and MSC in the circulating blood. We have previously shown that LLLT application to the BM of infarcted rats caused a 2 fold enhancement in the rate of proliferation of MSC in the BM [30]. Those cells that most probably leave the BM to the circulating blood in- deed show a significant elevation of their concentration (as reveled by the FACS analysis in the present paper) at 5 days post MI. Consequently these cells probably home in on the infarcted heart, and even migrate specifically to the infarcted area [30]. These cells may induce cardiac stem cells to differentiate to newly-formed cardiomyo- cytes, as suggested previously by Hatzistergos et al. [14]. Indeed, it was found that endogenous c-kit+ cardiac stem cells were increased by 20-fold in the rat infarcted heart compared to control, following transcardial injection of BM-derived MSCs [14]. Such induction may be enabled due to paracrine secretion of various growth factors by the laser-stimulated MSC that originated from the BM. The possibility that paracrine secretion occurs in im- planted stem cells during cell therapy to the heart post- MI has been suggested previously [34]. Another mecha- nism that may take place after homing of stem cells to the infarcted heart of the laser-stimulated rats is that these cells continue to proliferate in the appropriate mi-lieu of the interphase zone in the infarcted heart and then differentiate to cardiomyocytes [30].
Another possible mechanism that maybe associates with the reduction of infarct size is the significant increase in the concentration of macrophages in the circulation fol- lowing LLLT to the BM as revealed from the FACS analysis in the present study. These findings corroborate with studies indicating that macrophages activity in the infarcted area at early stages post MI cause reduction of scarring post MI [35,36]. Thus, it could be postulated that more macrophages that will eventually home in the infarcted area from the circulating blood in the laser treated rats will also contribute to the reduction of scar- ring.
Although the findings of the present study do not in- dicate the extent of regenerative capacity of the rat in- farcted heart post-laser-irradiation, they do reveal a shift from practically no cardiomyocytes in the tissue samples taken from the non-laser-treated hearts, to the presence of newly-formed cardiomyocytes in all the electron mi- croscope sections taken from the hearts of rats that are laser-treated to the BM.
In conclusion, to the best of our knowledge, this is the first study to demonstrate the appearance of newly-form- ed cardiomyocytes in the infarcted area following LLLT to autologous BM in the infarcted rat heart. The mecha- nisms associated with this phenomenon remain to be elu- cidated in further studies.
5. ACKNOWLEDGEMENTS
This study was partially supported by the Elizabeth and Nicholas Shle- zak Super-center for Cardiac Research and Medical Engineering. The authors wish to acknowledge N. Paz for editing the manuscript and V. Wexler for helping with preparation of the figures.
REFERENCES
[1]
Rumyantsev, P.P. (1977) Interrelations of the prolifera- tion and differentiation processes during cardiac myoge- nesis and regeneration. International Review of Cytology, 51, 186-273. doi:10.1016/S0074-7696(08)60228-4
[2]
Poss, K.D., Wilson, L.G. and Keating, M.T. (2002) Heart regeneration in zebrafish. Science, 298, 2188-2190. doi:10.1126/science.1077857
[3]
Rumyantsev, P.P. (1973) Post-injury DNA synthesis, mi- tosis and ultrastructural reorganization of adult frog car- diac myocytes. An electron microscopic-autoradiographic study. Z Zellforsch Mikrosk Anat, 139, 431-50. doi:10.1007/BF00306596
[4]
Barnett, P. and van den Hoff, M.J.B. (2011) Cardiac re- generation: Different cells same goal. Medical & Biologi- cal Engineering & Computing, 49, 723-732. doi:10.1007/s11517-011-0776-5
[5]
Bollini, S., Smart. N. and Riley, P.R. (2011) Resident car- diac progenitor cells: At the heart of regeneration. Jour- nal of Molecular and Cellular Cardiology, 50, 296-303. doi:10.1016/j.yjmcc.2010.07.006
[6]
Choi, W.Y. and Poss, K.D. (2012) Cardiac regeneration. Current Topics in Developmental Biology, 100, 319-343. doi:10.1016/B978-0-12-387786-4.00010-5
[7]
Laflamme, M.A. and Murry, C.E. (2011) Heart regenera- tion. Nature, 473, 326-335. doi:10.1038/nature10147
[8]
Steinhauser, M.L. and Lee, R.T. (2011) Regeneration of the heart. EMBO Molecular Medicine, 3, 701-712. doi:10.1002/emmm.201100175
[9]
Urbanek, K., Torella, D., Sheikh, F., De Angelis, A., Nur- zynska, D., Silvestri, F., Beltrami, C.A., Bussani, R., Bel- trami, A.P., Quaini, F., Bolli, R., Leri, A., Kajstura. J. and Anversa, P. (2005) Myocardial regeneration by activation of multipotent cardiac stem cells in ischemic heart failure. Proceedings of the National Academy of Sciences of the USA, 102, 8692-8697. doi:10.1073/pnas.0500169102
[10]
Bittner, R.E., Schofer, C., Weipoltshammer, K., Ivanova, S., Streubel, B., Hauser, E., Freilinger, M., Höger, H., Elbe- Bürger, A. and Wachtler, F. (1999) Recruitment of bone- marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anatomy and Embryology, 199, 391-396. doi:10.1007/s004290050237
[11]
Jackson, K.A., Majka, S.M., Wand, H., Pocius, J., Hart- ley, C.J., Majesky, M.W., Entman, M.L., Michael, L.H., Hirschi, K.K. and Goodell, M.A. (2001) Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. The Journal of Clinical Investigation, 107, 1395-1402. doi:10.1172/JCI12150
[12]
Pfister, O., Mouquet, F., Jain, M., Summer, R., Helmes, M., Fine, A., Colucci, W.S. and Liao, R. (2005) CD3- but not CD31+ cardiac side population cells exhibit functio- nal cardiomyogenic differentiation. Circulation Research, 97, 52-61. doi:10.1161/01.RES.0000173297.53793.fa
[13]
Balsam, L.B., Wagers, A.J., Christensen, J.L., Kofidis, T., Weissman, I.L. and Robbins, R.C. (2004) Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature, 428, 668-673. doi:10.1038/nature02460
[14]
Hatzistergos, K.E., Quevedo, H., Oskouei. B.N., Hu, Q., Feigenbaum, G.S., Margitich, I.S., Mazhari, R., Boyle, A.J., Zambrano, J.P., Rodriguez, J.E., Dulce, R., Pattany, P.M., Valdes, D., Revilla, C., Heldman, A.W., McNiece, I. and Hare, J.M. (2010) Bone marrow mesenchymal stem cells stimulate cardiac stem cell proliferation and differ- entiation. Circulation Research, 107, 913-922. doi:10.1161/CIRCRESAHA.110.222703
[15]
Porrello, E.R., Mahmoud, A.I., Simpson, E., Hill, J.A., Richardson, J.A., Olson, E.N. and Sadek, H.A. (2011) Transient regenerative potential of the neonatal mouse heart. Science, 331, 1078-1080. doi:10.1126/science.1200708
[16]
Conlan, M.J., Rapley, J.W. and Cobb, C.M. (1996) Bio- stimulation of wound healing by low energy laser irradia- tion. Journal of Clinical Periodontology, 23, 492-496. doi:10.1111/j.1600-051X.1996.tb00580.x
[17]
Karu, T. (2007) Ten lectures on basic science of laser photherapy. Prima Books, Gragesberg.
[18]
Karu, T. (2010) Mitochondrial mechanisms of photobio- modulation in context of new data about multiple roles of ATP. Photomedicine and Laser Surgery, 28, 159-160. doi:10.1089/pho.2010.2789
[19]
Bibikova, A. and Oron, U. (1993) Promotion of muscle regeneration in the toad (Bufo viridis) gastrocnemius mu- scle by low energy laser irradiation. Anatomical Record, 235, 374-380. doi:10.1002/ar.1092350306
[20]
Bibikova, A., Belkin, A. and Oron, U. (1994) Enhancement of angiogenesis in regenerating gastrocnemius muscle of the toad (Bufo viridis) by low energy laser irradiation. Anatomy and Embryology, 190, 597-602. doi:10.1007/BF00190110
[21]
Oron, U. (2006) Photoengineering of tissue repair in ske- letal and cardiac muscles. Photomedicine and Laser Sur- gery, 24, 111-120. doi:10.1089/pho.2006.24.111
[22]
Yaakobi, T., Shoshani, Y., Levkovitz, S., Rubin, O., Ben- Haim, S.A. and Oron, U. (2001) Long term effect of low energy laser irradiation on infarction and reperfusion in- jury in the rat heart. Journal of Applied Physiology, 90, 2411-2441.
[23]
Oron, U., Yaakobi, T., Oron, A., Hayam, G., Gepstein, L., Wolf, T., Rubin, O. and Ben Haim, S.A. (2001a) Attenu- ation of the formation of scar tissue in rats and dogs post myocardial infarction by low energy laser irradiation. La- sers in Surgery and Medicine, 28, 204-211. doi:10.1002/lsm.1039
[24]
Oron, U., Yaakobi, T., Oron, A., Mordechovitz, D., Shof- ti, R., Hayam, G., Dror, U., Gepstein, L., Wolf, T., Hau- denschild, C. and Ben Haim, S.A. (2001b) Low energy la- ser irradiation reduces formation of scar tissue following myocardial infarction in dogs. Circulation, 103, 296-301.doi:10.1161/01.CIR.103.2.296
[25]
Tuby, H., Maltz, L. and Oron, U. (2006) Modulations of VEGF and iNOS in the rat heart by low energy laser irra- diation are associated with cardioprotection and enhanced angiogenesis. Lasers in Surgery and Medicine, 38, 682- 688. doi:10.1002/lsm.20377
[26]
Oron, U., Ilic, S., De Taboada, L. and Streeter, J. (2007) Ga-As (808 nm) laser irradiation enhance ATP produc- tion in human neuronal cells in culture. Photomedicine and Laser Surgery, 25, 180-182. doi:10.1089/pho.2007.2064
[27]
Tuby, H., Maltz, L. and Oron, U. (2007) Low-level laser irradiation (LLLI) promotes proliferation of mesenchy- mal and cardiac stem cells in culture. Lasers in Surgery and Medicine, 39, 373-378. doi:10.1002/lsm.20492
[28]
Li, W.T., Leu, Y.C. and Wu, J.L. (2010) Red-light light- emitting diode irradiation increases the proliferation and osteogenic differentiation of rat bone marrow mesenchy- mal stem cells. Photomedicine and Laser Surgery, 28, S-157-S-165. doi:10.1089/pho.2009.2540
[29]
Mvula, B., Moore, T.J. and Abrahamse, H. (2010) Effect of low-level laser irradiation and epidermal growth factor on adult human adipose-derived stem cells. Lasers in Medical Science, 25, 33-39. doi:10.1007/s10103-008-0636-1
[30]
Tuby, H., Maltz, L. and Oron, U. (2011) Induction of au- tologous mesenchymal stem cells in the bone marrow by low-level laser therapy has profound beneficial effects on the infarcted rat heart. Lasers in Surgery and Medicine, 43, 401-409. doi:10.1002/lsm.21063
[31]
Oron, U. (2011) Light therapy and stem cells: A thera- peutic intervention of the future. Journal of Interventio- nal Cardiology, 3, 627-629.
[32]
Toma, C., Pittenger, M.F., Cahill, K.S., Byrne, B.J. and Kessler, P.D. (2002) Human mesenchymal stem cells dif- ferentiate to a cardiomyocyte phenotype in the adult mu- rine heart. Circulation, 105, 93-98. doi:10.1161/hc0102.101442
[33]
Oron, U. and Mandelberg, M. (1985) Focal regeneration in the rat myocardium following cold injury. Cell Tissue Research, 241, 459-463. doi:10.1007/BF00217194
[34]
Mummery, C.L., Davis, R.P. and Krieger, J.E. (2010) Challenges in using stem cells for cardiac repair. Science Translational Medicine, 14, 1-5.
[35]
van Amerongen, M.J., Harmsen, M.C., van Rooijen, N., Petersen, A.H. and van Luyn, M.J. (2007) Macrophage dep- letion impairs wound healing and increases left ventricu- lar remodeling after myocardial injury in mice. American Journal of Pathology, 170, 1093-1103. doi:10.2353/ajpath.2007.060547
[36]
Okazaki, T., Ebihara, S., Asada, M., Yamanda, S., Saijo, Y., Shiraishi, Y., Ebihara, T., Niu, K., Mei, H., Arai, H. and Yambe, T. (2007) Macrophage colony-stimulating factor improves cardiac function after ischemic injury by induc- ing vascular endothelial growth factor production and sur- vival of cardiomyocytes. American Journal of Pathology, 171, 1093-1103. doi:10.2353/ajpath.2007.061191
Original Source: http://www.scirp.org/journal/jbise
Influence of Low Level Laser Radiation on Migration of Stem Cells
Levon Gasparyan, Grigory Brill, Anu Makela - (Publication) 4468
This study showed a 26% increase in stem cell when they uses red and IR lasers continuous wave.
View Resource
Abstract: The long term effects of low level laser therapy can involve treatment mechanisms connected with activation of stem cells.
In the current study migration of stem cells was tested under the influence of laser light alone as well as in case of combined influence of light and stromal cell-derived factor-1α (SDF-1α). This cytokine plays a role in lymphocyte trafficking, hematopoietic progenitor cell and stem cell homing.
To investigate the light influence on stem cells, we analyzed factor-dependent cell-Patersen (FDCP)-mix multipotent progenitor cells.
Migration of the stem cell line was tested using Transwell system (Corning, NY) under influence of red diode laser (λ=659.6 nm, 19.5 mW) or infrared diode laser (λ=958 nm, 36 mW) during 15 min at continuous wave, as well as in case of applying 150 ng/ml SDF-1α.
Group 1 cells were a group of control, group 2 cells received only red light irradiation, while group 3 cells had IR light irradiation. Group 4 cells were treated with 150 ng/ml SDF-1α. Group 5 cells were irradiated with red laser light in addition to 150 ng/ml SDF-1α, and group 6 cells by IR light and 150 ng/ml SDF-1α.
The count of migrated cells was 1496,5±409 (100%) in case of control. Red and IR laser light increased migration activity of stem cells up to 1892±283 (126%) and 2255,5±510 (151%) accordingly. Influence of SDF-1α was more significant, than effects of light irradiation alone 3365,5±489 (225%). Combined effects of light irradiation and SDF-1α were significantly stronger 5813±1199 (388%) for SDF-1α and red laser light, and 6391,5±540 (427%) for SDF-1α and IR laser light irradiation.
Preliminary study results showed that laser light irradiation can activate stem cell migration in vitro. The results are more reliable in the case of combined application of light and SDF-1α. These results are giving ground to consider that stem cell reactions to light irradiation can be one of the factors of light therapy.
Key words: low level laser irradiation, low level laser therapy, stem cells, SDF-1, stromal cell-derived factor-1
INTRODUCTION
More than 30 years ago first reports about biological effects of low doses of laser light were presented. Currently low level laser therapy (LLLT) is successfully applied in the treatment of numerous diseases and pathological conditions. LLLT exhibits positive effects for the treatment of disorders, having in common failure of blood supply with development of acute or chronic tissue hypoxia, different level of destruction of tissues, following decreased regenerative abilities of tissues and organs, defects in immune system, and altered cell metabolism. At the same time some important mechanisms of influence of laser light on the body are still far to be fully understood [1 - 8].
Recent studies discovered important role of bone marrow hematopoetic stem cell (HSCs) for naturally occurred recovery and regeneration processes, following tissue hypoxia and injury. The three clinically important steps in this natural process are mobilization of stem cells from the bone marrow, homing of these cells to the site of injury, and differentiation of the stem cell into a functional cell of the injured tissue [9]. Different methods of stem cell therapy, the treatment method, based on mobilization and transplantation of stem cells, proves to be effective method of therapy for different disorders.
We proposed a hypothesis that wide range of positive effects following laser therapy can be connected to increased activity of stem cells in damaged tissues. To test that, we examined in vitro the influence of laser light on migration of stem cells in absence and in presence of stromal cell-derived factor-1 (SDF-1), a potent chemoattractor for lymphocytes, monocytes, HSCs, which plays a critical role in the stem cell migration towards areas of tissue injury and hypoxia.
MATERIALS AND METHODS
To investigate the light influence on stem cells, we analyzed factor-dependent cell-Patersen (FDCP)-mix multipotent progenitor cells. The FDCP-mix stem cell line was maintained in ISCOVE’S medium supplemented with 20% horse serum and penicillin/streptomycin in the presence of 20 ng/ml IL-3. The cells were supplied with fresh medium each 5 days. Migration of the stem cell line was tested using Transwell system (Corning, NY). The cells were washed with PBS once and re-suspended in the medium containing 0.1% BSA (2x106/ml). Then, 600 μl of the mixture was irradiated by red diode laser (λ=659.6 nm, 19.5 mW) or infrared diode laser (λ=958 nm, 36 mW) during 15 min at continuous wave. Next, 100 μl of the mixture (2x105 cells) was seeded into upper chambers of the Transwell system, and the filters were placed into the wells containing 600? μl of the medium with or without 150 ng/ml SDF-1α. The plate was incubated for 4 h (37°C, 5% CO2, humidified atmosphere), after which the cells were collected and counted by a FACS sorter (Beckton Dickinson) during 1 min. All samples were performed in duplicate.
Group 1 cells are control group, group 2 cells received only red light irradiation, while group 3 cells – only IR light irradiation. Group 4 cells were treated with 150 ng/ml SDF-1α. Group 5 cells were irradiated with red laser light in addition to 150 ng/ml SDF-1α, and group 6 cells – IR light and 150 ng/ml SDF-1α.
RESULTS
Small amount of stem cells can migrate without SDF-1α or laser light influence. The count of migrated cells in control group was 1496,5±409 (Fig). This amount was considered as 100%. Red and IR laser light at the above mentioned dosage and methods of irradiation increased migration activity of stem cells up to 1892±283 (126%) and 2255,5±510 (151%) accordingly. Influence of SDF-1α was more noticeable, than effects of red or IR laser light irradiation alone - 3365,5±489 (225%). It is important to stress attention on the finding, that rate of stem cell migration towards the filter and SDF-1α containing medium was much higher after laser irradiation of cells - 5813±1199 (388%) for red laser light, and 6391,5±540 (427%) for IR laser light irradiation.
DISCUSSION
The main scientific result of this study is the fact, that red and infrared laser light irradiation can activate migration of stem cells in vitro. Moreover, red and IR laser radiation can up-regulate the rate of stem cell migration towards higher SDF-1α gradient.
How to explain the direct effects of mobility of stem cells in vitro under red and IR laser light irradiation, and use this fact for better understanding the wide range of therapeutic effects of laser therapy?
Modern medical science has accepted that every pathologic condition or disease should be treated according to its clinical stage and symptoms, considering its pathogenesis and etiology. Similar treatment methods can be applied only for the treatment of different diseases, having common pathogenesis.
Not very many examples of successful application of the similar or close therapy method for the treatment of different pathologies are known in modern medicine. Steroid hormone therapy is one of such cases.
Another illustration of successful application of the similar treatment techniques for treatment of different disorders is stem cell therapy, a novel treatment method, which is still under development. Growing data suggests, that transplanted stem cell can successfully and for long period of time improve heart myocardial contractility and other heart functions after myocardial infarction, can support neoangiogenesis in areas of tissue infarction and damage, can replace several cell types in tissues, including β-cells in diabetes models, neurons, cardiomyocytes, hematopoetic cells of different lineages and so on, as well as be useful in the treatment of atherosclerosis [9].
The main principle of stem cell therapy is the idea of replacement of damaged and dead cells in injured tissues and organs with new healthy ones. It is known, that severe stress, tissue hypoxia and damage mobilizes some hematopoetic stem cells (HSCs) from bone marrow to peripheral bloodstream. After that HSCs can migrate towards hypoxic tissues and reach them. Finally they can start to proliferate to the cells types, typical for that damaged tissues. HSCs in the tissues are also able to produce several cytokines, chemokines, growthfactors, improve survival of damaged cells and limit apoptosis. As a result of some tissue regeneration, improvement in the function of a damaged organ can be achieved. Similar and even stronger regeneration and treatment effects can be displayed after transplantation of fetal or adult HSCs to recipient [10-12].
Low laser light irradiation is one other example of application of the same factor for the treatment of number of disorders, which, at first glance, have nothing or very little in common in their pathogenesis. Laser light can accelerate wound and burn healing, improve condition of patients after myocardial infarction and stroke, can support hematopoiesis of bone marrow after X-ray radiation or during cancer chemotherapy, can help for the treatment of diabetic angiopathy and neuropathy, as well as reduce atherosclerotic plaque formation. In cellular and tissue level LLLT exhibits positive effects for the treatment of disorders, having in common failure of blood supply with development of acute or chronic tissue hypoxia, different level of destruction of tissues, following with decreased regenerative abilities of cells, as well as altered cell metabolism [6, 7, 13, 14].
One can see that the therapeutic applications of LLLT and stem cell therapy are very close. So, earlier we proposed the hypotheses that one of the mechanisms of light therapy includes acceleration of tissue repair due to better mobilization of stem cells to the spot of injury after laser light irradiation [15]. That process should include several phases, including activation of stem cell migration towards area of tissue damage and hypoxia.
Stem cells are being investigated for their potential use in regenerative medicine. Stem cells share the following two defining characteristics: the capacity to differentiate into a spectrum of different cell types and the capacity to renew themselves [16]. The biological principle that underlies stem cell therapy is tissue-directed differentiation. For example, adult stem cells isolated from liver tissue and re-injected into liver become hepatocytes, whereas the same cells injected into myocardium become myocytes. [17] Stem cells have been engrafted into a broad spectrum of tissues, including regenerating bone, neural tissue, dystrophic skeletal muscle, and injured skeletal muscle. [18]. Myocardial regeneration is perhaps the most widely studied and debated example of stem cell plasticity. The most promising results have been obtained after transplantation and mobilization of bone marrow cells to the area of infarction.
The three clinically important steps in this natural process are mobilization of stem cells from the bone marrow, homing of these cells to the site of injury, and differentiation of the stem cell into a functional cell of the injured tissue [19].
Stem cell repair of cardiac and vascular tissue is a naturally occurring process after injury [20, 21] Circulating CD34+ mononuclear cell counts and plasma levels of endothelial growth factor are significantly increased in patients with acute myocardial infarction, peaking on day 7 after onset [22]. Due to limitations of the naturally occurring repair process after myocardium infarction and other injuries or pathologies several stem cell transplantation strategies were proposed and tested.
At present, however, enthusiasm for the therapeutic potential of strategies of stem cell transplantation is limited by certain practical considerations. For example, the number of stem cells, required for injection for the treatment of myocardial infarction, can be harvested approximately from 6 l of donor blood [23].
Other important limitation for autologous bone marrow stem/progenitor cell mobilization is a recent finding, that circulating endothelial progenitor cells in patients with coronary heart disease are impaired with respect to number and functional activity. Moreover, Heeschen et al [24] reported that regeneration and functional ability of bone marrow-derived mononuclear cells (BM-MNCs) in patients with chronic ischemic cardiomyopathy (ICMP) are also limited. In spite of the fact that, the number of BM-MNCs isolated from bone marrow aspirates of 18 patients with ICMP and 8 healthy subjects s did not differ, the colony-forming capacity of BM-MNCs from patients with ICMP was significantly lower compared with BM-MNCs from healthy controls. Likewise, the migratory response to SDF-1 and vascular endothelial growth factor (VEGF) was significantly reduced in BM-MNCs derived from patients with ICMP compared with BM-MNCs from healthy controls. The reduced neovascularization capacity in vivo of BM-MNCs derived from patients with ICMP closely correlated with the in vitro assessment of SDF-1-induced migration and colony-forming capacity.
The need for development of new methods for mobilization, as well as for homing of stem cells to the site of injury is therefore evident.
Several growth factors, chemokines and cytokines are involved in the regulation of stem cell mobilization, homing and differentiation. Stromal cell-derived factor-1 (SDF-1) is one of them. SDF-1 is a chemokine playing an important role in the trafficking of hematopoietic stem cells. SDF-1 is expressed on stromal cells of various tissues. CXCR4 is the only known receptor for SDF-1 [25]. SDF-1/CXCR4 interaction is reported to play an important physiological role during embryogenesis in hematopoiesis, vascular development, cardiogenesis, and cerebellar development [26-28].
Recently, several investigators have reported that CD34+ cells, classically considered to be hematopoietic stem cells, expressed CXCR4, and that SDF-1 could induce CD34+ cell migration in vitro [29]. Accordingly, SDF-1 is considered as one of the key regulators of hematopoietic stem cell trafficking between the peripheral circulation and bone marrow. SDF-1 has also been shown to effect CD34+ cell proliferation and mobilization and to induce angiogenesis in vivo [30 -32].
Hattori et al [31] reported that plasma elevation of SDF-1 induced mobilization of mature and immature hematopoietic progenitors and stem cells, including endothelial progenitor cells (EPCs). However, application of granulocyte colony-stimulating factor (G-CSF) for stem cell mobilization is widely accepted nowadays.
Yamaguchi et al [23] studied the effects of SDF-1 on migration and accumulation of EPCs. SDF-1 induced EPCs migration in a dose dependent manner in vitro. The magnitude of migration was similar to that induced by VEGF. Authors also reported that locally (in hind-limb ischemic muscle of experimental animals) administered SDF-1 could augment the local accumulation of transplanted EPCs from peripheral blood, thereby resulting in enhanced neovascularization. As a result, cell transplantation not only improved neovascularization but also reduced adverse biological consequences such as limb necrosis and auto-amputation in the mouse ischemic hind-limb model. These studies also disclosed that systemic EPCs transplantation improved myocardial neovascularization and cardiac function corresponding to reduced left ventricular scarring. Authors concluded that, at least under the experimental conditions used in the study, the effect of SDF-1 on neovascularization appears to result primarily from its ability to enhance the recruitment and incorporation of transplanted EPCs.
Damas at al. [33] reported that SDF-1α, at least in high concentrations, may mediate anti-inflammatory and matrix-stabilizing effects in unstable angina. These effects may promote plaque stabilization, and therapeutic intervention that enhances SDF-1 α activity could potentially be beneficial in acute coronary syndromes. Authors demonstrated significantly altered SDF-1/CXCR4 expression in patients with angina, with particularly marked changes in those with unstable disease, with low SDF-1 levels in plasma and altered expression of its corresponding receptor on peripheral blood mononuclear cells (PBMC). In contrast to the raised plasma levels of inflammatory chemokines in patients with angina plasma levels of SDF-1 and the surface expression of its corresponding receptor (CXCR4) on PBMC appear to be down-regulated in these patients. Thus, although persistent inflammation may involve up-regulation of inflammatory chemokines, recent studies suggest that inflammatory cytokines (eg, TNF-α and IL-1) may decrease the expression of SDF-1 and CXCR4.
Future progress of stem therapy techniques probably will include development of incubation methods for enhancement stem cell mobility and homing ability, as well as for faster proliferation into desire tissue cells. Increasing migration abilities will help to achieve better and faster results.
The ability of laser light to activate migration and mobility of different cells is well known. It was noticed, that irradiation of sperm cells in vitro can increase their mobility and fertility [34]. Moreover, this effect is more pronounced in case of damaged cells with low mobility rate. This gives a ground to assume that laser light irradiation in certain dosage and condition can improve functional abilities of cells. Future experiments are required to ascertain if stem cells respond to the laser light the same way.
The main finding on this study is that red and IR laser light can stimulate stem cell migration in vitro, and especially increase migration towards SDF-1α gradient. Stem cell ability to migrate towards tissues with higher SDF-1 concentration is one of the key mechanisms of stem cell homing. These results are giving ground to speculate that activation of stem cell migration can be one of the mechanisms of low level laser therapy. Taking into consideration that the combined of SDF-1 and laser irradiation had the strongest effect on stem cell homing, it would be reasonable to assume that this combination could be used in not only increasing the activity of stem cells but also in determining the main area of stem cell mobilization and homing. The current study did not aim to study the mechanisms of increased migration ability, which will be study in the future. But it is possible to suggest following explanation: laser irradiation can change the metabolism of stem cells, increase ATP production and so increase the migration, as well as up-regulate CXCR4 receptor expression or syntheses de novo. More studies are required to test if the laser light irradiation in vivo is able to make homing of transplanted stem cells to the area of damage more efficient, to check the influence of laser light on the mobilization rate of stem cells from bone marrow, to investigate if laser light can enhance functional abilities of stem cells. These studies would be desirable for better understanding of the mechanisms of laser therapy and for development of more effective methods of stem cell therapy.
References
1. Tuner J. and Hode L. Low Level Laser Therapy: Clinical Practice and Scientific Background, Prima Books, Grängesberg, Sweden, 1999.
2. Karu T. The Science of Low Power Laser Therapy, Gordon & Breach, London, 1998.
3. Baxter G.D. Therapeutic Lasers: Theory and Practice, Churchill Livingstone, London, 1994.
4. Simunovic Z., Ed. Lasers in Medicine and Dentistry, Vitgraf, Rijeka (Croatia), 2000.
5. Zhukov B.N. and Lysov N.A. Laser irradiation in experimental and clinical angiology (in Russian), Samara (Russia), 1996.
6. Kozlov V.I., et al. Bases of laser physio- and reflexo-therapy (in Russian), Zdorovje, Samara (Russia), 1993.
7. Paleev N.R. Ed. Phototherapy (in Russian), Meditsina, Moscow (Russia), 2001.
8. Skobelkin O. K. Ed. Application of low-intensive lasers in clinical practice (in Russian). Moscow, 1997.
9. Forrester J, Price M, Makkar R. Stem Cell Repair of Infarcted Myocardium. An Overview for Clinicians. Circulation. 2003;108:1139–1145.
10. Orlic D., Hill J., Arai A. Stem Cells for Myocardial Regeneration Circulation Research. 2002;91:1092.
11. Hodgson D., Behfar A., Zingman L.V., Kane G.C., Perez-Terzic C., Alekseev A.E., Puceat M., and Terzic A. Stable benefit of embryonic stem cell therapy in myocardial infarction. Am J Physiol Heart Circ Physiol, August 1, 2004; 287(2): H471 - H479.
12. Ozbaran M., Omay S. B., Nalbantgil S., Kultursay H., Kumanlioglu K., Nart D., and Pektok E. Autologous peripheral stem cell transplantation in patients with congestive heart failure due to ischemic heart disease. Eur. J. Cardiothorac. Surg., March 1, 2004; 25(3): 342 - 350.
13. Brill A.G., Shenkman B., Brill G.E. et al. Blood irradiation by He-Ne laser induces a decrease in platelet responses to physiological agonists and an increase in platelet cyclic GMP. Platelets. 2000. Vol. 11. P. 87-93.
14. Mester A. Biostimulative effect in wound healing by continuous wave 820 nm laser diode. Lasers in Med Science, abstract issue July 1988, No. 289.
15. Gasparyan L.V. Stem cells and therapeutic effect of light irradiation (in Russian). Collection of abstracts of the 10th International Conference of Quantum Medicine, Moscow, 2003, pp. 43-44.
16. Graf T. Differentiation plasticity of hematopoietic cells. Blood. 2002;99:3089–3101.
17. Malouf NN, Coleman WB, Girsham JW, et al. Adult-derived stem cells from the liver become myocytes in the heart in vivo. Am J Pathol. 2001;158:1929–1935.
18. Donovan PJ, Gearhart J. The end of the beginning for pluripotent stem cells. Nature. 2001;414:92–97.
19. Forrester J, Price M, Makkar R. Stem Cell Repair of Infarcted Myocardium. An Overview for Clinicians. Circulation. 2003;108:1139–1145.
20. Beltrami AP, Urbanek K, Kajstura J, et al. Evidence that human cardiac myocytes divide after myocardial infarction. N Engl J Med. 2001;344:1750–1757.
21. Gill M, Dias S, Hattori K, et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res. 2001;88:167–174.
22. Shintani S, Murohara T, Ikeda H, et al. Mobilization of endothelial progenitor cells in patients with acute myocardial infarction. Circulation. 2001;103:2776–2779.
23. Yamaguchi J, Kusano K, Masuo O, at al. Stromal Cell–Derived Factor-1 Effects on Ex Vivo Expanded Endothelial Progenitor Cell Recruitment for Ischemic Neovascularization. Circulation. 2003;107: 1322–1328.
24. Heeschen C, Lehmann R, Honold J, Assmus B, Aicher A, Walter DH, Martin H, Zeiher AM, Dimmeler S. Profoundly reduced neovascularization capacity of bone marrow mononuclear cells derived from patients with chronic ischemic heart disease. Circulation. 2004;109(13):1615-22.
25. Bleul CC, Farzan M, Choe H, et al. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature. 1996;382:829–833.
26. Nagasawa T, Hirota S, Tachibana K, et al. Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature. 1996;382:635–638.
27. Tachibana K, Hirota S, Iizasa H, et al. The chemokine receptor CXCR4 is essential for vascularization of the gastrointestinal tract. Nature. 1998; 393:591–594.
28. Zou YR, Kottmann AH, Kuroda M, et al. Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature. 1998;393:595–599.
29. Mohle R, Bautz F, Rafii S, et al. The chemokine receptor CXCR-4 is expressed on CD34+ hematopoietic progenitors and leukemic cells and mediates transendothelial migration induced by stromal cell-derived factor-1. Blood. 1998;91:4523–4530.
30. Lataillade JJ, Clay D, Dupuy C, et al. Chemokine SDF-1 enhances circulating CD34+ cell proliferation in synergy with cytokines: possible role in progenitor survival. Blood. 2000;95:756–768.
31. Hattori K, Heissig B, Tashiro K, et al. Plasma elevation of stromal cell-derived factor-1 induces mobilization of mature and immature hematopoietic progenitor and stem cells. Blood. 2001;97:3354–3360.
32. Salcedo R, Wasserman K, Young HA, et al. Vascular endothelial growth factor and basic fibroblast growth factor induce expression of CXCR4 on human endothelial cells: in vivo neovascularization induced by stromal derived factor-1α. Am J Pathol. 1999;154:1125–1135.
33. Damas J, Wæhre T, Yndestad A, et al. Stromal Cell–Derived Factor-1a in Unstable Angina. Circulation. 2002;106:36-42.
34. Pyrikova S.I et al. Effect of laser exposure on human seminal fluid (in Russian). Clinical and laboratory diagnosis. 1998;5:15-16.
Laser-induced regeneration of cartilage
Emil N. Sobol, Olga I. Baum, Anatoliy B. Shekhter, Anna Guller, Andrey V. Baskov - J. of Biomedical Optics, 16(8), 080902 (2011) (Publication) 4501
This article shows results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine
View Resource
Abstract
Laser radiation provides a means to control the fields of temperature and thermo mechanical stress, mass transfer, and modification of fine structure of the cartilage matrix. The aim of this outlook paper is to review physical and biological aspects of laser-induced regeneration of cartilage and to discuss the possibilities and prospects of its clinical applications. The problems and the pathways of tissue regeneration, the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches for cartilage repair; consider possible mechanisms of laser-induced regeneration. Finally, we present the results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine.
1.
Introduction
Cartilage is a kind of highly specialized connective tissue. The structural variety of the cartilage provides its unique biomechanical capacity to bear different kinds of static and dynamic loads over a wide range of intensity. Biological role of cartilage structures stems from their critical significance for growth and development as well as for all kinds of body movements. The exceptional importance of cartilage elements for individual survival is, probably, due to mechanisms of natural selection, resulting in limited reparative potential of this tissue. Scanty cellular sources and low metabolic rate along with avascularity of cartilage contribute to its decreased regeneration ability. As a result of these strong limitations, the injuries of cartilage caused by inflammation, traumas, degeneration, and aging usually become chronic and recalcitrant to any kind of medical treatment. In the USA, according to tentative estimations, the prevalence of all forms of arthritis has been calculated in order of 40 million people; and the annual medical care costs were about 65 billion USD.1Degenerative spine diseases are a major cause of back pain that deteriorates the quality of life of patients and often leads to disability. Direct and indirect medical expenses are estimated as more than 90 billion per year.2
High prevalence and incidence, as well as the social and economic significance of cartilage pathology, attract great interest to this problem. Considerable efforts have been devoted to study various approaches to restore cartilage structures and to stimulate intrinsic capabilities of the tissue to regeneration. There are several treatment modalities of cartilage restoration suggested for clinical use (see Ref. 3 and referred literature): 1. surgical techniques; 2. controllable cell delivery to the lesion; and 3. tissue engineering applications of biodegradable materials (scaffolds) with cell-seeding and modification of cartilage reparative response by different growth factors and cytokines. Although there is a wealth of information regarding the substitution of lost cartilage by the mentioned approaches, the problem of cartilage repair is still unsolved. The long term results show no completed cartilage regeneration; in many cases, the new growing tissue materially differs from the well organized original cartilage. The reasons of insufficient cartilage reparation are connected with its structural and functional organization and with the difficulties of the precise control of the external physical and chemical effects.4, 5 Regeneration of cartilage may be realized in accordance with the natural genetic program of the cells. The efficacy of any approach aimed to control the regeneration process depends on the solution of three tasks: 1. the ability to reproduce the normal cell differentiation sequence from the progenitor cells to mature chondrocytes, 2. stimulation of the specific subpopulations of the resident cells to proliferation and/or new matrix production, and (c) achievement of adequate spatial organization of the new growing tissue. Probably, the most important feature of the laser-based treatment is the involvement and activation of the intrinsic mechanisms of cartilage repair. Many papers are devoted to the effect of low-intensive lasers on cartilage functional state and reparative ability. However, the effectiveness, as well as the placebo-versus-treatment ratio for low level laser therapy, is still under considerable dispute. A more detailed discussion of this issue may be found elsewhere.6 This paper is mainly limited with a consideration of the effect of nonablative laser radiation on the cartilaginous cells through their matrix microenvironment to provide natural and optimal conditions for regeneration. Wide ranges of wavelengths, precise localization of the irradiated area, and temporal and spatial modulation of laser radiation are the main advantages of the laser technologies, which may result in specific tissue response. In particular, the laser-induced modification of the cartilage extracellular matrix (ECM) seems to be of great significance in view of some new data on the developmental roles of the matrix molecules and mechanical loads. Although the evidence of laser irradiation morphogenetic effects is still largely circumstantial, we consider the available observations to address some possible perspectives of the controlled regeneration of cartilage using nonablative laser treatment. So, the aim of this paper is to review physical and biological aspects of laser-induced regeneration of cartilage, to discuss the possibilities and prospects of its clinical applications. The problems and the ways of tissue regeneration and the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches to cartilage repair, consider possible mechanisms of laser-induced regeneration, present the results in laser regeneration of joints and spine disks cartilages, and finally, discuss some future medical applications of laser regeneration.
2.
Cartilage as a Subject of Regeneration
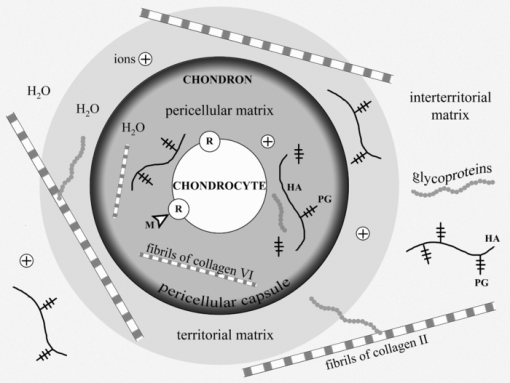
There are a number of detailed reviews describing the structure and vital functions of cartilages.3, 7, 8 The main components of cartilage are cells (chondrocytes) and ECM consisting of water (70 to 80%), collagens, proteoglycans (PGs), hyaluronic acid (HA), and glycoproteins (GP). The PGs consist of glycosaminoglycanes (chondroitin sulphate and keratan sulphate) linked to the core-protein, which, in turn, is bound with HA threads interweaving between collagen fibrils (Fig. 1). PGs have a lot of negative charged groups; and the electrical neutrality of cartilage is due to the presence of positive ions (K+, Na+, H+, Ca2+, Mg2+). There are three types of cartilage tissue: hyaline cartilage (costal, nasal septum, articular cartilage of the joints), fibrous cartilage (annulus fibrosis of the spine disks, Eustachian tube), and elastic cartilage (auricle, epiglottis). Hyaline cartilage first forms in embryos and later transforms into other types of cartilage and bone tissues. The distinguishing features of the ECM of hyaline cartilage are having a very high content of glycosaminoglycanes and the prevalence of collagen type II fibrils.9, 10, 11, 12, 13 Fibrous cartilage is characterized by predominance of collagen type I.14, 15, 16 Matrix of the elastic cartilage possesses elastic fibers. Nasal and some other cartilages are covered with a perichondrium playing an important role in nutrition and growth of the avascular tissue. Articular cartilage has no perichondrium; it gets nutrition from synovial liquid and subchondral bone. An articular cartilage surface is covered by a cell-free lamina splendens (LS) consisting mainly of the HA and phospholipids.17 An important structural and metabolic unit of articular cartilage is a chondron.11 It includes a chondrocyte and its pericellular matrix (PM) bordered with a pericellular capsule (PC). The chondron is surrounded by territorial and interterritorial matrices. The chondrons and their matrix environment have different mechanical properties.3, 11 The PM is enriched with HA, sulphated PGs, biglycan, and GPs, including link protein and laminin. The PC is predominantly composed of compact thin fibrils of collagen type VI and fibronectin. It is suggested that the PM and PC provide hydrodynamic protection for the chondrocyte against pressure loading and take a part in control of spatial and temporal distribution of newly synthesized macromolecules as well as in the cell-matrix interaction.11 Territorial and interterritorial matrices are characterized by different degrees of the PGs maturity and with a different proportion of the chondroitin sulphate and keratan sulphate. The heteropolymeric fibrils of collagen types II, IX, and XI (HCF) emerging in the territorial matrice become the major load-bearing element in the interterritorial matrice.16 These fibrils are in charge of the tissue protection against multidirectional tensions.
Fig. 1
Cartilage components and structure. PG –proteolycanes; HA – hyaluronic acid, GP – glycoproteins; M – morphogenes; R -molecular receptors of chondrocyte's membrane; (+) ions (K+, Na+, H+, Ca2+, Mg2+).
A number of molecules that possess signal roles in morphogenetic processes, including chondrogenesis from embryonic development to regeneration, may interact with the receptors of the cellular membrane of chondrocyte. Binding of such morphogenes to the membrane receptors triggers various intracellular signaling cascades to result in regulation of the expression of genes. Hydrostatic pressures and fluid flows as well as multidirectional tensions contribute to tissue water displacement leading to changes of local concentrations of ions and morphogens. The GP molecules (integrins, fibronectin, laminin, etc.) distributing over the ECM serve as important mediators of the signaling molecules. They play an important role in the cell-matrix interactions and operate on the growth of cartilage tissue.
Hyaline cartilage has a zonal structure:11, 18, 19 the superficial layer contains fibroblast-like chondrocytes of type I. It is characterized by a decreased level of the PG aggregates (aggrecanes) and by a high content of small leucine-rich PGs (decorin and biglycan). The cells in the middle layer are chondrocytes of type II. They form multicellular clones and keep a certain ability of proliferation. A smaller subpopulation of the middle layer cells is presented by the chondrocytes type III covered with lacunas. These nonproliferating cells are also presented in the deep layer of cartilage. Type IV cells belong to a degrading cell group. Chondrocytes synthesize and degrade all components of cartilage matrix through specialized enzymes (prolyl hydroxylase, lysil oxidase, collagenases, aggrecanases etc.).16, 20 Metabolic activity of the chondrocytes in cartilage is controlled by hormones, various cytokines, growth factors, and vitamins (A, C, and D).21, 22, 23, 24 Ultimately, the biosynthetic and catabolic activities of cartilage cells, as well as the kinetics of the cellular population are governed by the local concentrations of the humoral and insoluble morphogens near the external membranes of chondrocytes.
The main mechanism of cartilage nutrition is diffusion of water carrying low-molecular substances (ions, glucose, amino acids, etc.). As the chondrocytes kinetics are under conditions of hypoxia, their metabolism is generally realized by the anaerobic glycolysis pathway. That, in combination with the chondrocytes paucity, determines a low level of cartilage metabolism. Half life period is three or four years for aggrecans, and about 10 years for collagen.25 All types of cartilage, especially articular cartage and intervertebral disks, have low repair potential. There is a lot of literature on this topic. 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37 Extra-articular cartilage is usually repaired by the means of proliferation and chondrogenic differentiation of the perichondrial cells. The defects of hyaline cartilage and the extensive defects of costal and auricular cartilages are usually filled up with fibrous connective tissue or fibrous cartilage, which both do not have adequate functional properties; that determines persistent attempts to find new possibilities for cartilage regeneration. The healing of cartilage defects can be improved with mechanical stimulation, intra-articular application of HA, hormone therapy,38, 39, 40, 41 and also with the use of osteochondral or cartilaginous implants, in particular together with cultivated chondrocytes.42, 43 One of the current leading approaches is in vitro growth of the tissue engineering constructs followed with their implantation into cartilage lesion. Autologous chondrocyte implantation (ACI) resulted in the formation of hyaline-like tissue with a quite stable clinical outcome.44, 45 But according to the histological data, only 39% of the defects treated with ACI were filled with hyaline cartilage, while 43% were filled with fibrocartilage, and 18% did not show any healing response at all.46
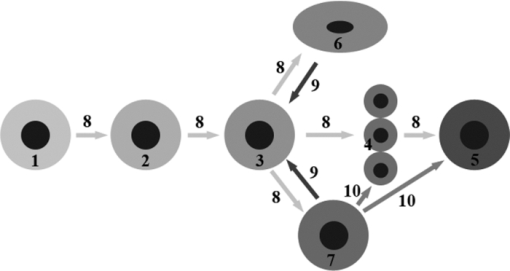
Regeneration process is associated with embryonic chondrogenesis mechanisms and partial dedifferentiation of mature cells. Figure 2 shows possible pathways of regeneration-related dedifferentiation of the cells in cartilage. Mesenchymal stem cells (MSC) can differentiate into cartilage cells of various types, including immature and mature chondrocytes, and notochordal and chondrocyte-like cells of the intervertebral disks. These processes are under multilevel control of signaling molecules and mechanical factors. Our main hypothesis is that differentiation and dedifferentiation of cartilage cells, as well as their metabolic activity, may be controlled by direct action of laser radiation on the cells and through laser-induced modification of the ECM.
Fig. 2
Differentiation of cartilage cells and possible pathways of their regeneration-related dedifferentiation. 1 – MSC, 2 – pre-chondrocytes, 3 – early chondrocytes (chondroblasts), 4 – columnar chondrocytes, 5 – hypertrophic chondrocytes, 6 – chondrocytes of fibrous cartilage, 7 – chondrocytes of hyaline cartilage, 8 – differentiation pathways, 9 – pathways of limited dedifferentiation, 10 – additional pathways of cellular differentiation (following the enchondral osteogenesis differentiation mechanism).
3.
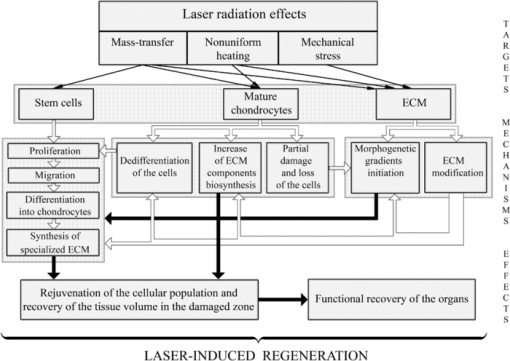
Targets for Laser Effect. Possible Types of Cartilage Response on Laser Radiation
To discuss possible ways of using lasers for cartilage regeneration, it is important to know what effect laser parameters have on (a) different types of the cells; (b) different components of the ECM; (c) signaling molecules produced by the cells and accumulated in the ECM; (d) intercellular and cell-matrix interactions; (e) differentiation and dedifferentiation of the cells, their migration and biosynthesis activity. Feasible pathways promoting cartilage regeneration include: 1. additional cellular supply from bone marrow and blood; 2. biosynthesis amplification of the ECM components, 3. stimulation of the motility of mature chondrocytes, and 4. activation of resident adult stem cells toward their proliferation, differentiation, and ECM production. The main reasons of the low regeneration potential of cartilage are advanced differentiation of the resident chondrocytes and relatively slow metabolism of the tissue. The nonablative laser radiation may provide controllable thermal and mechanical effects (as on the cells, as on the matrix) resulting in activation of the cellular biosynthesis. In particular, nonuniform laser heating of cartilage induces heterogeneous thermal expansion, stress, and also the movement of the interstitial water and ions (see Fig. 3 and Sec. 4).
Fig. 3
Targets and mechanisms of the laser-induced regeneration of cartilage. Thin straight arrows show the direct laser influence on the components of cartilage. White thick arrows represent the most important biological responses to laser action. Dark thick arrows show the processes leading directly to regeneration.
One of the major obstacles for regeneration of cartilage, including partial-thickness defect of articular cartilage, is its avascularity, which hampers the progenitor cell movement from the blood and marrow to the damaged areas of the tissue. Preventing an entry of unspecialized cells and diminishing the rate of cartilage repair that slow regeneration, nevertheless, may have its good point, as it may potentially result in the growth of well organized tissue of the hyaline cartilage. Rapid repair of the full-thickness cartilage defects usually leads to undesirable growth of fibrous connective tissue or fibrous cartilage due to the impact of blood and bone-marrow-derived cells. It can be better understood by the following analogy. It is known that skin wound healing resulting in a fibrous scar is going through emergency regeneration due to swift proliferation of unspecialized fibroblasts. Their sources are the precursor cells coming into the wound via blood. These cells have nonspecific genetic program and form scar.47 In a similar manner, the bone-marrow cells coming to the full-thickness defect of articular cartilage differentiate into the fibroblasts of nonspecific connective tissue or into the chondrocytes of the fibrous cartilage. This provides quick filling of the defect, but fails in functionality of the novel tissue. One of the possible ways to promote growth of the hyaline cartilage in the full-thickness defects of articular cartilage plates can be laser-induced coagulation of the bottom of the defect. This may prevent access of unspecialized precursor cells from the blood or bone-marrow in order to develop more specific, i.e., hyaline cartilage.
It is known that in the course of embryogenesis, the hyaline cartilage forms in the zones undergoing compression load (articular cartilage), whereas, the fibrous cartilage (meniscus, annulus fibrosis of the intervertebral disk) usually develops in the stretched or torsioned zones. Spatial and temporal modulation of laser radiation allows controlling the actual distribution of stretched and compressed zones in cartilage. The mechanical loads are important factors governing an orchestra of chondrogenesis, including the processes of cellular differentiation. Therefore, the nonablative laser treatment may play a triggering role in the differentiation of immature cartilage cells. Laser radiation may probably be responsible for the reverse process of dedifferentiation of the mature chondrocytes leading to the recovery of their ability to divide. Existing natural pathways of cells dedifferentiation (see Fig. 2) open possibilities for tissue correction, in particular, replacement of abnormally grown fibrous tissue by hyaline cartilage possessing adequate mechanical and functional properties (Fig. 3).
Laser radiation can also be used to stimulate proliferation and acquiring the specialized phenotype by resident stem cells or MSC coming through synovial liquid in order to promote their transformation into mature hyaline-like chondrocytes. This approach is critically significant for healing of the partial-thickness defects of articular cartilage. At the same time, as the cellular population in full-thickness cartilage defect is highly heterogenic, laser irradiation may effect the proliferation of different kinds of cells. Thereafter, the additional controlling factor of the ECM architecture should be taken into account. Laser modification of the fine structure of ECM does not change its general organization. This provides natural environments for chondrocytes and promotes restoration of the hyaline type cartilage. One of the important factors is cell movement velocity, which correlates with the alignment of the matrix fibrillar components.48, 49 Nonablative laser irradiation allows structure modification and diffusion properties of ECM. This may support cell movement and favor tissue regeneration.
Laser-induced growth of hyaline cartilage in elastic cartilage was established in the course of in vivoexperiments on laser reshaping of porcine ears.50 The effects of laser irradiation on gene expression of chondrocytes and collagen of ECM have been studied for rabbit septal cartilage using laser settings typical for laser reshaping procedure.51 It was shown that laser irradiation of cartilage does not result in the detection of collagen type I. Only collagen type II was observed after laser irradiation in the corresponding cell culture in vitro. This fact indicates that cartilage cellular response to nonablative laser irradiation differs from the reaction of conventional wound healing. Laser irradiation of cartilage can leave intact collagen and preserve general matrix architecture, which favors chondrocyte survival and promotes new tissue growth. Evidence of hyaline cartilage development in laser-irradiated intervertebral disks was revealed in the animal experiments (see Sec. 5.2). The advantage of the laser effect on chondrocytes proliferation compared to other thermal, mechanical, and chemical effects was demonstrated in Ref. 52. No evidence of chondrocyte DNA replication was observed in tissues heated using nonlaser methods, grown in TGF-β-contained media, or mechanically traumatized. In contrast, for laser irradiated chondrocytes, flow cytometry provided evidence that laser irradiation causes a proliferative response in cho...
Original Source: https://www.spiedigitallibrary.org/journals/journal-of-biomedical-optics/volume-16/issue-08/080902/Laser-induced-regeneration-of-cartilage/10.1117/1.3614565.full?SSO=1
Lasers, stem cells, and COPD
Feng Lin†, Steven F Josephs†, Doru T Alexandrescu†, Famela Ramos, Vladimir Bogin, Vincent Gammill, Constantin A Dasanu, Rosalia De Necochea-Campion, Amit N Patel, Ewa Carrier and David R Koos - 2010 (Publication) 4469
This well annotated paper looks at the biphasic response to try to find the appropriate dosage for several applications including COPD.
View Resource
Lasers, stem cells, and COPD
· Feng Lin†,
· Steven F Josephs†,
· Doru T Alexandrescu†,
· Famela Ramos,
· Vladimir Bogin,
· Vincent Gammill,
· Constantin A Dasanu,
· Rosalia De Necochea-Campion,
· Amit N Patel,
· Ewa Carrier and
· David R KoosEmail author
†Contributed equally
Journal of Translational Medicine20108:16
https://doi.org/10.1186/1479-5876-8-16
© Lin et al; licensee BioMed Central Ltd. 2010
Received: 7 January 2010
Accepted: 16 February 2010
Published: 16 February 2010
Abstract
The medical use of low level laser (LLL) irradiation has been occurring for decades, primarily in the area of tissue healing and inflammatory conditions. Despite little mechanistic knowledge, the concept of a non-invasive, non-thermal intervention that has the potential to modulate regenerative processes is worthy of attention when searching for novel methods of augmenting stem cell-based therapies. Here we discuss the use of LLL irradiation as a "photoceutical" for enhancing production of stem cell growth/chemoattractant factors, stimulation of angiogenesis, and directly augmenting proliferation of stem cells. The combination of LLL together with allogeneic and autologous stem cells, as well as post-mobilization directing of stem cells will be discussed.
Introduction (Personal Perspective)
We came upon the field of low level laser (LLL) therapy by accident. One of our advisors read a press release about a company using this novel technology of specific light wavelengths to treat stroke. Given the possible role of stem cells in post-stroke regeneration, we decided to cautiously investigate. As a background, it should be said that our scientific team has been focusing on the area of cord blood banking and manufacturing of disposables for processing of adipose stem cells for the past 3 years. Our board has been interested in strategically refocusing the company from services-oriented into a more research-focused model. An unbiased exploration into the various degenerative conditions that may be addressed by our existing know-how led us to explore the condition of chronic obstructive pulmonary disease (COPD), an umbrella term covering chronic bronchitis and emphysema, which is the 4th largest cause of death in the United States. As a means of increasing our probability of success in treatment of this condition, the decision was made to develop an adjuvant therapy that would augment stem cell activity. The field of LLL therapy attracted us because it appeared to be relatively unexplored scientific territory for which large amounts of clinical experience exist. Unfortunately, it was difficult to obtain the cohesive "state-of-the-art" description of the molecular/cellular mechanisms of this therapy in reviews that we have searched. Therefore we sought in this mini-review to discuss what we believe to be relevant to investigators attracted by the concept of "regenerative photoceuticals". Before presenting our synthesis of the field, we will begin by describing our rationale for approaching COPD with the autologous stem cell based approaches we are developing.
COPD as an Indication for Stem Cell Therapy
COPD possesses several features making it ideal for stem cell based interventions: a) the quality of life and lack of progress demands the ethical exploration of novel approaches. For example, bone marrow stem cells have been used in over a thousand cardiac patients with some indication of efficacy [1, 2]. Adipose-based stem cell therapies have been successfully used in thousands of race-horses and companion animals without adverse effects [3], as well as numerous clinical trials are ongoing and published human data reports no adverse effects (reviewed in ref [4]). Unfortunately, evaluation of stem cell therapy in COPD has lagged behind other areas of regenerative investigation; b) the underlying cause of COPD appears to be inflammatory and/or immunologically mediated. The destruction of alveolar tissue is associated with T cell reactivity [5, 6], pathological pulmonary macrophage activation [7], and auto-antibody production [8]. Mesenchymal stem cells have been demonstrated to potently suppress autoreactive T cells [9, 10], inhibit macrophage activation [11], and autoantibody responses [12]. Additionally, mesenchymal stem cells can be purified in high concentrations from adipose stromal vascular tissue together with high concentrations of T regulatory cells [4], which in animal models are approximately 100 more potent than peripheral T cells at secreting cytokines therapeutic for COPD such as IL-10 [13, 14]. Additionally, use of adipose derived cells has yielded promising clinical results in autoimmune conditions such as multiple sclerosis [4]; and c) Pulmonary stem cells capable of regenerating damaged parenchymal tissue have been reported [15]. Administration of mesenchymal stem cells into neonatal oxygen-damaged lungs, which results in COPD-like alveoli dysplasia, has been demonstrated to yield improvements in two recent publications [16, 17].
Based on the above rationale for stem cell-based COPD treatments, we began our exploration into this area by performing several preliminary experiments and filing patents covering combination uses of stem cells with various pharmacologically available antiinflammatories, as well as methods of immune modulation. These have served as the basis for two of our pipeline candidates, ENT-111, and ENT-894. As a commercially-oriented organization, we needed to develop a therapeutic candidate that not only has a great potential for efficacy, but also can be easily implemented as part of the standard of care. Our search led us to the area of low level laser (LLL) therapy. From our initial perception as neophytes to this field, the area of LLL therapy has been somewhat of a medical mystery. A pubmed search for "low level laser therapy" yields more than 1700 results, yet before stumbling across this concept, none of us, or our advisors, have ever heard of this area of medicine.
On face value, this field appeared to be somewhat of a panacea: clinical trials claiming efficacy for conditions ranging from alcoholism [18], to sinusitis [19], to ischemic heart disease [20]. Further confusing was that many of the studies used different types of LLL-generating devices, with different parameters, in different model systems, making comparison of data almost impossible. Despite this initial impression, the possibility that a simple, non-invasive methodology could exist that augments regenerative potential in a tissue-focused manner became very enticing to us. Specific uses envisioned, for which intellectual property was filed included using light to concentrate stem cells to an area of need, to modulate effects of stem cells once they are in that specific area, or even to use light together with other agents to modulate endogenous stem cells.
The purpose of the current manuscript is to overview some of the previous work performed in this area that was of great interest to our ongoing work in regenerative medicine. We believe that greater integration of the area of LLL with current advancements in molecular and cellular biology will accelerate medical progress. Unfortunately, in our impression to date, this has been a very slow process.
What is Low Level Laser Irradiation?
Lasers (Light amplification by stimulated emission of radiation) are devices that typically generate electromagnetic radiation which is relatively uniform in wavelength, phase, and polarization, originally described by Theodore Maiman in 1960 in the form of a ruby laser [21]. These properties have allowed for numerous medical applications including uses in surgery, activation of photodynamic agents, and various ablative therapies in cosmetics that are based on heat/tissue destruction generated by the laser beam [22, 23, 24]. These applications of lasers are considered "high energy" because of their intensity, which ranges from about 10-100 Watts. The subject of the current paper will be another type of laser approach called low level lasers (LLL) that elicits effects through non-thermal means. This area of investigation started with the work of Mester et al who in 1967 reported non-thermal effects of lasers on mouse hair growth [25]. In a subsequent study [26], the same group reported acceleration of wound healing and improvement in regenerative ability of muscle fibers post wounding using a 1 J/cm2 ruby laser. Since those early days, numerous in vitro and in vivo studies have been reported demonstrating a wide variety of therapeutic effects involving LLL, a selected sample of which will be discussed below. In order to narrow our focus of discussion, it is important to first begin by establishing the current definition of LLL therapy. According to Posten et al [27], there are several parameters of importance: a) Power output of laser being 10-3 to 10-1 Watts; b) Wavelength in the range of 300-10,600 nm; c) Pulse rate from 0, meaning continuous to 5000 Hertz (cycles per second); d) intensity of 10-2-10 W/cm(2) and dose of 0.01 to 100 J/cm2. Most common methods of administering LLL radiation include lasers such as ruby (694 nm), Ar (488 and 514 nm), He-Ne (632.8 nm), Krypton (521, 530, 568, and 647 nm), Ga-Al-As (805 or 650 nm), and Ga-As (904 nm). Perhaps one of the most distinguishing features of LLL therapy as compared to other photoceutical modalities is that effects are mediated not through induction of thermal effects but rather through a process that is still not clearly defined called "photobiostimulation". It appears that this effect of LLL is not depend on coherence, and therefore allows for use of non-laser light generating devices such as inexpensive Light Emitting Diode (LED) technology [28].
To date several mechanisms of biological action have been proposed, although none are clearly established. These include augmentation of cellular ATP levels [29], manipulation of inducible nitric oxide synthase (iNOS) activity [30, 31], suppression of inflammatory cytokines such as TNF-alpha, IL-1beta, IL-6 and IL-8 [32, 33, 34, 35, 36], upregulation of growth factor production such as PDGF, IGF-1, NGF and FGF-2 [36, 37, 38, 39], alteration of mitochondrial membrane potential [29, 40, 41, 42] due to chromophores found in the mitochondrial respiratory chain [43, 44] as reviewed in [45], stimulation of protein kinase C (PKC) activation [46], manipulation of NF-κB activation [47], direct bacteriotoxic effect mediated by induction of reactive oxygen species (ROS) [48], modification of extracellular matrix components [49], inhibition of apoptosis [29], stimulation of mast cell degranulation [50], and upregulation of heat shock proteins [51]. Unfortunately these effects have been demonstrated using a variety of LLL devices in non-comparable models. To add to confusion, dose-dependency seems to be confined to such a narrow range or does not seem to exist in that numerous systems therapeutic effects disappear with increased dose.
In vitro studies of LLL
In areas of potential phenomenology, it is important to begin by assessing in vitro studies reported in the literature in which reproducibility can be attained with some degree of confidence, and mechanistic dissection is simpler as compared with in vivo systems. In 1983, one of the first studies to demonstrate in vitro effects of LLL was published. The investigators used a helium neon (He-Ne) laser to generate a visible red light at 632.8 nm for treatment of porcine granulosa cells. The paper described upregulation of metabolic and hormone-producing activity of the cells when exposed for 60 seconds to pulsating low power (2.8 mW) irradiation [52]. The possibility of modulating biologically-relevant signaling proteins by LLL was further assessed in a study using an energy dose of 1.5 J/cm2 in cultured keratinocytes. Administration of He-Ne laser emitted light resulted in upregulated gene expression of IL-1 and IL-8 [53]. Production of various growth factors in vitro suggests the possibility of enhanced cellular mitogenesis and mobility as a result of LLL treatment. Using a diode-based method to generate a similar wavelength to the He-Ne laser (363 nm), Mvula et al reported in two papers that irradiation at 5 J/cm2 of adipose derived mesenchymal stem cells resulted in enhanced proliferation, viability and expression of the adhesion molecule beta-1 integrin as compared to control [54, 55]. In agreement with possible regenerative activity based on activation of stem cells, other studies have used an in vitro injury model to examine possible therapeutic effects. Migration of fibroblasts was demonstrated to be enhanced in a "wound assay" in which cell monolayers are scraped with a pipette tip and amount of time needed to restore the monolayer is used as an indicator of "healing". The cells exposed to 5 J/cm2 generated by an He-Ne laser migrated rapidly across the wound margin indicating a stimulatory or positive influence of phototherapy. Higher doses (10 and 16 J/cm2) caused a decrease in cell viability and proliferation with a significant amount of damage to the cell membrane and DNA [56]. In order to examine whether LLL may positively affect healing under non-optimal conditions that mimic clinical situations treatment of fibroblasts from diabetic animals was performed. It was demonstrated that with the He-Ne laser dosage of 5 J/cm2 fibroblasts exhibited an enhanced migration activity, however at 16 J/cm2 activity was negated and cellular damage observed [57]. Thus from these studies it appears that energy doses from 1.5 J/cm2 to 5 J/cm2 are capable of eliciting "biostimulatory effects" in vitro in the He-Ne-based laser for adherent cells that may be useful in regeneration such as fibroblasts and mesenchymal stem cells.
Studies have also been performed in vitro on immunological cells. High intensity He-Ne irradiation at 28 and 112 J/cm2 of human peripheral blood mononuclear cells, a heterogeneous population of T cells, B cells, NK cells, and monocytes has been described to induce chromatin relaxation and to augment proliferative response to the T cell mitogen phytohemaglutin [58]. In human peripheral blood mononuclear cells (PBMC), another group reported in two papers that interleukin-1 alpha (IL-1 alpha), tumor necrosis factor-alpha (TNF-alpha), interleukin-2 (IL-2), and interferon-gamma (IFN-gamma) at a protein and gene level in PBMC was increased after He-Ne irradiation at 18.9 J/cm2 and decreased with 37.8 J/cm2 [59, 60]. Stimulation of human PBMC proliferation and murine splenic lymphocytes was also reported with He-Ne LLL [61, 62]. In terms of innate immune cells, enhanced phagocytic activity of murine macrophages have been reported with energy densities ranging from 100 to 600 J/cm2, with an optimal dose of 200 J/cm2 [63]. Furthermore, LLL has been demonstrated to augment human monocyte killing mycobacterial cells at similar densities, providing a functional correlation [64].
Thus from the selected in vitro studies discussed, it appears that modulation of proliferation and soluble factor production by LLL can be reliably reproduced. However the data may be to some extent contradictory. For example, the over-arching clinical rationale for use of LLL in conditions such as sinusitis [65], arthritis [66, 67], or wound healing [68] is that treatment is associated with anti-inflammatory effects. However the in vitro studies described above suggested LLL stimulates proinflammatory agents such as TNF-alpha or IL-1 [59, 60]. This suggests the in vivo effects of LLL may be very complex, which to some extent should not be surprising. Factors affecting LLL in vivo actions would include degree of energy penetration through the tissue, the various absorption ability of cells in the various tissues, and complex chemical changes that maybe occurring in paracrine/autocrine manner. Perhaps an analogy to the possible discrepancy between LLL effects in vitro versus in vivo may be made with the medical practice of extracorporeal ozonation of blood. This practice is similar to LLL therapy given that it is used in treatment of conditions such as atherosclerosis, non-healing ulcers, and various degenerative conditions, despite no clear mechanistic understanding [69, 70, 71]. In vitro studies have demonstrated that ozone is a potent oxidant and inducer of cell apoptosis and inflammatory signaling [72, 73, 74]. In contrast, in vivo systemic changes subsequent to administration of ozone or ozonized blood in animal models and patients are quite the opposite. Numerous investigators have published enhanced anti-oxidant enzyme activity such as elevations in Mg-SOD and glutathione-peroxidase levels, as well as diminishment of inflammation-associated pathology [75, 76, 77, 78]. Regardless of the complexity of in vivo situations, the fact that reproducible, in vitro experiments, demonstrate a biological effect provided support for us that there is some basis for LLL and it is not strictly an area of phenomenology.
Animal Studies with LLL
As early as 1983, Surinchak et al reported in a rat skin incision healing model that wounds exposed He-Ne radiation of fluency 2.2 J/cm2 for 3 min twice daily for 14 days demonstrated a 55% increase in breaking strength over control rats. Interestingly, higher doses yielded poorer healing [79]. This application of laser light was performed directly on shaved skin. In a contradictory experiment, it was reported that rats irradiated for 12 days with four levels of laser light (0.0, 0.47, 0.93, and 1.73 J/cm2) a possible strengthening of wounds tension was observed at the highest levels of irradiation (1.73 J/cm2), however it did not reach significance when analyzed by resampling statistics [80]. In another wound-healing study Ghamsari et al reported accelerated healing in the cranial surface of teats in dairy cows by administration of He-Ne irradiation at 3.64 J/cm2 dose of low-level laser, using a helium-neon system with an output of 8.5 mW, continuous wave [81]. Collagen fibers in LLL groups were denser, thicker, better arranged and more continuous with existing collagen fibers than those in non-LLL groups. The mean tensile strength was significantly greater in LLL groups than in non-LLL groups [82]. In the random skin flap model, the use of He-Ne laser irradiation with 3 J/cm2 energy density immediately after the surgery and for the four subsequent days was evaluated in 4 experimental groups: Group 1 (control) sham irradiation with He-Ne laser; Group 2 irradiation by punctual contact technique on the skin flap surface; Group 3 laser irradiation surrounding the skin flap; and Group 4 laser irradiation both on the skin flap surface and around it. The percentage of necrotic area of the four groups was determined on day 7-post injury. The control group had an average necrotic area of 48.86%; the group irradiated on the skin flap surface alone had 38.67%; the group irradiated around the skin flap had 35.34%; and the group irradiated one the skin flap surface and around it had 22.61%. All experimental groups reached statistically significant values when compared to control [83]. Quite striking results were obtained in an alloxan-induced diabetes wound healing model in which a circular 4 cm2 excisional wound was created on the dorsum of the diabetic rats. Treatment with He-Ne irradiation at 4.8 J/cm2 was performed 5 days a week until the wound healed completely and compared to sham irradiated animals. The laser-treated group healed on average by the 18th day whereas, the control group healed on average by the 59th day [84].
In addition to mechanically-induced wounds, beneficial effects of LLL have been obtained in burn-wounds in which deep second-degree burn wounds were induced in rats and the effects of daily He-Ne irradiation at 1.2 and 2.4 J/cm2 were assessed in comparison to 0.2% nitrofurazone cream. The number of macrophages at day 16, and the depth of new epidermis at day 30, was significantly less in the laser treated groups in comparison with control and nitrofurazone treated groups. Additionally, infections with S. epidermidis and S. aureus were significantly reduced [85].
While numerous studies have examined dermatological applications of LLL, which may conceptually be easier to perform due to ability to topically apply light, extensive investigation has also been made in the area of orthopedic applications. Healing acceleration has been observed in regeneration of the rat mid-cortical diaphysis of the tibiae, which is a model of post-injury bone healing. A small hole was surgically made with a dentistry burr in the tibia and the injured area and LLL was administered over a 7 or 14 day course transcutaneously starting 24 h from surgery. Incident energy density dosages of 31.5 and 94.5 J/cm2 were applied during the period of the tibia wound healing. Increased angiogenesis was observed after 7 days irradiation at an energy density of 94.5 J/cm2, but significantly decreased the number of vessels in the 14-day irradiated tibiae, independent of the dosage [86]. In an osteoarthritis model treatment with He-Ne resulted in augmentation of heat shock proteins and pathohistological improvement of arthritic cartilage [87]. The possibility that a type of preconditioning response is occurring, which would involve induction of genes such as hemoxygenase-1 [88], remains to be investigated. Effects of LLL therapy on articular cartilage were confirmed by another group. The experiment consisted of 42 young Wistar rats whose hind limbs were operated on in order to immobilize the knee joint. One week after operation they were assigned to three groups; irradiance 3.9 W/cm2, 5.8 W/cm2, and sham treatment. After 6 times of treatment for another 2 weeks significantpreservation of articular cartilage stiffness with 3.9 and 5.8 W/cm2therapy was observed [89].
Muscle regeneration by LLL was demonstrated in a rat model of disuse atrophy in which eight-week-old rats were subjected to hindlimb suspension for 2 weeks, after which they were released and recovered. During the recovery period, rats underwent daily LLL irradiation (Ga-Al-As laser; 830 nm; 60 mW; total, 180 s) to the right gastrocnemius muscle through the skin. After 2-weeks the number of capillaries and fibroblast growth factor levels exhibited significant elevation relative to those of the LLL-untreated muscles. LLL treatment induced proliferation in satellite cells as detected by BRdU [90].
Other animal studies of LLL have demonstrated effects in areas that appear unrelated such as suppression of snake venom induced muscle death [91], decreasing histamine-induced vasospasms [92], inhibition of post-injury restenosis [93], and immune stimulation by thymic irradiation [94].
Clinical Studies Using LLL
Growth factor secretion by LLL and its apparent regenerative activities have stimulated studies in radiation-induced mucositis. A 30 patient randomized trial of carcinoma patients treated by radiotherapy alone (65 Gy at a rate of 2 Gy/fraction, 5 fractions per week) without prior surgery or concomitant chemotherapy suffering from radiation-induced mucositis was performed using a He-Ne 60 mW laser. Grade 3 mucositis occured with a frequency of 35.2% in controls and at 7.6% of treated patients. Furthermore, a decrease in "severe pain" (grade 3) was observed in that 23.8% in the control group experienced this level of pain, as compared to 1.9% in the treatment group [95]. A subsequent study reported similar effects [96].
Healing ability of lasers was also observed in a study of patients with gingival flap incisions. Fifty-eight extraction patients had one of two gingival flap incisions lased with a 1.4 mW He-Ne (670 nm) at 0.34 J/cm2. Healing rates were evaluated clinically and photographically. Sixty-nine percent of the irradiated incisions healed faster than the control incisions. No significant difference in healing was noted when patients were compared by age, gender, race, and anatomic location of the incision [97]. Another study evaluating healing effects of LLL in dental practice examined 48 patients subjected to surgical removal of their lower third molars. Treated patients were administered Ga-Al-As diode generated 808 nm at a dose of 12 J. The study demonstrated that extraoral LLL is more effective than intraoral LLL, which was more effective than control for the reduction of postoperative trismus and swelling after extraction of the lower third molar [98].
Given the predominance of data supporting fibroblast proliferative ability and animal wound healing effects of LLL therapy, a clinical trial was performed on healing of ulcers. In a double-blinded fashion 23 diabetic leg ulcers from 14 patients were divided into two groups. Phototherapy was applied (<1.0 J/cm2) twice per week, using a Dynatron Solaris 705(R) LED device that concurrently emits 660 and 890 nm energies. At days 15, 30, 45, 60, 75, and 90 mean ulcer granulation and healing rates were significantly higher for the treatment group as compared to control. By day 90, 58.3% of the ulcers in the LLL treated group were fully healed and 75% achieved 90-100% healing. In the placebo group only one ulcer healed fully [68].
As previously mentioned, LLL appears to have some angiogenic activity. One of the major problems in coronary artery disease is lack of collateralization. In a 39 patient study advanced CAD, two sessions of irradiation of low-energy laser light on skin in the chest area from helium-neon B1 lasers. The time of irradiation was 15 minutes while operations were performed 6 days a week for one month. Reduction in Canadian Cardiology Society (CCS) score, increased exercise capacity and time, less frequent angina symptoms during the treadmill test, longer distance of 6-minute walk test and a trend towards less frequent 1 mm ST depression lasting 1 min during Holter recordings was noted after therapy [99].
Perhaps one of the largest clinical trials with LLL was the NEST trial performed by Photothera. In this double blind trial 660 stroke patients were recruited and randomized: 331 received LLL and 327 received sham. No prespecified test achieved significance, but a post hoc analysis of patients with a baseline National Institutes of Health Stroke Scale score of <16 showed a favorable outcome at 90 days on the primary end point (P < 0.044) [100]. Currently Photothera is in the process of repeating this trial with modified parameters.
Relevance of LLL to COPD
A therapeutic intervention in COPD would require addressing the issues of inflammation and regeneration. Although approaches such as administration of bone marrow stem cells, or fat derived cellular components have both regenerative and anti-inflammatory activity in animal models, the need to enhance their potency for clinical applications can be seen in the recent Osiris's COPD trial interim data which reported no significant improvement in pulmonary function [101]. Accordingly, we sought to develop a possible rationale for how LLL may be useful as an adjunct to autologous stem cell therapy.
Table 1 depicts some of the properties of LLL that provide a rationale for the combined use with stem cells. One of the basic properties of LLL seems to be ability to inhibit inflammation at the level of innate immune activation. Representative studies showed that LLL was capable of suppressing inflammatory genes and/or pathology after administration of lipopolysaccharide (LPS) as a stimulator of monocytes [102] and bronchial cells [34], in vitro, and leukocyte infiltration in vivo [103, 104]. Inflammation induced by other stimulators such as zymosan, carrageenan, and TNF-alpha was also inhibited by LLL [32, 105, 106]. Growth factor stimulating activity of LLL was demonstrated in both in vitro and in vivo experiments in which augmentation of FGF-2, PDGF and IGF-1 was observed [36, 37, 107]. Endogenous production of these growth factors may be useful in regeneration based on activation of endogenous pulmonary stem cells [108, 109]. Another aspect of LLL activities of relevance is ability to stimulate angiogenesis. In COPD, the constriction of blood vessels as a result of poor oxygen uptake is results in a feedback loop culminating in pulmonary hypertension. Administration of angiogenic factors has been demonstrated to be beneficial in several animal models of pulmonary pathology [110, 111]. The ability of LLL to directly induce proliferation of HUVEC cells [112], as well as to augment production of angiogenic factors such as VEGF [113], supports the possibility of creation of an environment hospitable to neoangiogenesis which is optimal for stem cell growth. In fact, a study demonstrated in vivo induction of neocapillary formation subsequent to LLL administration in a hindlimb ischemia model [114]. The critical importance of angiogenesis in stem cell mediated regeneration has previously been demonstrated in the stroke model, where the major therapeutic activity of exogenous stem cells has been attributed to angiogenic as opposed to transdifferentiation effects [115].
Table 1
Examples of LLL Properties Relevant to COPD
COPD Property
LLL Experiment
Original Source: https://translational-medicine.biomedcentral.com/articles/10.1186/1479-5876-8-16
Lasers, stem cells, and COPD
Feng Lin1†, Steven F Josephs1†, Doru T Alexandrescu2†, Famela Ramos1, Vladimir Bogin3, Vincent Gammill4, Constantin A Dasanu5, Rosalia De Necochea-Campion6, Amit N Patel7, Ewa Carrier6, David R Koos1* - (Publication) 4488
View Resource
Abstract
The medical use of low level laser (LLL) irradiation has been occurring for decades, primarily in the area of tissue
healing and inflammatory conditions. Despite little mechanistic knowledge, the concept of a non-invasive, nonthermal
intervention that has the potential to modulate regenerative processes is worthy of attention when searching
for novel methods of augmenting stem cell-based therapies. Here we discuss the use of LLL irradiation as a
“photoceutical” for enhancing production of stem cell growth/chemoattractant factors, stimulation of angiogenesis,
and directly augmenting proliferation of stem cells. The combination of LLL together with allogeneic and autologous
stem cells, as well as post-mobilization directing of stem cells will be discussed.
Introduction (Personal Perspective)
We came upon the field of low level laser (LLL) therapy
by accident. One of our advisors read a press release
about a company using this novel technology of specific
light wavelengths to treat stroke. Given the possible role
of stem cells in post-stroke regeneration, we decided to
cautiously investigate. As a background, it should be
said that our scientific team has been focusing on the
area of cord blood banking and manufacturing of disposables
for processing of adipose stem cells for the past 3
years. Our board has been interested in strategically
refocusing the company from services-oriented into a
more research-focused model. An unbiased exploration
into the various degenerative conditions that may be
addressed by our existing know-how led us to explore
the condition of chronic obstructive pulmonary disease
(COPD), an umbrella term covering chronic bronchitis
and emphysema, which is the 4th largest cause of death
in the United States. As a means of increasing our probability
of success in treatment of this condition, the
decision was made to develop an adjuvant therapy that
would augment stem cell activity. The field of LLL therapy
attracted us because it appeared to be relatively
unexplored scientific territory for which large amounts
of clinical experience exist. Unfortunately, it was difficult
to obtain the cohesive “state-of-the-art” description of
the molecular/cellular mechanisms of this therapy in
reviews that we have searched. Therefore we sought in
this mini-review to discuss what we believe to be relevant
to investigators attracted by the concept of “regenerative
photoceuticals”. Before presenting our synthesis
of the field, we will begin by describing our rationale for
approaching COPD with the autologous stem cell based
approaches we are developing.
COPD as an Indication for Stem Cell Therapy
COPD possesses several features making it ideal for
stem cell based interventions: a) the quality of life and
lack of progress demands the ethical exploration of
novel approaches. For example, bone marrow stem cells
have been used in over a thousand cardiac patients with
some indication of efficacy [1,2]. Adipose-based stem
cell therapies have been successfully used in thousands
of race-horses and companion animals without adverse
effects [3], as well as numerous clinical trials are
ongoing and published human data reports no adverse
effects (reviewed in ref [4]). Unfortunately, evaluation of
stem cell therapy in COPD has lagged behind other
areas of regenerative investigation; b) the underlying
cause of COPD appears to be inflammatory and/or
immunologically mediated. The destruction of alveolar
tissue is associated with T cell reactivity [5,6], pathological
pulmonary macrophage activation [7], and auto-antibody
production [8]. Mesenchymal stem cells have been
demonstrated to potently suppress autoreactive T cells
[9,10], inhibit macrophage activation [11], and autoantibody
responses [12]. Additionally, mesenchymal stem
cells can be purified in high concentrations from adipose
stromal vascular tissue together with high concentrations of T regulatory cells [4], which in animal
models are approximately 100 more potent than peripheral
T cells at secreting cytokines therapeutic for COPD
such as IL-10 [13,14]. Additionally, use of adipose
derived cells has yielded promising clinical results in
autoimmune conditions such as multiple sclerosis [4];
and c) Pulmonary stem cells capable of regenerating
damaged parenchymal tissue have been reported [15].
Administration of mesenchymal stem cells into neonatal
oxygen-damaged lungs, which results in COPD-like
alveoli dysplasia, has been demonstrated to yield
improvements in two recent publications [16,17].
Based on the above rationale for stem cell-based
COPD treatments, we began our exploration into this
area by performing several preliminary experiments and
filing patents covering combination uses of stem cells
with various pharmacologically available antiinflammatories,
as well as methods of immune modulation. These
have served as the basis for two of our pipeline candidates,
ENT-111, and ENT-894. As a commerciallyoriented
organization, we needed to develop a therapeutic
candidate that not only has a great potential for efficacy,
but also can be easily implemented as part of the
standard of care. Our search led us to the area of low
level laser (LLL) therapy. From our initial perception as
neophytes to this field, the area of LLL therapy has been
somewhat of a medical mystery. A pubmed search for
“low level laser therapy” yields more than 1700 results,
yet before stumbling across this concept, none of us, or
our advisors, have ever heard of this area of medicine.
On face value, this field appeared to be somewhat of a
panacea: clinical trials claiming efficacy for conditions
ranging from alcoholism [18], to sinusitis [19], to
ischemic heart disease [20]. Further confusing was that
many of the studies used different types of LLL-generating
devices, with different parameters, in different model
systems, making comparison of data almost impossible.
Despite this initial impression, the possibility that a simple,
non-invasive methodology could exist that augments
regenerative potential in a tissue-focused manner
became very enticing to us. Specific uses envisioned, for
which intellectual property was filed included using light
to concentrate stem cells to an area of need, to modulate
effects of stem cells once they are in that specific
area, or even to use light together with other agents to
modulate endogenous stem cells.
The purpose of the current manuscript is to overview
some of the previous work performed in this area that was
of great interest to our ongoing work in regenerative medicine.
We believe that greater integration of the area of
LLL with current advancements in molecular and cellular
biology will accelerate medical progress. Unfortunately, in
our impression to date, this has been a very slow process.
What is Low Level Laser Irradiation?
Lasers (Light amplification by stimulated emission of
radiation) are devices that typically generate electromagnetic
radiation which is relatively uniform in wavelength,
phase, and polarization, originally described by Theodore
Maiman in 1960 in the form of a ruby laser [21]. These
properties have allowed for numerous medical applications
including uses in surgery, activation of photodynamic
agents, and various ablative therapies in cosmetics that are
based on heat/tissue destruction generated by the laser
beam [22-24]. These applications of lasers are considered
“high energy” because of their intensity, which ranges
from about 10-100 Watts. The subject of the current
paper will be another type of laser approach called low
level lasers (LLL) that elicits effects through non-thermal
means. This area of investigation started with the work of
Mester et al who in 1967 reported non-thermal effects of
lasers on mouse hair growth [25]. In a subsequent study
[26], the same group reported acceleration of wound healing
and improvement in regenerative ability of muscle
fibers post wounding using a 1 J/cm2 ruby laser. Since
those early days, numerous in vitro and in vivo studies
have been reported demonstrating a wide variety of therapeutic
effects involving LLL, a selected sample of which
will be discussed below. In order to narrow our focus of
discussion, it is important to first begin by establishing the
current definition of LLL therapy. According to Posten et
al [27], there are several parameters of importance: a)
Power output of laser being 10-3 to 10-1 Watts; b) Wavelength
in the range of 300-10,600 nm; c) Pulse rate from 0,
meaning continuous to 5000 Hertz (cycles per second); d)
intensity of 10-2-10 W/cm(2) and dose of 0.01 to 100 J/
cm2. Most common methods of administering LLL radiation
include lasers such as ruby (694 nm), Ar (488 and 514
nm), He-Ne (632.8 nm), Krypton (521, 530, 568, and 647
nm), Ga-Al-As (805 or 650 nm), and Ga-As (904 nm).
Perhaps one of the most distinguishing features of LLL
therapy as compared to other photoceutical modalities is
that effects are mediated not through induction of thermal
effects but rather through a process that is still not clearly
defined called “photobiostimulation”. It appears that this
effect of LLL is not depend on coherence, and therefore
allows for use of non-laser light generating devices such as
inexpensive Light Emitting Diode (LED) technology [28].
To date several mechanisms of biological action have
been proposed, although none are clearly established.
These include augmentation of cellular ATP levels [29],
manipulation of inducible nitric oxide synthase (iNOS)
activity [30,31], suppression of inflammatory cytokines
such as TNF-alpha, IL-1beta, IL-6 and IL-8 [32-36],
upregulation of growth factor production such as PDGF,
IGF-1, NGF and FGF-2 [36-39], alteration of mitochondrial
membrane potential [29,40-42] due to chromophores found in the mitochondrial respiratory
chain [43,44] as reviewed in [45], stimulation of protein
kinase C (PKC) activation [46], manipulation of NF-!B
activation [47], direct bacteriotoxic effect mediated by
induction of reactive oxygen species (ROS) [48], modification
of extracellular matrix components [49], inhibition
of apoptosis [29], stimulation of mast cell
degranulation [50], and upregulation of heat shock proteins
[51]. Unfortunately these effects have been demonstrated
using a variety of LLL devices in noncomparable
models. To add to confusion, dose-dependency
seems to be confined to such a narrow range or
does not seem to exist in that numerous systems therapeutic
effects disappear with increased dose.
In vitro studies of LLL
In areas of potential phenomenology, it is important to
begin by assessing in vitro studies reported in the literature
in which reproducibility can be attained with some
degree of confidence, and mechanistic dissection is simpler
as compared with in vivo systems. In 1983, one of
the first studies to demonstrate in vitro effects of LLL
was published. The investigators used a helium neon
(He-Ne) laser to generate a visible red light at 632.8 nm
for treatment of porcine granulosa cells. The paper
described upregulation of metabolic and hormone-producing
activity of the cells when exposed for 60 seconds
to pulsating low power (2.8 mW) irradiation [52]. The
possibility of modulating biologically-relevant signaling
proteins by LLL was further assessed in a study using an
energy dose of 1.5 J/cm2 in cultured keratinocytes.
Administration of He-Ne laser emitted light resulted in
upregulated gene expression of IL-1 and IL-8 [53]. Production
of various growth factors in vitro suggests the
possibility of enhanced cellular mitogenesis and mobility
as a result of LLL treatment. Using a diode-based
method to generate a similar wavelength to the He-Ne
laser (363 nm), Mvula et al reported in two papers that
irradiation at 5 J/cm2 of adipose derived mesenchymal
stem cells resulted in enhanced proliferation, viability
and expression of the adhesion molecule beta-1 integrin
as compared to control [54,55]. In agreement with possible
regenerative activity based on activation of stem
cells, other studies have used an in vitro injury model to
examine possible therapeutic effects. Migration of fibroblasts
was demonstrated to be enhanced in a “wound
assay” in which cell monolayers are scraped with a pipette
tip and amount of time needed to restore the
monolayer is used as an indicator of “healing”. The cells
exposed to 5 J/cm2 generated by an He-Ne laser
migrated rapidly across the wound margin indicating a
stimulatory or positive influence of phototherapy.
Higher doses (10 and 16 J/cm2) caused a decrease in
cell viability and proliferation with a significant amount
of damage to the cell membrane and DNA [56]. In
order to examine whether LLL may positively affect
healing under non-optimal conditions that mimic clinical
situations treatment of fibroblasts from diabetic animals
was performed. It was demonstrated that with the
He-Ne laser dosage of 5 J/cm2 fibroblasts exhibited an
enhanced migration activity, however at 16 J/cm2 activity
was negated and cellular damage observed [57]. Thus
from these studies it appears that energy doses from 1.5
J/cm2 to 5 J/cm2 are capable of eliciting “biostimulatory
effects” in vitro in the He-Ne-based laser for adherent
cells that may be useful in regeneration such as fibroblasts
and mesenchymal stem cells.
Studies have also been performed in vitro on immunological
cells. High intensity He-Ne irradiation at 28
and 112 J/cm2 of human peripheral blood mononuclear
cells, a heterogeneous population of T cells, B cells, NK
cells, and monocytes has been described to induce chromatin
relaxation and to augment proliferative response
to the T cell mitogen phytohemaglutin [58]. In human
peripheral blood mononuclear cells (PBMC), another
group reported in two papers that interleukin-1 alpha
(IL-1 alpha), tumor necrosis factor-alpha (TNF-alpha),
interleukin-2 (IL-2), and interferon-gamma (IFNgamma)
at a protein and gene level in PBMC was
increased after He-Ne irradiation at 18.9 J/cm2 and
decreased with 37.8 J/cm2 [59,60]. Stimulation of human
PBMC proliferation and murine splenic lymphocytes
was also reported with He-Ne LLL [61,62]. In terms of
innate immune cells, enhanced phagocytic activity of
murine macrophages have been reported with energy
densities ranging from 100 to 600 J/cm2, with an optimal
dose of 200 J/cm2 [63]. Furthermore, LLL has been
demonstrated to augment human monocyte killing
mycobacterial cells at similar densities, providing a functional
correlation [64].
Thus from the selected in vitro studies discussed, it
appears that modulation of proliferation and soluble factor
production by LLL can be reliably reproduced. However
the data may be to some extent contradictory. For
example, the over-arching clinical rationale for use of
LLL in conditions such as sinusitis [65], arthritis [66,67],
or wound healing [68] is that treatment is associated
with anti-inflammatory effects. However the in vitro studies
described above suggested LLL stimulates proinflammatory
agents such as TNF-alpha or IL-1 [59,60].
This suggests the in vivo effects of LLL may be very
complex, which to some extent should not be surprising.
Factors affecting LLL in vivo actions would include
degree of energy penetration through the tissue, the various
absorption ability of cells in the various tissues, and
complex chemical changes that maybe occurring in
paracrine/autocrine manner. Perhaps an analogy to the
possible discrepancy between LLL effects in vitro versus in vivo may be made with the medical practice of extracorporeal
ozonation of blood. This practice is similar to
LLL therapy given that it is used in treatment of conditions
such as atherosclerosis, non-healing ulcers, and
various degenerative conditions, despite no clear
mechanistic understanding [69-71]. In vitro studies have
demonstrated that ozone is a potent oxidant and inducer
of cell apoptosis and inflammatory signaling [72-74].
In contrast, in vivo systemic changes subsequent to
administration of ozone or ozonized blood in animal
models and patients are quite the opposite. Numerous
investigators have published enhanced anti-oxidant
enzyme activity such as elevations in Mg-SOD and glutathione-
peroxidase levels, as well as diminishment of
inflammation-associated pathology [75-78]. Regardless
of the complexity of in vivo situations, the fact that
reproducible, in vitro experiments, demonstrate a biological
effect provided support for us that there is some
basis for LLL and it is not strictly an area of
phenomenology.
Animal Studies with LLL
As early as 1983, Surinchak et al reported in a rat skin
incision healing model that wounds exposed He-Ne
radiation of fluency 2.2 J/cm2 for 3 min twice daily for
14 days demonstrated a 55% increase in breaking
strength over control rats. Interestingly, higher doses
yielded poorer healing [79]. This application of laser
light was performed directly on shaved skin. In a contradictory
experiment, it was reported that rats irradiated
for 12 days with four levels of laser light (0.0, 0.47, 0.93,
and 1.73 J/cm2) a possible strengthening of wounds tension
was observed at the highest levels of irradiation
(1.73 J/cm2), however it did not reach significance when
analyzed by resampling statistics [80]. In another
wound-healing study Ghamsari et al reported accelerated
healing in the cranial surface of teats in dairy cows
by administration of He-Ne irradiation at 3.64 J/cm2
dose of low-level laser, using a helium-neon system with
an output of 8.5 mW, continuous wave [81]. Collagen
fibers in LLL groups were denser, thicker, better
arranged and more continuous with existing collagen
fibers than those in non-LLL groups. The mean tensile
strength was significantly greater in LLL groups than in
non-LLL groups [82]. In the random skin flap model,
the use of He-Ne laser irradiation with 3 J/cm2 energy
density immediately after the surgery and for the four
subsequent days was evaluated in 4 experimental
groups: Group 1 (control) sham irradiation with He-Ne
laser; Group 2 irradiation by punctual contact technique
on the skin flap surface; Group 3 laser irradiation surrounding
the skin flap; and Group 4 laser irradiation
both on the skin flap surface and around it. The percentage
of necrotic area of the four groups was determined
on day 7-post injury. The control group had an average
necrotic area of 48.86%; the group irradiated on the skin
flap surface alone had 38.67%; the group irradiated
around the skin flap had 35.34%; and the group irradiated
one the skin flap surface and around it had
22.61%. All experimental groups reached statistically significant
values when compared to control [83]. Quite
striking results were obtained in an alloxan-induced diabetes
wound healing model in which a circular 4 cm2
excisional wound was created on the dorsum of the diabetic
rats. Treatment with He-Ne irradiation at 4.8 J/
cm2 was performed 5 days a week until the wound
healed completely and compared to sham irradiated animals.
The laser-treated group healed on average by the
18th day whereas, the control group healed on average
by the 59th day [84].
In addition to mechanically-induced wounds, beneficial
effects of LLL have been obtained in burn-wounds
in which deep second-degree burn wounds were
induced in rats and the effects of daily He-Ne irradiation
at 1.2 and 2.4 J/cm2 were assessed in comparison to
0.2% nitrofurazone cream. The number of macrophages
at day 16, and the depth of new epidermis at day 30,
was significantly less in the laser treated groups in comparison
with control and nitrofurazone treated groups.
Additionally, infections with S. epidermidis and S. aureus
were significantly reduced [85].
While numerous studies have examined dermatological
applications of LLL, which may conceptually be
easier to perform due to ability to topically apply light,
extensive investigation has also been made in the area
of orthopedic applications. Healing acceleration has
been observed in regeneration of the rat mid-cortical
diaphysis of the tibiae, which is a model of post-injury
bone healing. A small hole was surgically made with a
dentistry burr in the tibia and the injured area and LLL
was administered over a 7 or 14 day course transcutaneously
starting 24 h from surgery. Incident energy density
dosages of 31.5 and 94.5 J/cm2 were applied during
the period of the tibia wound healing. Increased angiogenesis
was observed after 7 days irradiation at an
energy density of 94.5 J/cm2, but significantly decreased
the number of vessels in the 14-day irradiated tibiae,
independent of the dosage [86]. In an osteoarthritis
model treatment with He-Ne resulted in augmentation
of heat shock proteins and pathohistological improvement
of arthritic cartilage [87]. The possibility that a
type of preconditioning response is occurring, which
would involve induction of genes such as hemoxygenase-
1 [88], remains to be investigated. Effects of LLL
therapy on articular cartilage were confirmed by another
group. The experiment consisted of 42 young Wistar
rats whose hind limbs were operated on in order to
immobilize the knee joint. One week after operation they were assigned to three groups; irradiance 3.9 W/
cm2, 5.8 W/cm2, and sham treatment. After 6 times of
treatment for another 2 weeks significantpreservation of
articular cartilage stiffness with 3.9 and 5.8 W/cm2 therapy
was observed [89].
Muscle regeneration by LLL was demonstrated in a rat
model of disuse atrophy in which eight-week-old rats
were subjected to hindlimb suspension for 2 weeks,
after which they were released and recovered. During
the recovery period, rats underwent daily LLL irradiation
(Ga-Al-As laser; 830 nm; 60 mW; total, 180 s) to
the right gastrocnemius muscle through the skin. After
2-weeks the number of capillaries and fibroblast growth
factor levels exhibited significant elevation relative to
those of the LLL-untreated muscles. LLL treatment
induced proliferation in satellite cells as detected by
BRdU [90].
Other animal studies of LLL have demonstrated
effects in areas that appear unrelated such as suppression
of snake venom induced muscle death [91],
decreasing histamine-induced vasospasms [92], inhibition
of post-injury restenosis [93], and immune stimulation
by thymic irradiation [94].
Clinical Studies Using LLL
Growth factor secretion by LLL and its apparent regenerative
activities have stimulated studies in radiationinduced
mucositis. A 30 patient randomized trial of carcinoma
patients treated by radiotherapy alone (65 Gy at
a rate of 2 Gy/fraction, 5 fractions per week) without
prior surgery or concomitant chemotherapy suffering
from radiation-induced mucositis was performed using a
He-Ne 60 mW laser. Grade 3 mucositis occured with a
frequency of 35.2% in controls and at 7.6% of treated
patients. Furthermore, a decrease in “severe pain” (grade
3) was observed in that 23.8% in the control group
experienced this level of pain, as compared to 1.9% in
the treatment group [95]. A subsequent study reported
similar effects [96].
Healing ability of lasers was also observed in a study
of patients with gingival flap incisions. Fifty-eight extraction
patients had one of two gingival flap incisions lased
with a 1.4 mW He-Ne (670 nm) at 0.34 J/cm2. Healing
rates were evaluated clinically and photographically.
Sixty-nine percent of the irradiated incisions healed faster
than the control incisions. No significant difference
in healing was noted when patients were compared by
age, gender, race, and anatomic location of the incision
[97]. Another study evaluating healing effects of LLL in
dental practice examined 48 patients subjected to surgical
removal of their lower third molars. Treated patients
were administered Ga-Al-As diode generated 808 nm at
a dose of 12 J. The study demonstrated that extraoral
LLL is more effective than intraoral LLL, which was
more effective than control for the reduction of postoperative
trismus and swelling after extraction of the
lower third molar [98].
Given the predominance of data supporting fibroblast
proliferative ability and animal wound healing effects of
LLL therapy, a clinical trial was performed on healing of
ulcers. In a double-blinded fashion 23 diabetic leg ulcers
from 14 patients were divided into two groups. Phototherapy
was applied (<1.0 J/cm2) twice per week, using a
Dynatron Solaris 705(R) LED device that concurrently
emits 660 and 890 nm energies. At days 15, 30, 45, 60,
75, and 90 mean ulcer granulation and healing rates
were significantly higher for the treatment group as
compared to control. By day 90, 58.3% of the ulcers in
the LLL treated group were fully healed and 75%
achieved 90-100% healing. In the placebo group only
one ulcer healed fully [68].
As previously mentioned, LLL appears to have some
angiogenic activity. One of the major problems in coronary
artery disease is lack of collateralization. In a 39
patient study advanced CAD, two sessions of irradiation
of low-energy laser light on skin in the chest area from
helium-neon B1 lasers. The time of irradiation was 15
minutes while operations were performed 6 days a week
for one month. Reduction in Canadian Cardiology
Society (CCS) score, increased exercise capacity and
time, less frequent angina symptoms during the treadmill
test, longer distance of 6-minute walk test and a
trend towards less frequent 1 mm ST depression lasting
1 min during Holter recordings was noted after therapy
[99].
Perhaps one of the largest clinical trials with LLL was
the NEST trial performed by Photothera. In this double
blind trial 660 stroke patients were recruited and randomized:
331 received LLL and 327 received sham. No
prespecified test achieved significance, but a post hoc
analysis of patients with a baseline National Institutes of
Health Stroke Scale score of <16 showed a favorable
outcome at 90 days on the primary end point (P <
0.044) [100]. Currently Photothera is in the process of
repeating this trial with modified parameters.
Relevance of LLL to COPD
A therapeutic intervention in COPD would require
addressing the issues of inflammation and regeneration.
Although approaches such as administration of bone marrow
stem cells, or fat derived cellular components have
both regenerative and anti-inflammatory activity in animal
models, the need to enhance their potency for clinical
applications can be seen in the recent Osiris’s COPD trial
interim data which reported no significant improvement
in pulmonary function [101]. Accordingly, we sought to
develop a possible rationale for how LLL may be useful as
an adjunct to autologous stem cell therapy.
Table 1 Examples of LLL Properties Relevant to COPD
COPD
Property
LLL Experiment LLL Details Ref
Inflammation In vivo. Decreased joint inflammation in zymosan-induced
arthritis
Semiconductor laser (685 nm and 830 nm) at (2.5 J/cm2)
In vitro. Suppression of LPS-induced bronchial inflammation and
TNF-alpha.
655 nm at of 2.6 J/cm2
In vivo. Carrageenan-induced pleurisy had decreased leukocyte
infiltration and cytokine (TNF-alpha, IL-6, and MCP)
660 nm at 2.1 J/cm2
In vitro. LPS stimulated Raw 264.7 monocytes had reduced gene
expression of MCP-1, IL-1 and IL-6
780 nm diode laser at 2.2 J/cm2)
In vivo. Suppression of LPS-stimulated neutrophil influx,
myeloperoxidase activity and IL-1beta in bronchoalveolar lavage
fluid.
660 nm diode laser at 7.5 J/cm2
In vitro. Inhibition of TNF-alpha induced IL-1, IL-8 and TNF-alpha
mRNA in human synoviocytes
810 nm (5 J/cm2) suppressed IL-1 and TNF, (25 J/cm2) also
suppressed IL-8
In vivo. Reduction of TNF-alpha in diaphragm muscle after
intravenous LPS injection.
4 sessions in 24 h with diode Ga-AsI-Al laser of 650 nm and
a total dose of 5.2 J/cm2
In vivo. Inhibition of LPS induced peritonitis and neutrophil influx 3 J/cm2 and 7.5 J/cm2
Growth Factor Production
In vivo. Upregulation of TGF-b and PDGF in rat gingiva after
incision.
He-Ne laser (632.8 nm) at a dose of 7.5 J/cm2
In vitro. Osteoblast-like cells were isolated from fetal rat calvariae
had increased IGF-1
Ga-Al-As laser (830 nm) at (3.75 J/cm2).
In vitro. Upregulated production of IGF-1 and FGF-2 in human
gingival fibroblasts.
685 nm, for 140 s, 2 J/cm2
Angiogenesis
In vivo. Increased fiber to capillary ratio in rabbits with ligated
femoral arteries.
Gallium-aluminum-arsenide (Ga-Al-As) diode laser, 904 nm
and power of 10 mW
In vitro. Stimulation of HUVEC proliferation by conditioned media
from LLL-treated T cells
820 nm at 1.2 and 3.6 J/cm2.
In vitro. 7-fold increased production of VEGF by cardiomyocytes,
1.6-fold increase by smooth muscle cells (SMC) and fibroblasts.
Supernatant of SMC had increased HUVEC-stimulating potential.
He:Ne continuous wave laser (632 nm). 0.5 J/cm2 for SMC,
2.1 J/cm2 for fibroblasts and 1.05 J/cm2 for cardiomyocytes.
In vitro. Direct stimulation of HUVEC proliferation 670 nm diode device at 2 and 8 J/cm2
Direct Stem Cell Effects
In vivo. LLL precondition significantly enhanced early cell survival
rate by 2-fold, decreased the apoptotic percentage of implanted
BMSCs in infarcted myocardium and increased the number of
newly formed capillaries.
635 nm at 0.96 J/cm2
In vitro. LLL stimulated MSC proliferation, VEGF and NGF
production, and myogenic differentiation after 5-aza induction.
635 nm diode laser at 0.5 J/cm2 for MSC proliferation, 5 J/
cm2 for VEGF and NGF production and for augmentation of
induced myogenic differentiation
In vitro. Increased proliferation of rat MSC. Red light LED 630 nm at 2 and 4 J/cm(2)
In vitro. Augmented proliferation of bone marrow and cardiac
specific stem cells.
GA-Al-As 810 nm at 1 and 3 J/cm2
In vitro/In vivo. Administration of LLL-treated MSC resulted 53%
reduction in infarct size, 5- and 6.3-fold significant increase in cell
density that positively immunoreacted to BrdU and c-kit,
respectively, and 1.4- and 2-fold higher level of angiogenesis and
vascular endothelial growth factor, respectively, when compared
to non-laser-treated implanted cells
Ga-Al-As laser (810 nm wavelength), 1 J/cm2
In vitro. Enhanced proliferation of adipose derived MSC in
presence of EGF.
636 nm diode, 5 J/cm2
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
Table 1: Examples of LLL Properties Relevant to COPD (Continued)
In vitro. Enhanced proliferation and beta-1 integrin expression of
adipose derived MSC.
635 nm diode laser, at 5 J/cm2
Clinical. 660 stroke patients: 331 received LLL and 327 received
sham. No prespecified test achieved significance, but a post hoc
analysis of patients with a baseline National Institutes of Health
Stroke Scale score of <16 showed a favorable outcome at 90
days on the primary end point (P < 0.044).
808 nm. No density disclosed.
Table 1 depicts some of the properties of LLL that provide
a rationale for the combined use with stem cells. One
of the basic properties of LLL seems to be ability to inhibit
inflammation at the level of innate immune activation.
Representative studies showed that LLL was capable of
suppressing inflammatory genes and/or pathology after
administration of lipopolysaccharide (LPS) as a stimulator
of monocytes [102] and bronchial cells [34], in vitro, and
leukocyte infiltration in vivo [103,104]. Inflammation
induced by other stimulators such as zymosan, carrageenan,
and TNF-alpha was also inhibited by LLL
[32,105,106]. Growth factor stimulating activity of LLL
was demonstrated in both in vitro and in vivo experiments
in which augmentation of FGF-2, PDGF and IGF-1 was
observed [36,37,107]. Endogenous production of these
growth factors may be useful in regeneration based on
activation of endogenous pulmonary stem cells [108,109].
Another aspect of LLL activities of relevance is ability to
stimulate angiogenesis. In COPD, the constriction of
blood vessels as a result of poor oxygen uptake is results
in a feedback loop culminating in pulmonary hypertension.
Administration of angiogenic factors has been
demonstrated to be beneficial in several animal models of
pulmonary pathology [110,111]. The ability of LLL to
directly induce proliferation of HUVEC cells [112], as well
as to augment production of angiogenic factors such as
VEGF [113], supports the possibility of creation of an
environment hospitable to neoangiogenesis which is optimal
for stem cell growth. In fact, a study demonstrated in
vivo induction of neocapillary formation subsequent to
LLL administration in a hindlimb ischemia model [114].
The critical importance of angiogenesis in stem cell
mediated regeneration has previously been demonstrated
in the stroke model, where the major therapeutic activity
of exogenous stem cells has been attributed to angiogenic
as opposed to transdifferentiation effects [115].
Direct evidence of LLL stimulating stem cells has been
obtained using mesenchymal stem cells derived both
from the bone marrow and from the adipose tissue
[116,117]. Interestingly in vivo administration of LLL stimulated
MSC has resulted in 50% decrease in cardiac
infarct size [118]. Clinical translation of LLL has been
performed in the area of stroke, in which a 660 patient
trial demonstrated statistically significant effects in post
trial subset analysis [100].
Conclusions
Despite clinical use of LLL for decades, the field is still
in its infancy. As is obvious from the wide variety of
LLL sources, frequencies, and intensities used, no standard
protocols exist. The ability of LLL to induce
growth factor production, inhibition of inflammation,
stimulation of angiogenesis, and direct effects on stem
cells suggests the urgent need for combining this modality
with regenerative medicine, giving birth to the new
field of “regenerative photoceuticals”. Development of a
regenerative treatment for COPD as well as for other
degenerative diseases would be of considerable benefit.
Regarding COPD, such treatment would be life-saving/
life extending for thousands of affected individuals.
Ceasing smoking or not starting to smoke would considerably
impact this disease.
Acknowledgements
The authors thank Victoria Dardov and Matthew Gandjian for critical
discussions and input.
Author details
1Entest BioMedical, San Diego, CA, USA. 2Georgetown Dermatology,
Washington DC, USA. 3Cromos Pharma Services, Longview, WA, USA. 4Center
for the Study of Natural Oncology, Del Mar, CA, USA. 5Department of
Hematology and Medical Oncology, St Francis Hospital and Medical Center,
Hartford, CT, USA. 6Moores Cancer Center, University of California San Diego,
CA, USA. 7Department of Cardiothoracic Surgery, University of Utah, Salt
Lake City, UT, USA.
Authors’ contributions
FL, SFJ, DTA, FR, VB, VG, CAD, RDNC, ANP, EC, DRK contributed to literature
review, analysis and discussion, synthesis of concepts, writing of the
manuscript and proof-reading of the final draft.
Competing interests
David R Koos is a shareholder, as well as Chairman and CEO of Entest Bio.
Feng Lin is research director of Entest Bio. All other authors declare no
competing interest.
Received: 7 January 2010
Accepted: 16 February 2010 Published: 16 February 2010
References
1. Abdel-Latif A, Bolli R, Tleyjeh IM, Montori VM, Perin EC, Hornung CA, Zuba-
Surma EK, Al-Mallah M, Dawn B: Adult bone marrow-derived cells for
cardiac repair: a systematic review and meta-analysis. Arch Intern Med
2007, 167:989-997.
2. Martin-Rendon E, Brunskill SJ, Hyde CJ, Stanworth SJ, Mathur A, Watt SM:
Autologous bone marrow stem cells to treat acute myocardial infarction:
a systematic review. Eur Heart J 2008, 29:1807-1818.
3. Vet-Stem Regenerative Veterinary Medicine. http://www.vet-stem.com.
4. Riordan NH, Ichim TE, Min WP, Wang H, Solano F, Lara F, Alfaro M,
Rodriguez JP, Harman RJ, Patel AN, Murphy MP, Lee RR, Minev B: Nonexpanded
adipose stromal vascular fraction cell therapy for multiple
sclerosis. J Transl Med 2009, 7:29.
5. Motz GT, Eppert BL, Sun G, Wesselkamper SC, Linke MJ, Deka R,
Borchers MT: Persistence of lung CD8 T cell oligoclonal expansions upon
smoking cessation in a mouse model of cigarette smoke-induced
emphysema. J Immunol 2008, 181:8036-8043.
6. Maeno T, Houghton AM, Quintero PA, Grumelli S, Owen CA, Shapiro SD:
CD8+ T Cells are required for inflammation and destruction in cigarette
smoke-induced emphysema in mice. J Immunol 2007, 178:8090-8096.
7. Woodruff PG, Koth LL, Yang YH, Rodriguez MW, Favoreto S, Dolganov GM,
Paquet AC, Erle DJ: A distinctive alveolar macrophage activation state
induced by cigarette smoking. Am J Respir Crit Care Med 2005,
172:1383-1392.
8. Stefanska AM, Walsh PT: Chronic obstructive pulmonary disease: evidence
for an autoimmune component. Cell Mol Immunol 2009, 6:81-86.
9. Gonzalez-Rey E, Gonzalez MA, Varela N, O’Valle F, Hernandez-Cortes P,
Rico L, Buscher D, Delgado M: Human adipose-derived mesenchymal
stem cells reduce inflammatory and T cell responses and induce
regulatory T cells in vitro in rheumatoid arthritis. Ann Rheum Dis
69:241-248.
10. Lepelletier Y, Lecourt S, Arnulf B, Vanneaux V, Fermand JP, Menasche P,
Domet T, Marolleau JP, Hermine O, Larghero J: Galectin-1 and
Semaphorin-3A are two soluble factors conferring T cell
immunosuppression to bone marrow mesenchymal stem cell. Stem Cells
Dev 2009.
11. Tsyb AF, Petrov VN, Konoplyannikov AG, Saypina EV, Lepechina LA,
Kalsina S, Semenkova IV, Agaeva EV: In vitro inhibitory effect of
mesenchymal stem cells on zymosan-induced production of reactive
oxygen species. Bull Exp Biol Med 2008, 146:158-164.
12. Sun L, Akiyama K, Zhang H, Yamaza T, Hou Y, Zhao S, Xu T, Le A, Shi S:
Mesenchymal stem cell transplantation reverses multiorgan dysfunction
in systemic lupus erythematosus mice and humans. Stem Cells 2009,
27:1421-1432.
13. Feuerer M, Herrero L, Cipolletta D, Naaz A, Wong J, Nayer A, Lee J,
Goldfine AB, Benoist C, Shoelson S, Mathis D: Lean, but not obese, fat is
enriched for a unique population of regulatory T cells that affect
metabolic parameters. Nat Med 2009, 15:930-939.
14. Ogawa Y, Duru EA, Ameredes BT: Role of IL-10 in the resolution of airway
inflammation. Curr Mol Med 2008, 8:437-445.
15. Serrano-Mollar A, Nacher M, Gay-Jordi G, Closa D, Xaubet A, Bulbena O:
Intratracheal transplantation of alveolar type II cells reverses bleomycininduced
lung fibrosis. Am J Respir Crit Care Med 2007, 176:1261-1268.
16. Aslam M, Baveja R, Liang OD, Fernandez-Gonzalez A, Lee C, Mitsialis SA,
Kourembanas S: Bone marrow stromal cells attenuate lung injury in a
murine model of neonatal chronic lung disease. Am J Respir Crit Care Med
2009, 180:1122-1130.
17. van Haaften T, Byrne R, Bonnet S, Rochefort GY, Akabutu J, Bouchentouf M,
Rey-Parra GJ, Galipeau J, Haromy A, Eaton F, Chen M, Hashimoto K,
Abley D, Korbutt G, Archer SL, Thébaud B: Airway delivery of
mesenchymal stem cells prevents arrested alveolar growth in neonatal
lung injury in rats. Am J Respir Crit Care Med 2009, 180:1131-1142.
18. Zalewska-Kaszubska J, Obzejta D: Use of low-energy laser as adjunct
treatment of alcohol addiction. Lasers Med Sci 2004, 19:100-104.
19. Nikitin AV, Esaulenko IE, Shatalova OL: [Effectiveness of laser puncture in
elderly patients with bronchial asthma accompanied by chronic
rhinosinusitis]. Adv Gerontol 2008, 21:424-426.
20. Vasil’ev AP, Strel’tsova NN, Iu Senatorov N: [Laser irradiation in the
treatment of ischemic heart disease]. Vopr Kurortol Fizioter Lech Fiz Kult
2001, 10-13.
21. Maiman TH: Stimulated optical radiation in Ruby. Nature 187:493.
22. Roy D: Ablative facial resurfacing. Dermatol Clin 2005, 23:549-559, viii.
23. Brown MC: An evidence-based approach to patient selection for laser
vision correction. J Refract Surg 2009, 25:S661-667.
24. Brancaleon L, Moseley H: Laser and non-laser light sources for
photodynamic therapy. Lasers Med Sci 2002, 17:173-186.
25. Mester ESB, Tota JG: “Effect of laser on hair growth of mice”. Kiserl
Orvostud 1967, 19:628-631.
26. Mester E, Korenyi-Both A, Spiry T, Tisza S: The effect of laser irradiation on
the regeneration of muscle fibers (preliminary report). Z Exp Chir 1975,
8:258-262.
27. Posten W, Wrone DA, Dover JS, Arndt KA, Silapunt S, Alam M: Low-level
laser therapy for wound healing: mechanism and efficacy. Dermatol Surg
2005, 31:334-340.
28. Vladimirov YA, Osipov AN, Klebanov GI: Photobiological principles of
therapeutic applications of laser radiation. Biochemistry (Mosc) 2004,
69:81-90.
29. Hu WP, Wang JJ, Yu CL, Lan CC, Chen GS, Yu HS: Helium-neon laser
irradiation stimulates cell proliferation through photostimulatory effects
in mitochondria. J Invest Dermatol 2007, 127:2048-2057.
30. Moriyama Y, Nguyen J, Akens M, Moriyama EH, Lilge L: In vivo effects of
low level laser therapy on inducible nitric oxide synthase. Lasers Surg
Med 2009, 41:227-231.
31. Samoilova KA, Zhevago NA, Petrishchev NN, Zimin AA: Role of nitric oxide
in the visible light-induced rapid increase of human skin
microcirculation at the local and systemic levels: II. healthy volunteers.
Photomed Laser Surg 2008, 26:443-449.
32. Yamaura M, Yao M, Yaroslavsky I, Cohen R, Smotrich M, Kochevar IE: Low
level light effects on inflammatory cytokine production by rheumatoid
arthritis synoviocytes. Lasers Surg Med 2009, 41:282-290.
33. Shiba H, Tsuda H, Kajiya M, Fujita T, Takeda K, Hino T, Kawaguchi H,
Kurihara H: Neodymium-doped yttrium-aluminium-garnet laser
irradiation abolishes the increase in interleukin-6 levels caused by
peptidoglycan through the p38 mitogen-activated protein kinase
pathway in human pulp cells. J Endod 2009, 35:373-376.
34. de Lima Mafra F, Costa MS, Albertini R, Silva JA Jr, Aimbire F: Low level
laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-
adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat
bronchi segments in E. coli lipopolysaccharide-induced airway
inflammation by a NF-kappaB dependent mechanism. Lasers Surg Med
2009, 41:68-74.
35. Aimbire F, de Oliveira Ligeiro AP, Albertini R, Correa JC, de Campos
Ladeira CB, Lyon JP, Silva JA Jr, Costa MS: Low level laser therapy (LLLT)
decreases pulmonary microvascular leakage, neutrophil influx and IL-
1beta levels in airway and lung from rat subjected to LPS-induced
inflammation. Inflammation 2008, 31:189-197.
36. Safavi SM, Kazemi B, Esmaeili M, Fallah A, Modarresi A, Mir M: Effects of
low-level He-Ne laser irradiation on the gene expression of IL-1beta,
TNF-alpha, IFN-gamma, TGF-beta, bFGF, and PDGF in rat’s gingiva. Lasers
Med Sci 2008, 23:331-335.
37. Saygun I, Karacay S, Serdar M, Ural AU, Sencimen M, Kurtis B: Effects of
laser irradiation on the release of basic fibroblast growth factor (bFGF),
insulin like growth factor-1 (IGF-1), and receptor of IGF-1 (IGFBP3) from
gingival fibroblasts. Lasers Med Sci 2008, 23:211-215.
38. Schwartz F, Brodie C, Appel E, Kazimirsky G, Shainberg A: Effect of helium/
neon laser irradiation on nerve growth factor synthesis and secretion in
skeletal muscle cultures. J Photochem Photobiol B 2002, 66:195-200.
39. Yu W, Naim JO, Lanzafame RJ: The effect of laser irradiation on the
release of bFGF from 3T3 fibroblasts. Photochem Photobiol 1994,
59:167-170.
40. Zungu IL, Hawkins Evans D, Abrahamse H: Mitochondrial responses of
normal and injured human skin fibroblasts following low level laser
irradiation–an in vitro study. Photochem Photobiol 2009, 85:987-996.
41. Wu S, Xing D, Gao X, Chen WR: High fluence low-power laser irradiation
induces mitochondrial permeability transition mediated by reactive
oxygen species. J Cell Physiol 2009, 218:603-611.
42. Lan CC, Wu CS, Chiou MH, Chiang TY, Yu HS: Low-energy helium-neon
laser induces melanocyte proliferation via interaction with type IV
collagen: visible light as a therapeutic option for vitiligo. Br J Dermatol
2009, 161:273-280.
43. Karu T: Photobiology of low-power laser effects. Health Phys 1989,
56:691-704.
44. Tiphlova O, Karu T: Role of primary photoacceptors in low-power laser
effects: action of He-Ne laser radiation on bacteriophage T4-Escherichia
coli interaction. Lasers Surg Med 1989, 9:67-69.
45. Karu TI: Mitochondrial signaling in mammalian cells activated by red and
near-IR radiation. Photochem Photobiol 2008, 84:1091-1099.
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
46. Zhang L, Xing D, Zhu D, Chen Q: Low-power laser irradiation inhibiting
Abeta25-35-induced PC12 cell apoptosis via PKC activation. Cell Physiol
Biochem 2008, 22:215-222.
47. Aimbire F, Santos FV, Albertini R, Castro-Faria-Neto HC, Mittmann J,
Pacheco-Soares C: Low-level laser therapy decreases levels of lung
neutrophils anti-apoptotic factors by a NF-kappaB dependent
mechanism. Int Immunopharmacol 2008, 8:603-605.
48. Lipovsky A, Nitzan Y, Lubart R: A possible mechanism for visible lightinduced
wound healing. Lasers Surg Med 2008, 40:509-514.
49. Ignatieva N, Zakharkina O, Andreeva I, Sobol E, Kamensky V, Lunin V: Effects
of laser irradiation on collagen organization in chemically induced
degenerative annulus fibrosus of lumbar intervertebral disc. Lasers Surg
Med 2008, 40:422-432.
50. Silveira LB, Prates RA, Novelli MD, Marigo HA, Garrocho AA, Amorim JC,
Sousa GR, Pinotti M, Ribeiro MS: Investigation of mast cells in human
gingiva following low-intensity laser irradiation. Photomed Laser Surg
2008, 26:315-321.
51. Coombe AR, Ho CT, Darendeliler MA, Hunter N, Philips JR, Chapple CC,
Yum LW: The effects of low level laser irradiation on osteoblastic cells.
Clin Orthod Res 2001, 4:3-14.
52. Gregoraszczuk E, Dobrowolski JW, Galas J: Effect of low intensity laser
beam on steroid dehydrogenase activity and steroid hormone
production in cultured porcine granulosa cells. Folia Histochem Cytochem
(Krakow) 1983, 21:87-92.
53. Yu HS, Chang KL, Yu CL, Chen JW, Chen GS: Low-energy helium-neon
laser irradiation stimulates interleukin-1 alpha and interleukin-8 release
from cultured human keratinocytes. J Invest Dermatol 1996, 107:593-596.
54. Mvula B, Mathope T, Moore T, Abrahamse H: The effect of low level laser
irradiation on adult human adipose derived stem cells. Lasers Med Sci
2008, 23:277-282.
55. Mvula B, Moore TJ, Abrahamse H: Effect of low-level laser irradiation and
epidermal growth factor on adult human adipose-derived stem cells.
Lasers Med Sci 25:33-39.
56. Hawkins DH, Abrahamse H: The role of laser fluence in cell viability,
proliferation, and membrane integrity of wounded human skin
fibroblasts following helium-neon laser irradiation. Lasers Surg Med 2006,
38:74-83.
57. Houreld N, Abrahamse H: In vitro exposure of wounded diabetic
fibroblast cells to a helium-neon laser at 5 and 16 J/cm2. Photomed Laser
Surg 2007, 25:78-84.
58. Smol’yaninova NK, Karu TI, Fedoseeva GE, Zelenin AV: Effects of He-Ne
laser irradiation on chromatin properties and synthesis of nucleic acids
in human peripheral blood lymphocytes. Biomed Sci 1991, 2:121-126.
59. Funk JO, Kruse A, Neustock P, Kirchner H: Helium-neon laser irradiation
induces effects on cytokine production at the protein and the mRNA
level. Exp Dermatol 1993, 2:75-83.
60. Funk JO, Kruse A, Kirchner H: Cytokine production after helium-neon laser
irradiation in cultures of human peripheral blood mononuclear cells. J
Photochem Photobiol B 1992, 16:347-355.
61. Gulsoy M, Ozer GH, Bozkulak O, Tabakoglu HO, Aktas E, Deniz G, Ertan C:
The biological effects of 632.8-nm low energy He-Ne laser on peripheral
blood mononuclear cells in vitro. J Photochem Photobiol B 2006,
82:199-202.
62. Novoselova EG, Cherenkov DA, Glushkova OV, Novoselova TV,
Chudnovskii VM, Iusupov VI, Fesenko EE: [Effect of low-intensity laser
radiation (632.8 nm) on immune cells isolated from mice]. Biofizika 2006,
51:509-518.
63. Dube A, Bansal H, Gupta PK: Modulation of macrophage structure and
function by low level He-Ne laser irradiation. Photochem Photobiol Sci
2003, 2:851-855.
64. Hemvani N, Chitnis DS, Bhagwanani NS: Helium-neon and nitrogen laser
irradiation accelerates the phagocytic activity of human monocytes.
Photomed Laser Surg 2005, 23:571-574.
65. Moustsen PA, Vinter N, Aas-Andersen L, Kragstrup J: [Laser treatment of
sinusitis in general practice assessed by a double-blind controlled
study]. Ugeskr Laeger 1991, 153:2232-2234.
66. Shen X, Zhao L, Ding G, Tan M, Gao J, Wang L, Lao L: Effect of combined
laser acupuncture on knee osteoarthritis: a pilot study. Lasers Med Sci
2009, 24:129-136.
67. Ekim A, Armagan O, Tascioglu F, Oner C, Colak M: Effect of low level laser
therapy in rheumatoid arthritis patients with carpal tunnel syndrome.
Swiss Med Wkly 2007, 137:347-352.
68. Minatel DG, Frade MA, Franca SC, Enwemeka CS: Phototherapy promotes
healing of chronic diabetic leg ulcers that failed to respond to other
therapies. Lasers Surg Med 2009, 41:433-441.
69. Bocci V, Travagli V, Zanardi I: May oxygen-ozone therapy improves
cardiovascular disorders?. Cardiovasc Hematol Disord Drug Targets 2009,
9:78-85.
70. Bocci V, Borrelli E, Travagli V, Zanardi I: The ozone paradox: ozone is a
strong oxidant as well as a medical drug. Med Res Rev 2009, 29:646-682.
71. Re L, Mawsouf MN, Menendez S, Leon OS, Sanchez GM, Hernandez F:
Ozone therapy: clinical and basic evidence of its therapeutic potential.
Arch Med Res 2008, 39:17-26.
72. Damera G, Zhao H, Wang M, Smith M, Kirby C, Jester WF, Lawson JA,
Panettieri RA Jr: Ozone modulates IL-6 secretion in human airway
epithelial and smooth muscle cells. Am J Physiol Lung Cell Mol Physiol
2009, 296:L674-683.
73. Manzer R, Dinarello CA, McConville G, Mason RJ: Ozone exposure of
macrophages induces an alveolar epithelial chemokine response
through IL-1alpha. Am J Respir Cell Mol Biol 2008, 38:318-323.
74. McDonald RJ, Usachencko J: Neutrophils injure bronchial epithelium after
ozone exposure. Inflammation 1999, 23:63-73.
75. Rodriguez ZZ, Guanche D, Alvarez RG, Rosales FH, Alonso Y, Schulz S:
Preconditioning with ozone/oxygen mixture induces reversion of some
indicators of oxidative stress and prevents organic damage in rats with
fecal peritonitis. Inflamm Res 2009.
76. Zamora ZB, Borrego A, Lopez OY, Delgado R, Gonzalez R, Menendez S,
Hernandez F, Schulz S: Effects of ozone oxidative preconditioning on
TNF-alpha release and antioxidant-prooxidant intracellular balance in
mice during endotoxic shock. Mediators Inflamm 2005, 2005:16-22.
77. Borrego A, Zamora ZB, Gonzalez R, Romay C, Menendez S, Hernandez F,
Montero T, Rojas E: Protection by ozone preconditioning is mediated by
the antioxidant system in cisplatin-induced nephrotoxicity in rats.
Mediators Inflamm 2004, 13:13-19.
78. Martinez-Sanchez G, Al-Dalain SM, Menendez S, Re L, Giuliani A, Candelario-
Jalil E, Alvarez H, Fernandez-Montequin JI, Leon OS: Therapeutic efficacy of
ozone in patients with diabetic foot. Eur J Pharmacol 2005, 523:151-161.
79. Surinchak JS, Alago ML, Bellamy RF, Stuck BE, Belkin M: Effects of low-level
energy lasers on the healing of full-thickness skin defects. Lasers Surg
Med 1983, 2:267-274.
80. Broadley C, Broadley KN, Disimone G, Riensch L, Davidson JM: Low-energy
helium-neon laser irradiation and the tensile strength of incisional
wounds in the rat. Wound Repair Regen 1995, 3:512-517.
81. Ghamsari SM, Taguchi K, Abe N, Acorda JA, Yamada H: Histopathological
effect of low-level laser therapy on sutured wounds of the teat in dairy
cattle. Vet Q 1996, 18:17-21.
82. Ghamsari SM, Taguchi K, Abe N, Acorda JA, Sato M, Yamada H: Evaluation
of low level laser therapy on primary healing of experimentally induced
full thickness teat wounds in dairy cattle. Vet Surg 1997, 26:114-120.
83. Pinfildi CE, Liebano RE, Hochman BS, Ferreira LM: Helium-neon laser in
viability of random skin flap in rats. Lasers Surg Med 2005, 37:74-77.
84. Maiya GA, Kumar P, Rao L: Effect of low intensity helium-neon (He-Ne)
laser irradiation on diabetic wound healing dynamics. Photomed Laser
Surg 2005, 23:187-190.
85. Bayat M, Vasheghani MM, Razavi N, Taheri S, Rakhshan M: Effect of lowlevel
laser therapy on the healing of second-degree burns in rats: a
histological and microbiological study. J Photochem Photobiol B 2005,
78:171-177.
86. Garavello I, Baranauskas V, da Cruz-Hofling MA: The effects of low laser
irradiation on angiogenesis in injured rat tibiae. Histol Histopathol 2004,
19:43-48.
87. Lin YS, Huang MH, Chai CY, Yang RC: Effects of helium-neon laser on
levels of stress protein and arthritic histopathology in experimental
osteoarthritis. Am J Phys Med Rehabil 2004, 83:758-765.
88. Jamieson RW, Friend PJ: Organ reperfusion and preservation. Front Biosci
2008, 13:221-235.
89. Akai M, Usuba M, Maeshima T, Shirasaki Y, Yasuoka S: Laser’s effect on
bone and cartilage change induced by joint immobilization: an
experiment with animal model. Lasers Surg Med 1997, 21:480-484.
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
90. Nakano J, Kataoka H, Sakamoto J, Origuchi T, Okita M, Yoshimura T: Lowlevel
laser irradiation promotes the recovery of atrophied gastrocnemius
skeletal muscle in rats. Exp Physiol 2009, 94:1005-1015.
91. Doin-Silva R, Baranauskas V, Rodrigues-Simioni L, da Cruz-Hofling MA: The
ability of low level laser therapy to prevent muscle tissue damage
induced by snake venom. Photochem Photobiol 2009, 85:63-69.
92. Gal D, Chokshi SK, Mosseri M, Clarke RH, Isner JM: Percutaneous delivery of
low-level laser energy reverses histamine-induced spasm in
atherosclerotic Yucatan microswine. Circulation 1992, 85:756-768.
93. Kipshidze N, Sahota H, Komorowski R, Nikolaychik V, Keelan MH Jr:
Photoremodeling of arterial wall reduces restenosis after balloon
angioplasty in an atherosclerotic rabbit model. J Am Coll Cardiol 1998,
31:1152-1157.
94. Novoselova EG, Glushkova OV, Cherenkov DA, Chudnovsky VM, Fesenko EE:
Effects of low-power laser radiation on mice immunity. Photodermatol
Photoimmunol Photomed 2006, 22:33-38.
95. Bensadoun RJ, Franquin JC, Ciais G, Darcourt V, Schubert MM, Viot M,
Dejou J, Tardieu C, Benezery K, Nguyen TD, Laudoyer Y, Dassonville O,
Poissonnet G, Vallicioni J, Thyss A, Hamdi M, Chauvel P, Demard F: Lowenergy
He/Ne laser in the prevention of radiation-induced mucositis. A
multicenter phase III randomized study in patients with head and neck
cancer. Support Care Cancer 1999, 7:244-252.
96. Arun Maiya G, Sagar MS, Fernandes D: Effect of low level helium-neon
(He-Ne) laser therapy in the prevention & treatment of radiation
induced mucositis in head & neck cancer patients. Indian J Med Res 2006,
124:399-402.
97. Neiburger EJ: Rapid healing of gingival incisions by the helium-neon
diode laser. J Mass Dent Soc 1999, 48:8-13, 40..
98. Aras MH, Gungormus M: Placebo-controlled randomized clinical trial of
the effect two different low-level laser therapies (LLLT)-intraoral and
extraoral-on trismus and facial swelling following surgical extraction of
the lower third molar. Lasers Med Sci 2009.
99. Zycinski P, Krzeminska-Pakula M, Peszynski-Drews C, Kierus A, Trzos E,
Rechcinski T, Figiel L, Kurpesa M, Plewka M, Chrzanowski L, Drozdz J: Laser
biostimulation in end-stage multivessel coronary artery disease–a
preliminary observational study. Kardiol Pol 2007, 65:13-21, discussion 22-
13..
100. Zivin JA, Albers GW, Bornstein N, Chippendale T, Dahlof B, Devlin T,
Fisher M, Hacke W, Holt W, Ilic S, Kasner S, Lew R, Nash M, Perez J,
Rymer M, Schellinger P, Schneider D, Schwab S, Veltkamp R, Walker M,
Streeter J, NeuroThera Effectiveness and Safety Trial-2 Investigators:
Effectiveness and safety of transcranial laser therapy for acute ischemic
stroke. Stroke 2009, 40:1359-1364.
101. Osiris Therapeutics Reports Interim Data for COPD Trial. http://www.
medicalnewstoday.com/articles/155267.php.
102. Gavish L, Perez LS, Reissman P, Gertz SD: Irradiation with 780 nm diode
laser attenuates inflammatory cytokines but upregulates nitric oxide in
lipopolysaccharide-stimulated macrophages: implications for the
prevention of aneurysm progression. Lasers Surg Med 2008, 40:371-378.
103. Correa F, Lopes Martins RA, Correa JC, Iversen VV, Joenson J, Bjordal JM:
Low-level laser therapy (GaAs lambda = 904 nm) reduces inflammatory
cell migration in mice with lipopolysaccharide-induced peritonitis.
Photomed Laser Surg 2007, 25:245-249.
104. Aimbire F, Lopes-Martins RA, Castro-Faria-Neto HC, Albertini R,
Chavantes MC, Pacheco MT, Leonardo PS, Iversen VV, Bjordal JM: Low-level
laser therapy can reduce lipopolysaccharide-induced contractile force
dysfunction and TNF-alpha levels in rat diaphragm muscle. Lasers Med
Sci 2006, 21:238-244.
105. de Morais NC, Barbosa AM, Vale ML, Villaverde AB, de Lima CJ, Cogo JC,
Zamuner SR: Anti-Inflammatory Effect of Low-Level Laser and Light-
Emitting Diode in Zymosan-Induced Arthritis. Photomed Laser Surg 2009.
106. Boschi ES, Leite CE, Saciura VC, Caberlon E, Lunardelli A, Bitencourt S,
Melo DA, Oliveira JR: Anti-Inflammatory effects of low-level laser therapy
(660 nm) in the early phase in carrageenan-induced pleurisy in rat.
Lasers Surg Med 2008, 40:500-508.
107. Shimizu N, Mayahara K, Kiyosaki T, Yamaguchi A, Ozawa Y, Abiko Y: Lowintensity
laser irradiation stimulates bone nodule formation via insulinlike
growth factor-I expression in rat calvarial cells. Lasers Surg Med 2007,
39:551-559.
108. Hackett TL, Shaheen F, Johnson A, Wadsworth S, Pechkovsky DV,
Jacoby DB, Kicic A, Stick SM, Knight DA: Characterization of side
population cells from human airway epithelium. Stem Cells 2008,
26:2576-2585.
109. Irwin D, Helm K, Campbell N, Imamura M, Fagan K, Harral J, Carr M,
Young KA, Klemm D, Gebb S, Dempsey EC, West J, Majka S: Neonatal lung
side population cells demonstrate endothelial potential and are altered
in response to hyperoxia-induced lung simplification. Am J Physiol Lung
Cell Mol Physiol 2007, 293:L941-951.
110. Thebaud B, Ladha F, Michelakis ED, Sawicka M, Thurston G, Eaton F,
Hashimoto K, Harry G, Haromy A, Korbutt G, Archer SL: Vascular
endothelial growth factor gene therapy increases survival, promotes
lung angiogenesis, and prevents alveolar damage in hyperoxia-induced
lung injury: evidence that angiogenesis participates in alveolarization.
Circulation 2005, 112:2477-2486.
111. Thebaud B: Angiogenesis in lung development, injury and repair:
implications for chronic lung disease of prematurity. Neonatology 2007,
91:291-297.
112. Schindl A, Merwald H, Schindl L, Kaun C, Wojta J: Direct stimulatory effect
of low-intensity 670 nm laser irradiation on human endothelial cell
proliferation. Br J Dermatol 2003, 148:334-336.
113. Kipshidze N, Nikolaychik V, Keelan MH, Shankar LR, Khanna A, Kornowski R,
Leon M, Moses J: Low-power helium: neon laser irradiation enhances
production of vascular endothelial growth factor and promotes growth
of endothelial cells in vitro. Lasers Surg Med 2001, 28:355-364.
114. Ihsan FR: Low-level laser therapy accelerates collateral circulation and
enhances microcirculation. Photomed Laser Surg 2005, 23:289-294.
115. Taguchi A, Soma T, Tanaka H, Kanda T, Nishimura H, Yoshikawa H,
Tsukamoto Y, Iso H, Fujimori Y, Stern DM, Naritomi H, Matsuyama T:
Administration of CD34+ cells after stroke enhances neurogenesis via
angiogenesis in a mouse model. J Clin Invest 2004, 114:330-338.
116. Li WT, Leu YC: Effects of low level red-light irradiation on the
proliferation of mesenchymal stem cells derived from rat bone marrow.
Conf Proc IEEE Eng Med Biol Soc 2007, 2007:5830-5833.
117. Tuby H, Maltz L, Oron U: Low-level laser irradiation (LLLI) promotes
proliferation of mesenchymal and cardiac stem cells in culture. Lasers
Surg Med 2007, 39:373-378.
118. Tuby H, Maltz L, Oron U: Implantation of low-level laser irradiated
mesenchymal stem cells into the infarcted rat heart is associated with
reduction in infarct size and enhanced angiogenesis. Photomed Laser
Surg 2009, 27:227-233.
119. Agaiby AD, Ghali LR, Wilson R, Dyson M: Laser modulation of angiogenic
factor production by T-lymphocytes. Lasers Surg Med 2000, 26:357-363.
120. Zhang H, Hou JF, Shen Y, Wang W, Wei YJ, Hu S: Low Level Laser
Irradiation Precondition to Create Friendly Milieu of Infarcted
Myocardium and Enhance Early Survival of Transplanted Bone Marrow
Cells. J Cell Mol Med 2009.
121. Hou JF, Zhang H, Yuan X, Li J, Wei YJ, Hu SS: In vitro effects of low-level
laser irradiation for bone marrow mesenchymal stem cells: proliferation,
growth factors secretion and myogenic differentiation. Lasers Surg Med
2008, 40:726-733.
doi:10.1186/1479-5876-8-16
Cite this article as: Lin et al.: Lasers, stem cells, and COPD. Journal of
Translational Medicine 2010 8:16.
Original Source: http://www.translational-medicine.com/content/8/1/16
Light Therapy to Stem Cells: A New Therapeutic Approach in Regenerative Medicine – A Mini Review
Amir Oron1 and Uri Oron2* - 15 April 2015 (Publication) 4489
View Resource
Abstract
Regenerative capacity following injury or an ischemic event is confined to non mammalian vertebrates. Mammals have a limited capacity to restore organs
following injury to organs like the liver and skeletal muscles but practically no ability to regenerate organs like the heart or brain following an ischemic event
or injury. We tried a new approach in cell based therapy to improve regeneration in various organs following ischemic injury. Low-level laser therapy (LLLT)
which has photobiostimulating effects on cells was delivered to autologous bone marrow (BM) that is enriched with stem cells and various progenitor cells, in
order to induce the cells in the BM for the benefit of the injured /ischemic organs. In a model of induced myocardial infarction (MI) in rats laser application
to the BM caused a marked and significant decrease (79%) in infarct size (scarring) 3 weeks post-MI. It was also found that a significantly higher density of
c-kit positive cells (a marker of mesenchymal stem cells) in the myocardium of laser-treated rats relative to non-treated rat’s post-MI. The novel approach
presented in this study, of the use of stem cells for cell therapy to the infracted heart, avoids the need to isolate millions of stem cells, to grow them in vitro and
to inject them back into the patient. In the same line of rationale we tried to find whether LLLT to the BM could be beneficial also to kidney impairment after
ischemic reperfusion injury (IRI) to the rat kidney. C-kit positive cell density in kidneys post-IRI and laser-treatment was significantly (p=0.05) 2.4-fold higher
compared to the non laser treated group. Creatinine, blood urea nitrogen, and cystatin-C levels were significantly lower in the laser-treated rats as compared
to non-treated ones. The effect of LLLT delivery to BM was also tested on Alzheimer’s disease (AD) mice in their late stage of the disease. Mice were given
multiple (every 10 days) LLLT to BM from age 4 to 6 months. It was found that in the treated AD-mice neurological tests (Fear and Cognitive tests) revealed
a significantly (p<0.05) better neurological performance and cognitive capacity compared to the non-treated AD mice. Furthermore, concomitantly with the
improved neurological performance, ß-amyloid density in the hippocampal region of the brains was revealed to be significantly less in the laser-treated mice
as compared to control. In conclusion, a novel approach, of applying LLLT to autologeous BM in order to induce stem cells that are consequently recruited to the
injured/ischemic organ leading to a marked beneficial effect post-ischemic event or degenerative process is presented. This approach is novel in the respect
that it is stimulating the patient’s own abilities to initiate a regenerative response in an organ by the utilization of light. The possibility that this approach can
also be applied to other ischemic/injured organs or organs undergoing degenerative processes (i.e. neurodegenerative diseases), with consequent beneficial
effects, cannot be ruled out
CELL THERAPY FOR INJURED/ISCHEMIC ORGANS
Clinical trials have lately been implemented in a growing
abundance due to the extensive research and new approaches of
cell based therapies for the reconstruction of impaired organs.
Regenerative capacity following injury or an ischemic event
is confined to non mammalian vertebrates. In particular, fish
and primitive amphibians can regenerate organs like the heart,
brain and limbs. However, mammals have a limited capacity
to restore organs following injury to organs like the liver and
skeletal muscles but practically no ability to regenerate organs
like the heart or brain following an ischemic event or injury.
The mammalian heart, including the human heart, for example,
has a very limited capacity to regenerate following damage or
an acute ischemic event like myocardial infarction (MI). This is
due to the very low level of cardiomyocyte proliferation and the
limited number of cells expressing stem-cell marker proteins.
Stem-cell-based therapy was suggested as a potential solution
to the above situation. In recent years, cell-based therapy for
cardiac repair in particular has undergone a rapid transition
from basic science research to clinical reality [1-3]. The general
outcome of the clinical trials was that the procedures and longterm
outcome post-stem-cell implantation to the heart via the
coronary arteries are safe. However, improvement in long-term
functional performance of the heart was either not achieved or
was marginal [1-3].
There are several central issues pertaining to the use of cell
implantation in stem-cell therapy: the number of implanted stem
cells has to be high since there is massive cell death following
implantation or injection of cells into the heart or the blood
circulation. Another central issue in stem-cell implantation for
organ repair is the creation of a receptive cell environment in the
ischemic organ. Several factors (e.g. inhibition of inflammation
and apoptosis, secretion of cell growth factors etc.) are necessary
for optimal cell implantation [4]. The injected cells may have to
migrate from the circulating blood to the ischemic niche. They
can then remain active and secrete growth factors, exerting a paracrine effect on the ischemic tissue [5]. Alternatively, they
may stimulate the small population of stem cells in the ischemic
organ (such as the heart), to proliferate and differentiate so as to
enhance cardiac repair post-MI [6]. Another issue is the timing
of injection of the stem cells to the infarcted heart and effect of
MI (inflammatory phase) on the BM [7]. Photobiostimulation
of cells in the bone marrow (BM), that is enriched with various
progenitor cells, by low level laser therapy (LLLT) may suggest a
new approach that may overcome some of the above limitation.
This new approach will be discussed in the present mini review
[8-11].
LOW LEVEL LASER THERAPY FOR THE ISCHEMIC
HEART
In general LLLT has been found to modulate various biological
processes, such as increasing mitochondrial respiration and ATP
synthesis, facilitating wound healing, and promoting the process
of skeletal muscle regeneration and angiogenesis [12,13]. It
was previously shown that LLLT can enhance skeletal muscle
regeneration following partial excision in the rat hind limb
muscles when the laser was delivered directly to the injured
organ multiple times (for 2 min each time) following injury [14].
This phenomenon was even more prominent following cold
injury to the frog skeletal muscles indicating that enhancement
of regeneration by LLLT is probably a general phenomenon in
vertebrates and maybe more effective in cold blooded animals
which innately have a lower metabolic rate in their cells [13-15].
In an experimental model of the infarcted heart in rats and dogs, it
was demonstrated that LLLT (Diode –Ga-Al-As 810nm at a power
density of 5 mW/cm2 for 120sec duration of laser exposure
comprising 0.6 J/cm2), application directly to the infarcted area
in the heart at optimal power parameters significantly reduces
infarct size (scar tissue formation) [16,17]. This phenomenon
was partially attributed to a significant elevation in ATP content,
heat shock proteins, vascular endothelial growth factor (VEGF),
and angiogenesis in the ischemic zone of the laser-irradiated
rats, as compared to non-irradiated rats [16,17]. The mechanism
associated with the photobiostimulation by LLLT is not yet
clearly understood [12]. There is evidence that cytochrome c
oxidase and perhaps also plasma membranes in cells function as
photoacceptors of the photons, and thereafter a cascade of events
occur in the mitochondria, leading to effects on various processes
like ATP production, up-regulation of VEGF, etc [12].
The effect of photobiostimulation on stem cells or progenitor
cells has not been extensively studied [18-21]. It was previously
shown that laser application (Diode laser at 50mW/cm2 for
100sec, energy density 0.5 mW/cm2) to the mesenchymal stem
cells isolated from bone marrow or cardiac stem cells causes a
significant increase in their proliferation in vitro [20]. Based on
previous studies that showed an increase in cytoprotective effect
on the ischemic heart following LLLT, a new approach was taken
to apply laser irradiation to stem cells grown in culture prior
to their implantation to the infarcted heart as a cell therapy for
heart repair [21]. In that study it was demonstrated that MSCs
that were laser treated prior to their implantation to the rat
infarcted heart caused a significant reduction in infarct size
as compared to MSCs that were injected to the heart without
prior laser treatment. This phenomenon was also associated
with significant elevation of vascular endothelial growth factor
(VEGF) in the myocardium of the rats that received the lasertreated
MSCs. In a recent study [8] the possibility of recruiting
autologous stem cells stimulated by LLLT in the BM to the
infarcted heart was addressed. The rationale behind the attempt
to use LLLT to induce the “crude” BM in the bone was, and still is,
that one cannot significantly affect the complex process post-MI
or ischemic injury to the kidney with a single type of stem cell. The
native BM is known for its many types and subtypes of stem cells,
which are defined by their reactivity to various antibodies. The
BM also contains many progenitor cells (i.e. monocytes) that can
further differentiate, for example to macrophages. Macrophages
have been shown recently to have a crucial role in the scarring
process post-MI. Thus LLLT may induce concomitantly in the BM
various types of cells that will increase in number in the blood
circulation following their enhanced proliferation in the BM.
These cells will probably, eventually, and to a certain extent and
under certain circumstances, home in on the ischemic zone in the
ischemic organ (heart, kidney etc.). In this study [8] it was found
that when LLLT was applied in vivo to the BM, and MSCs were
isolated from that BM 3 and 6 weeks later and grown in vitro, they
grew at a higher rate of proliferation relative to MSCs isolated
from non-laser-treated BM. This indicated that the MSCs when
in the BM, following LLLT application in vivo can be induced to
proliferate to a higher rate than non-treated MSCs. Furthermore,
laser application (Diode laser 808nm at power density of 10mW/
cm2 for 100 sec comprising 1J/cm2 energy density) to the BM (at
about 20 min post-MI) caused a marked and significant decrease
(79%) in infarct size 3 weeks post-MI. This extent of infarct size
reduction was even more effective in reducing scarring than that
of laser application directly to the infarcted heart, as also found
in previous studies with infarcted rat and dog hearts [10]. Even
when laser was applied 4 hours post-MI to the BM of infarcted
rats, a marked and significant reduction in the infarcted area
was observed in the laser-treated rats compared to control.
We also found a significantly higher density of c-kit+ (a marker
of MSCs) cells in the myocardium of laser-treated rats relative
to non-treated rat’s post-MI. Moreover, it was demonstrated in
this study that c-kit+ cells post-laser application to the BM of MIinduced
rats, homed specifically in on the infarcted heart and
not on uninjured organs (i.e. liver, kidney) in the same rat [16].
It can be hypothesized that the increased number of c-kit+ cells
found in the myocardium came from proliferating MSCs in the
BM that had migrated to the circulating blood and homed onto
the infarcted heart. Another finding of this study was that of
the preferred homing of the recruited or endogenous c-kit+ cells
in on the infarcted area, rather than their random deposition
throughout the left ventricle in the heart. Indeed, at 3-weeks
post-MI the density of c-kit+ cells in the infarcted area was 27-
fold higher in the rats whose BM had been treated with LLLT as
compared to control rats. Similarly, Hatzistergos et al. [6] found
that endogenous c-kit+ cardiac stem cells increased by 20-fold
in the porcine infarcted heart as compared to control following
Effect of autologous mesenchymal stem cells induced by low level laser therapy on cardiogenesis in the infarcted area following myocardial infarction in rats
Hana Tuby1, Tali Yaakobi1, Lidya Maltz1, Yaakov Delarea2, Orit Sagi-Assif2, Uri Oron1* - (Publication) 4467This study showed rats that were give a heart attack and then treated with the laser on their shins saw a 55% reduction in infarction size in the heart showing that the stem cells released from the bone migrated to the heart.
1Department of Zoology, The George S. Wise Faculty of Life Sciences, Tel-Aviv University, Tel-Aviv, Israel
2Department of Cell Biology and Immunology, The George S. Wise Faculty of Life Sciences, Tel-Aviv University, Tel-Aviv, Israel
Email: *oronu@post.tau.ac.il
Received 27 May 2013; revised 29 June 2013; accepted 16 July 2013
ABSTRACT
In this study, we investigated the hypothesis that photo- biostimulation by low-energy laser therapy (LLLT) applied to the bone marrow (BM) of myocardial in- farcted rats may attenuate the scarring processes that follow myocardial infarction (MI). Wistar rats under- went experimental MI. LLLT (Ga-Al-As diode laser) was applied to the BM of the exposed tibia at differ- ent time intervals post-MI (4 hrs, 48 hrs and 5 days). Sham-operated infarcted rats served as control. In- farct size was significantly reduced (55%) in the la- ser-treated rats as compared to the control non-treat- ed rats, at 2 weeks post-MI. A significant 3-fold in- crease was observed in the density of desmin immu- nopositive stained cells 14 days post-MI in the infarc- ted area of the laser-treated rats as compared to the non-laser-treated controls. The electron microscopy from the control infarcted rat hearts revealed a typi- cal interphase area between the intact myocardium and the infarcted area, with conspicuous fibroblasts with collagen deposition dispersed among them. In rats that were laser treated (to BM), the interphase zone demonstrated cells with different intracellular struc- tures. There was also a significant increase in the per- centage of c-kit positive cells and macrophages in the circulating blood of the laser treated rats as compar- ed to control non treated ones. In the majority of the cells clusters of myofibrils anchored to well-developed Z-lines and structures resembling the morphological characteristics of mature intact cardiomyocytes were evident. In conclusion, LLLT to the BM of rats post- MI induces cardiogenesis mainly at the borders of the infarcted area in the heart.
Keywords: Low-Level Laser Therapy; Myocardial Infarction; Macrophage; Desmin; Ultrastructure; c-Kit Positive Cells
1. INTRODUCTION
Regenerative capacity and mitotic activity in the heart are confined mainly to the lower vertebrates [1]. Amputation of ~20% of the zebrafish’s ventricular myocardium re- sulted in full regeneration without scarring [2]. In am- phibians, heart injury was associated with increased cell proliferation of myocytes and enhanced regeneration [3]. The adult mammalian heart was traditionally considered to be a post-mitotic organ with terminally differentiated cardiac myocytes. However, this dogma has recently been challenged by several studies and reviews [4-8]. These studies have suggested that cardiac myocytes are replaced throughout the lifespan even in the human heart, and that myocytes can regenerate from resident cardiac progenitor cells (CPC) as well as from bone marrow (BM). Studies in human infarcted hearts have shown evidence of cytoki- nesis of cells in the heart and evidence of cardiac stem cells that are activated in response to ischemic injury. This growth response is attenuated in chronic heart fail- ure [9]. Some studies have reported that cardiac myocyt- es can be derived from BM; specifically, side population precursor cells following induction of myocardial infarc- tion (MI) by left anterior descending artery (LAD) liga- tion [10-12]. Contradicting these findings, other laborato- ries using genetic markers have reported that lineage ne- gative, c-kit+ BM cells did not differentiate into cardio- myocytes [13]. It was also suggested that BM-derived stem cells may stimulate the small population of stem cells in the ischemic heart to proliferate and differentiate to enhance cardiac repair post-MI [14]. In a recent study transient regenerative potential in the mouse heart was demonstrated during the neonatal period [15].
Low-level laser therapy (LLLT) has been found to modulate various biological processes [16,17], such as increasing mitochondrial respiration and ATP synthesis [18], facilitating wound healing and promoting the proc- ess of skeletal muscle regeneration and angiogenesis [19- 21]. In an experimental model of the infarcted heart in rats and dogs, it was demonstrated that LLLT application directly to the infarcted area in the heart at optimal power parameters significantly reduced scar tissue formation [22-24]. This phenomenon was partially attributed to a significant elevation in ATP content, heat shock proteins, vascular endothelial growth factor (VEGF), inducible ni- tric oxide (NO) synthase, and angiogenesis in the ischemic zone of the laser-irradiated rats, as compared to non- irradiated rats [25].
The effect of photobiostimulation on stem cells or pro- genitor cells has not been extensively studied. LLLT ap- plication to normal human neural progenitor cells signi- ficantly increases ATP production in these cells [26]. LLLT delivery to MSCs and cardiac stem cells in vitro caused a significant enhancement in their proliferation rate [27,28]. LLLT has also been shown to increase the proliferation rate of adipose-derived stem cells in vitro [29]. Recently, we demonstrated that LLLT application to autologous BM could induce mesenchymal stem cells (MSCs) in the BM to proliferate and cause their recruit- ment and specific homing in on the infarcted rat heart and not on other organs [30,31]. The laser treatment to the BM also caused a marked and statistically significant reduction of 79% in the scarring and ventricular dilata- tion followed MI as compared to infarcted non-laser- treated rats. The aim of the present study was to investi- gate the possibility that induction of stem cells in the BM of rats by LLLT could also affect cardiogenesis in the in- farcted rat heart.
2. MATERIALS AND METHODS
2.1. Experimental Procedures
A total of 21 Wistar male rats, weighing 200 - 250 gr, that underwent ligation of the LAD artery to induce MI, were used as described by us previously [23]. All the ex- perimental procedures were approved by the animal care committee of Tel-Aviv University. Briefly, rats were anes- thetized with Avertin (1 ml/100 g body weight I.P.) and the lungs were ventilated. Thoractomy was performed by invasion of the intercostals muscles between the 5th and 6th rib to expose the heart. The LAD artery was occluded 2 mm from the origin with 5-0 polypropylene thread (Ethicon Inc., Cincinnati, OH). Following LAD artery occlusion the chest muscles and skin were sutured and the rats were ventilated until they woke up. The infarcted rats were divided randomly into two groups. In one group LLLT was applied directly to the BM 4 hrs, 48 hrs and 5 days post-MI (see below). The second group was non-laser-treated (the rat’s bone was exposed for the same duration as the laser-treated group but the laser was not turned on). Food and water were supplied ad libitum. Rats were sacrificed 14 days post-MI.
2.2. Laser Application
After induction of MI rats were randomly assigned to a laser-treated or control non-laser-treated group. A diode (Ga-Al-As) laser, wavelength 804 nm with a tunable po- wer output of maximum of 400 mW (Lasotronic Inc., Zug, Switzerland) for application to the BM was used. The laser device was equipped with a metal-backed glass fiber optic (1.5 mm diameter). An infrared viewer (Laso- tronic Inc. Zug, Switzerland) and infrared-sensitive de-tecting card (Newport, Inc., Irvine, CA) were used to de- termine the infrared irradiation area. Laser application was done by a 10 mm longitudinal cut in the skin above the medial aspect, and further delicate cleaning of the bone surface was carried out. The tip of the fiber optic (1.5 mm diameter) was placed perpendicularly to the center of the exposed medial aspect of the tibia and power den- sity of 10 mW/cm2 was applied to the BM. The laser was applied for a duration of 100 sec (energy density 1.0 J/cm2). Left or right exposed tibias were chosen at random for LLLT application. In sham-operated infarcted rats that served as control the tibias were exposed and the fi- ber optic was placed as described above but the laser beam was not turned on.
2.3. Histology and Electron Microscopy
A defined cross-section sample (2 mm thick) from the central part of the infarcted area was taken from all hearts for histology. Eight micron paraffin sections were pre- pared from the tissue samples of each heart. Infarct size was determined using Masson’s trichrome staining as described by us previously [23]. Three observers, blinded to control or laser-treated rats, analyzed infarct size. Six microscopic slides from the infarcted area of each heart were chosen at random for determination of infarct size. Infarct size was expressed as the percentage of the total infarcted area relative to the total area of the left ventri- cle (LV) in each section, using image analysis software Sigma Scan Pro (Sigma, St. Louis, MO).
For electron microscopy three tissue samples from each of the control and laser-irradiated rat hearts were taken from the interphase zone between the infarcted and non-infarcted tissue by macroscopic examination. Fixa- tion was performed in 3.5% glutaraldehyde in 0.1 M ca- codylate buffer for 24 hrs followed by embedment in Epon-812. Semi-thin sections (1 micron) were prepared in order to localize the interphase zone. Thin sections were then prepared and stained with uranyl acetate and lead citrate followed by examination with a Jeol electron microscope.
2.4. Immunohistochemistry
The total number of cells immunostained for desmin (bone marrow cells or newly formed) in the infarcted area were determined using a desmin kit (Zytomed Laboratory, Ber- lin, Germany). The procedure was performed at room temperature with anti-mouse (dilution 1:25 - 1:50) primary antibody for 60 min. Following washing, slides were in- cubated with HRP secondary antibody for mouse for 30 min followed by DAB Chromogen system (Covance Inc., Dedham). Slides were rinsed again in wash buffer, stain- ed in Hematoxylin for nuclei detection, mounted and viewed using a Zeiss microscope equipped with a camera and video screen. The total number of desmin immuno- stained cells within the infarcted area was counted and their density expressed as the percentage of the total area of the infarct using SigmaPro software.
2.5. Flow Cytometry Analysis
Blood samples were taken 2 and 7 days post-IR injury for fluorescence-activated cell sorting (FACS) analysis. 100 μl of blood were mixed with different antibodies: anti-mouse CD117 (c-kit) PE (eBioscience San Diego, USA) and rat IgG2b isotype control PE (eBioscience San Diego, USA) and anti-rat macrophage marker PE (eBio- science San Diego, USA) and mouse IgG2a K isotype control PE (eBioscience San Diego, USA), were used for the FACS analysis according to the manufacturer’s guide- lines. Forty five min post incubation of the whole fresh blood with the relevant antibodies, 2 ml of Fix/Lyse so- lution (eBioscience, San Diego, USA) was added. After mixture the suspended cells were left for 60 min in the dark at room temperature. Centrifugation was performed for 10 min, supernatant was removed and washing of the pellet was performed with 2 ml of Flow Cytometry Stain- ing Buffer Solution (eBioscience, San Diego, California, USA). After another centrifugation for 10 minutes the supernatant was decanted. The pellet containing mono- nucleated cells was resuspended in 200 μl of flow stain buffer for FACS analysis.
2.6. Statistical Analysis
The SigmaStat 2.0 (Sigma, St. Luis, USA) software was used for statistical analysis. Tests were performed first for normality distribution, followed by parametric (stu- dent’s t-test) test.
3. RESULTS
Application of LLLT to the infarcted heart caused a sig- nificant (p = 0.049) reduction of 55% in infarct size as compared to control. The present of macrophages and c- kit positive cells in the blood was determined by FACS analysis (Figure 1). It was found that at 5 days post MI there was a statistical significant 2-fold higher concentra- tion of macrophages and significant 1.4-fold higher c-kit positive cells (mesenchymal cells) in the laser treated rats as compared to the infarcted non laser treated rats. Des- min immunostaining of histological sections of the in- farcted zone from laser-treated rats demonstrated a higher density of positively stained cells than in the non laser-treated ones (Figures 2-4). In the interphase zone, cells extending from the myocardium towards the in 
Figure 1. Percent (out of total mononucleated cells) of macro- phages and c-kit positive cells in blood of control and laser treated rats (to the bone marrow) 5 days post MI as revealed by FACS analysis. The results are mean ± S.E.M of 15 rats at each group. Statistical significance *p < 0.05; **p < 0.01.

Figure 2. Representative desmin immunostained light micro- graphs of the infarcted zone of non-laser-treated rats (a, c) and laser-treated rats (to the bone marrow at 4 and 48 hrs and 5 days) (b, d) taken 2 weeks post-MI. Note that the zone in the control non-laser-treated rats contains mainly collageneous mate- rial with a few desmin immunopositive cells in the infarcted area (a, c); while in the laser-treated rats the zone displays posi- tive desmin staining in extended outgrowths (arrow) from the myocardium (MC) in (b), and in the cytoplasm of many cells in the infarcted area in (d). IF, Infarcted area. Bar = 50 μm.
farcted area showed higher immunostaining for desmin in the laser-treated rat hearts as compared to the control non-treated ones (Figure 2). The cell density of desmin immune-positive cells was also determined quantitatively in histological sections of both the infarcted laser-treated rats and infarcted non-laser-treated rats. The cell density was significantly (p < 0.01) 3-fold higher in the infarcted area of the laser-treated rats as compared to the non-la- ser-treated controls (Figure 4).
The electron micrographs of all samples taken from the control non-laser-treated infarcted rat hearts revealed a typical interphase area between intact and infarcted heart (Figure 5(a)). Adjacent to the non-ischemic intact myocardium there were conspicuous fibroblasts with col- lagen deposition dispersed among them (Figure 5(a)). In all samples taken from the laser-irradiated hearts the in- terphase zone between intact and infarcted area demon- strated different characteristics to those of the non-laser- treated infarcted rat hearts. Cells with newly-formed or- ganized contractile myofilaments dispersed in the cyto- plasm were detected in groups of several cells (Figure 5(b)). In these cells numerous mitochondria, clusters of ribosomes, and conspicuous clusters of contractile pro- teins were evident in the cytoplasm (Figures 6-8). Some cells contained dispersed contractile myofilaments in the cytoplasm that were still in an early stage of organization (Figure 6). The organization of newly-formed contractile myofilaments in the cytoplasm was observed in various

Figure 3. Representative desmin immunostained light micro- graphs of the interphase of the infarcted zone of laser-treated rats. Note that desmin positively stained cross-sections of myo- fibers (arrows) intermingled in the infarcted zone in (a). In (b) immunopositively stained cross-sections of myofibers (arrow) are visible in the infarcted area (IF). In (c) newly-formed car- diomyocytes (NC) are seen, with the desmin immunostaining mainly confined to the Z-line. Bar = 50 μm.

Figure 4. Density of desmin positively stained area (relative to total area) in the infarcted areas of control (non-laser-treated) and laser-treated (to the bone marrow) rats at 14 days post-MI. Results are mean+ S.E.M from 6 - 8 rats in each group. **p < 0.01.

Figure 5. Electron micrographs of typical interphase zone be- tween myocardium and infarcted area of control non-laser- treated (a) and laser-treated (b) to bone marrow rats. Note intact myocardium (MY) and adjacent fibroblast (FB) in the infarcted area surrounded by collagen (CL) deposition in (a). In (b) sev- eral newly-formed cardiomyocytes (marked with asterix) with conspicuous well-organized myofilaments (MF) in their cyto- plasm are evident adjacent to blood capillaries (CA). EN, En- dothelial cell.
degrees of maturation in those cells. In some cells the myofilaments were dispersed in the cytoplasm and in others they were organized in clusters anchored to well- developed Z-lines (Figure 7(a)). In certain cells the myo- filaments were organized parallel to the longitudinal di- rection of the cells, resembling the morphological char- acteristics of mature intact cardiomyocytes (Figure 7(b)). Some of the cells were also seen in a process of forma- tion of typical intercalated disc between them (Figure 9).
4. DISCUSSION AND CONCLUSION
The most significant outcome of this study was the ap- pearance of newly-formed cardiomyocytes following laser treatment to the BM, as indicated by light and electron microscopy. There was a 3-fold increase in the density of

Figure 6. Electron micrographs of most probably newly-formed cardiomyocytes at an early stage of organization of contractile myofilaments. Note myofilaments (MF) in the cytoplasm. M, Mitochondrion. Bar = 1 μm.

Figure 7. Electron micrographs of most probably newly-formed cardiomyocytes with early (a) and late (b) stages of the organi- zation of the contractile myofilaments in the cytoplasm. Note contractile myofilaments that are dispersed (DMF) in the cyto- plasm with a few organized in clusters anchored to Z-lines (Z) in (a). In (b) myofilaments (MF) are organized in parallel to the longitudinal axis of the cardiomyocyte, resembling their orga- nization in mature cardiomyocyte. N, Nucleus. Bar = 1 μm.
desmin immunostained cells in the infarcted rat hearts that had been laser treated. Desmin is a protein found in the cytoplasm of developing myocytes and cardiomyo- cytes [32]. The significantly higher occurrence of des- min-positive cells in the infarcted area of the laser- treated hearts may indicate the synthesis of new contrac- tile proteins in the developing new cardiomyocytes, re- sembling the process that takes place during embryonic development. The ultrastructural features of the cells in the interphase between the intact myocardium and the

Figure 8. Electron micrographs of typical interphase zone be- tween myocardium and infarcted area of laser-treated infarcted rat heart. Note numerous mitochondria (M) in the cytoplasm of the cardiomyocytes in (a) and (b). Also note organized contrac- tile myofilament with well-developed Z-lines (Z), some dis- persed myofilaments and clusters of ribosomes (R). Bar = 1 μm.

Figure 9. Electron micrographs of typical intercalated disk formation in the interphase region of the infarcted heart of la- ser-treated rats. Formation of intercalated disks (ID) between cells (marked with asterix) is evident. Note that the most proba- bly newly-formed cardiomyocytes contain clusters of myofila- ments (MF) in the cytoplasm that are conspicuous in their obli- que or cross-sections (arrows). Bar = 1 μm.
infarcted myocardium of the laser-treated rats, as shown in this study, clearly resemble the characteristics of car- diomyocytes during embryonic development of the heart [33]. Furthermore, the clusters of ribosomes and the nu- merous clusters of mitochondria in the cytoplasm of these cells may characterize cells that are active in the synthe- sis of proteins. It was previously demonstrated that direct LLLT to the infarcted hearts of rats, dogs and pigs caus- ed a significant reduction of scarring post-MI [23,24]. It was suggested that part of this reduction could be ex- plained by the regenerative response that takes place in the interphase zone [24].
The results of the present study indicate that the LLLT
applied to autologous BM attenuates the concentration of macrophages and MSC in the circulating blood. We have previously shown that LLLT application to the BM of infarcted rats caused a 2 fold enhancement in the rate of proliferation of MSC in the BM [30]. Those cells that most probably leave the BM to the circulating blood in- deed show a significant elevation of their concentration (as reveled by the FACS analysis in the present paper) at 5 days post MI. Consequently these cells probably home in on the infarcted heart, and even migrate specifically to the infarcted area [30]. These cells may induce cardiac stem cells to differentiate to newly-formed cardiomyo- cytes, as suggested previously by Hatzistergos et al. [14]. Indeed, it was found that endogenous c-kit+ cardiac stem cells were increased by 20-fold in the rat infarcted heart compared to control, following transcardial injection of BM-derived MSCs [14]. Such induction may be enabled due to paracrine secretion of various growth factors by the laser-stimulated MSC that originated from the BM. The possibility that paracrine secretion occurs in im- planted stem cells during cell therapy to the heart post- MI has been suggested previously [34]. Another mecha- nism that may take place after homing of stem cells to the infarcted heart of the laser-stimulated rats is that these cells continue to proliferate in the appropriate mi-lieu of the interphase zone in the infarcted heart and then differentiate to cardiomyocytes [30].
Another possible mechanism that maybe associates with the reduction of infarct size is the significant increase in the concentration of macrophages in the circulation fol- lowing LLLT to the BM as revealed from the FACS analysis in the present study. These findings corroborate with studies indicating that macrophages activity in the infarcted area at early stages post MI cause reduction of scarring post MI [35,36]. Thus, it could be postulated that more macrophages that will eventually home in the infarcted area from the circulating blood in the laser treated rats will also contribute to the reduction of scar- ring.
Although the findings of the present study do not in- dicate the extent of regenerative capacity of the rat in- farcted heart post-laser-irradiation, they do reveal a shift from practically no cardiomyocytes in the tissue samples taken from the non-laser-treated hearts, to the presence of newly-formed cardiomyocytes in all the electron mi- croscope sections taken from the hearts of rats that are laser-treated to the BM.
In conclusion, to the best of our knowledge, this is the first study to demonstrate the appearance of newly-form- ed cardiomyocytes in the infarcted area following LLLT to autologous BM in the infarcted rat heart. The mecha- nisms associated with this phenomenon remain to be elu- cidated in further studies.
5. ACKNOWLEDGEMENTS
This study was partially supported by the Elizabeth and Nicholas Shle- zak Super-center for Cardiac Research and Medical Engineering. The authors wish to acknowledge N. Paz for editing the manuscript and V. Wexler for helping with preparation of the figures.
REFERENCES
[1]
Rumyantsev, P.P. (1977) Interrelations of the prolifera- tion and differentiation processes during cardiac myoge- nesis and regeneration. International Review of Cytology, 51, 186-273. doi:10.1016/S0074-7696(08)60228-4
[2]
Poss, K.D., Wilson, L.G. and Keating, M.T. (2002) Heart regeneration in zebrafish. Science, 298, 2188-2190. doi:10.1126/science.1077857
[3]
Rumyantsev, P.P. (1973) Post-injury DNA synthesis, mi- tosis and ultrastructural reorganization of adult frog car- diac myocytes. An electron microscopic-autoradiographic study. Z Zellforsch Mikrosk Anat, 139, 431-50. doi:10.1007/BF00306596
[4]
Barnett, P. and van den Hoff, M.J.B. (2011) Cardiac re- generation: Different cells same goal. Medical & Biologi- cal Engineering & Computing, 49, 723-732. doi:10.1007/s11517-011-0776-5
[5]
Bollini, S., Smart. N. and Riley, P.R. (2011) Resident car- diac progenitor cells: At the heart of regeneration. Jour- nal of Molecular and Cellular Cardiology, 50, 296-303. doi:10.1016/j.yjmcc.2010.07.006
[6]
Choi, W.Y. and Poss, K.D. (2012) Cardiac regeneration. Current Topics in Developmental Biology, 100, 319-343. doi:10.1016/B978-0-12-387786-4.00010-5
[7]
Laflamme, M.A. and Murry, C.E. (2011) Heart regenera- tion. Nature, 473, 326-335. doi:10.1038/nature10147
[8]
Steinhauser, M.L. and Lee, R.T. (2011) Regeneration of the heart. EMBO Molecular Medicine, 3, 701-712. doi:10.1002/emmm.201100175
[9]
Urbanek, K., Torella, D., Sheikh, F., De Angelis, A., Nur- zynska, D., Silvestri, F., Beltrami, C.A., Bussani, R., Bel- trami, A.P., Quaini, F., Bolli, R., Leri, A., Kajstura. J. and Anversa, P. (2005) Myocardial regeneration by activation of multipotent cardiac stem cells in ischemic heart failure. Proceedings of the National Academy of Sciences of the USA, 102, 8692-8697. doi:10.1073/pnas.0500169102
[10]
Bittner, R.E., Schofer, C., Weipoltshammer, K., Ivanova, S., Streubel, B., Hauser, E., Freilinger, M., Höger, H., Elbe- Bürger, A. and Wachtler, F. (1999) Recruitment of bone- marrow-derived cells by skeletal and cardiac muscle in adult dystrophic mdx mice. Anatomy and Embryology, 199, 391-396. doi:10.1007/s004290050237
[11]
Jackson, K.A., Majka, S.M., Wand, H., Pocius, J., Hart- ley, C.J., Majesky, M.W., Entman, M.L., Michael, L.H., Hirschi, K.K. and Goodell, M.A. (2001) Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. The Journal of Clinical Investigation, 107, 1395-1402. doi:10.1172/JCI12150
[12]
Pfister, O., Mouquet, F., Jain, M., Summer, R., Helmes, M., Fine, A., Colucci, W.S. and Liao, R. (2005) CD3- but not CD31+ cardiac side population cells exhibit functio- nal cardiomyogenic differentiation. Circulation Research, 97, 52-61. doi:10.1161/01.RES.0000173297.53793.fa
[13]
Balsam, L.B., Wagers, A.J., Christensen, J.L., Kofidis, T., Weissman, I.L. and Robbins, R.C. (2004) Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature, 428, 668-673. doi:10.1038/nature02460
[14]
Hatzistergos, K.E., Quevedo, H., Oskouei. B.N., Hu, Q., Feigenbaum, G.S., Margitich, I.S., Mazhari, R., Boyle, A.J., Zambrano, J.P., Rodriguez, J.E., Dulce, R., Pattany, P.M., Valdes, D., Revilla, C., Heldman, A.W., McNiece, I. and Hare, J.M. (2010) Bone marrow mesenchymal stem cells stimulate cardiac stem cell proliferation and differ- entiation. Circulation Research, 107, 913-922. doi:10.1161/CIRCRESAHA.110.222703
[15]
Porrello, E.R., Mahmoud, A.I., Simpson, E., Hill, J.A., Richardson, J.A., Olson, E.N. and Sadek, H.A. (2011) Transient regenerative potential of the neonatal mouse heart. Science, 331, 1078-1080. doi:10.1126/science.1200708
[16]
Conlan, M.J., Rapley, J.W. and Cobb, C.M. (1996) Bio- stimulation of wound healing by low energy laser irradia- tion. Journal of Clinical Periodontology, 23, 492-496. doi:10.1111/j.1600-051X.1996.tb00580.x
[17]
Karu, T. (2007) Ten lectures on basic science of laser photherapy. Prima Books, Gragesberg.
[18]
Karu, T. (2010) Mitochondrial mechanisms of photobio- modulation in context of new data about multiple roles of ATP. Photomedicine and Laser Surgery, 28, 159-160. doi:10.1089/pho.2010.2789
[19]
Bibikova, A. and Oron, U. (1993) Promotion of muscle regeneration in the toad (Bufo viridis) gastrocnemius mu- scle by low energy laser irradiation. Anatomical Record, 235, 374-380. doi:10.1002/ar.1092350306
[20]
Bibikova, A., Belkin, A. and Oron, U. (1994) Enhancement of angiogenesis in regenerating gastrocnemius muscle of the toad (Bufo viridis) by low energy laser irradiation. Anatomy and Embryology, 190, 597-602. doi:10.1007/BF00190110
[21]
Oron, U. (2006) Photoengineering of tissue repair in ske- letal and cardiac muscles. Photomedicine and Laser Sur- gery, 24, 111-120. doi:10.1089/pho.2006.24.111
[22]
Yaakobi, T., Shoshani, Y., Levkovitz, S., Rubin, O., Ben- Haim, S.A. and Oron, U. (2001) Long term effect of low energy laser irradiation on infarction and reperfusion in- jury in the rat heart. Journal of Applied Physiology, 90, 2411-2441.
[23]
Oron, U., Yaakobi, T., Oron, A., Hayam, G., Gepstein, L., Wolf, T., Rubin, O. and Ben Haim, S.A. (2001a) Attenu- ation of the formation of scar tissue in rats and dogs post myocardial infarction by low energy laser irradiation. La- sers in Surgery and Medicine, 28, 204-211. doi:10.1002/lsm.1039
[24]
Oron, U., Yaakobi, T., Oron, A., Mordechovitz, D., Shof- ti, R., Hayam, G., Dror, U., Gepstein, L., Wolf, T., Hau- denschild, C. and Ben Haim, S.A. (2001b) Low energy la- ser irradiation reduces formation of scar tissue following myocardial infarction in dogs. Circulation, 103, 296-301.doi:10.1161/01.CIR.103.2.296
[25]
Tuby, H., Maltz, L. and Oron, U. (2006) Modulations of VEGF and iNOS in the rat heart by low energy laser irra- diation are associated with cardioprotection and enhanced angiogenesis. Lasers in Surgery and Medicine, 38, 682- 688. doi:10.1002/lsm.20377
[26]
Oron, U., Ilic, S., De Taboada, L. and Streeter, J. (2007) Ga-As (808 nm) laser irradiation enhance ATP produc- tion in human neuronal cells in culture. Photomedicine and Laser Surgery, 25, 180-182. doi:10.1089/pho.2007.2064
[27]
Tuby, H., Maltz, L. and Oron, U. (2007) Low-level laser irradiation (LLLI) promotes proliferation of mesenchy- mal and cardiac stem cells in culture. Lasers in Surgery and Medicine, 39, 373-378. doi:10.1002/lsm.20492
[28]
Li, W.T., Leu, Y.C. and Wu, J.L. (2010) Red-light light- emitting diode irradiation increases the proliferation and osteogenic differentiation of rat bone marrow mesenchy- mal stem cells. Photomedicine and Laser Surgery, 28, S-157-S-165. doi:10.1089/pho.2009.2540
[29]
Mvula, B., Moore, T.J. and Abrahamse, H. (2010) Effect of low-level laser irradiation and epidermal growth factor on adult human adipose-derived stem cells. Lasers in Medical Science, 25, 33-39. doi:10.1007/s10103-008-0636-1
[30]
Tuby, H., Maltz, L. and Oron, U. (2011) Induction of au- tologous mesenchymal stem cells in the bone marrow by low-level laser therapy has profound beneficial effects on the infarcted rat heart. Lasers in Surgery and Medicine, 43, 401-409. doi:10.1002/lsm.21063
[31]
Oron, U. (2011) Light therapy and stem cells: A thera- peutic intervention of the future. Journal of Interventio- nal Cardiology, 3, 627-629.
[32]
Toma, C., Pittenger, M.F., Cahill, K.S., Byrne, B.J. and Kessler, P.D. (2002) Human mesenchymal stem cells dif- ferentiate to a cardiomyocyte phenotype in the adult mu- rine heart. Circulation, 105, 93-98. doi:10.1161/hc0102.101442
[33]
Oron, U. and Mandelberg, M. (1985) Focal regeneration in the rat myocardium following cold injury. Cell Tissue Research, 241, 459-463. doi:10.1007/BF00217194
[34]
Mummery, C.L., Davis, R.P. and Krieger, J.E. (2010) Challenges in using stem cells for cardiac repair. Science Translational Medicine, 14, 1-5.
[35]
van Amerongen, M.J., Harmsen, M.C., van Rooijen, N., Petersen, A.H. and van Luyn, M.J. (2007) Macrophage dep- letion impairs wound healing and increases left ventricu- lar remodeling after myocardial injury in mice. American Journal of Pathology, 170, 1093-1103. doi:10.2353/ajpath.2007.060547
[36]
Okazaki, T., Ebihara, S., Asada, M., Yamanda, S., Saijo, Y., Shiraishi, Y., Ebihara, T., Niu, K., Mei, H., Arai, H. and Yambe, T. (2007) Macrophage colony-stimulating factor improves cardiac function after ischemic injury by induc- ing vascular endothelial growth factor production and sur- vival of cardiomyocytes. American Journal of Pathology, 171, 1093-1103. doi:10.2353/ajpath.2007.061191
Original Source: http://www.scirp.org/journal/jbise
Influence of Low Level Laser Radiation on Migration of Stem Cells
Levon Gasparyan, Grigory Brill, Anu Makela - (Publication) 4468This study showed a 26% increase in stem cell when they uses red and IR lasers continuous wave.
Abstract: The long term effects of low level laser therapy can involve treatment mechanisms connected with activation of stem cells.
In the current study migration of stem cells was tested under the influence of laser light alone as well as in case of combined influence of light and stromal cell-derived factor-1α (SDF-1α). This cytokine plays a role in lymphocyte trafficking, hematopoietic progenitor cell and stem cell homing.
To investigate the light influence on stem cells, we analyzed factor-dependent cell-Patersen (FDCP)-mix multipotent progenitor cells.
Migration of the stem cell line was tested using Transwell system (Corning, NY) under influence of red diode laser (λ=659.6 nm, 19.5 mW) or infrared diode laser (λ=958 nm, 36 mW) during 15 min at continuous wave, as well as in case of applying 150 ng/ml SDF-1α.
Group 1 cells were a group of control, group 2 cells received only red light irradiation, while group 3 cells had IR light irradiation. Group 4 cells were treated with 150 ng/ml SDF-1α. Group 5 cells were irradiated with red laser light in addition to 150 ng/ml SDF-1α, and group 6 cells by IR light and 150 ng/ml SDF-1α.
The count of migrated cells was 1496,5±409 (100%) in case of control. Red and IR laser light increased migration activity of stem cells up to 1892±283 (126%) and 2255,5±510 (151%) accordingly. Influence of SDF-1α was more significant, than effects of light irradiation alone 3365,5±489 (225%). Combined effects of light irradiation and SDF-1α were significantly stronger 5813±1199 (388%) for SDF-1α and red laser light, and 6391,5±540 (427%) for SDF-1α and IR laser light irradiation.
Preliminary study results showed that laser light irradiation can activate stem cell migration in vitro. The results are more reliable in the case of combined application of light and SDF-1α. These results are giving ground to consider that stem cell reactions to light irradiation can be one of the factors of light therapy.
Key words: low level laser irradiation, low level laser therapy, stem cells, SDF-1, stromal cell-derived factor-1
INTRODUCTION
More than 30 years ago first reports about biological effects of low doses of laser light were presented. Currently low level laser therapy (LLLT) is successfully applied in the treatment of numerous diseases and pathological conditions. LLLT exhibits positive effects for the treatment of disorders, having in common failure of blood supply with development of acute or chronic tissue hypoxia, different level of destruction of tissues, following decreased regenerative abilities of tissues and organs, defects in immune system, and altered cell metabolism. At the same time some important mechanisms of influence of laser light on the body are still far to be fully understood [1 - 8].
Recent studies discovered important role of bone marrow hematopoetic stem cell (HSCs) for naturally occurred recovery and regeneration processes, following tissue hypoxia and injury. The three clinically important steps in this natural process are mobilization of stem cells from the bone marrow, homing of these cells to the site of injury, and differentiation of the stem cell into a functional cell of the injured tissue [9]. Different methods of stem cell therapy, the treatment method, based on mobilization and transplantation of stem cells, proves to be effective method of therapy for different disorders.
We proposed a hypothesis that wide range of positive effects following laser therapy can be connected to increased activity of stem cells in damaged tissues. To test that, we examined in vitro the influence of laser light on migration of stem cells in absence and in presence of stromal cell-derived factor-1 (SDF-1), a potent chemoattractor for lymphocytes, monocytes, HSCs, which plays a critical role in the stem cell migration towards areas of tissue injury and hypoxia.
MATERIALS AND METHODS
To investigate the light influence on stem cells, we analyzed factor-dependent cell-Patersen (FDCP)-mix multipotent progenitor cells. The FDCP-mix stem cell line was maintained in ISCOVE’S medium supplemented with 20% horse serum and penicillin/streptomycin in the presence of 20 ng/ml IL-3. The cells were supplied with fresh medium each 5 days. Migration of the stem cell line was tested using Transwell system (Corning, NY). The cells were washed with PBS once and re-suspended in the medium containing 0.1% BSA (2x106/ml). Then, 600 μl of the mixture was irradiated by red diode laser (λ=659.6 nm, 19.5 mW) or infrared diode laser (λ=958 nm, 36 mW) during 15 min at continuous wave. Next, 100 μl of the mixture (2x105 cells) was seeded into upper chambers of the Transwell system, and the filters were placed into the wells containing 600? μl of the medium with or without 150 ng/ml SDF-1α. The plate was incubated for 4 h (37°C, 5% CO2, humidified atmosphere), after which the cells were collected and counted by a FACS sorter (Beckton Dickinson) during 1 min. All samples were performed in duplicate.
Group 1 cells are control group, group 2 cells received only red light irradiation, while group 3 cells – only IR light irradiation. Group 4 cells were treated with 150 ng/ml SDF-1α. Group 5 cells were irradiated with red laser light in addition to 150 ng/ml SDF-1α, and group 6 cells – IR light and 150 ng/ml SDF-1α.
RESULTS
Small amount of stem cells can migrate without SDF-1α or laser light influence. The count of migrated cells in control group was 1496,5±409 (Fig). This amount was considered as 100%. Red and IR laser light at the above mentioned dosage and methods of irradiation increased migration activity of stem cells up to 1892±283 (126%) and 2255,5±510 (151%) accordingly. Influence of SDF-1α was more noticeable, than effects of red or IR laser light irradiation alone - 3365,5±489 (225%). It is important to stress attention on the finding, that rate of stem cell migration towards the filter and SDF-1α containing medium was much higher after laser irradiation of cells - 5813±1199 (388%) for red laser light, and 6391,5±540 (427%) for IR laser light irradiation.
DISCUSSION
The main scientific result of this study is the fact, that red and infrared laser light irradiation can activate migration of stem cells in vitro. Moreover, red and IR laser radiation can up-regulate the rate of stem cell migration towards higher SDF-1α gradient.
How to explain the direct effects of mobility of stem cells in vitro under red and IR laser light irradiation, and use this fact for better understanding the wide range of therapeutic effects of laser therapy?
Modern medical science has accepted that every pathologic condition or disease should be treated according to its clinical stage and symptoms, considering its pathogenesis and etiology. Similar treatment methods can be applied only for the treatment of different diseases, having common pathogenesis.
Not very many examples of successful application of the similar or close therapy method for the treatment of different pathologies are known in modern medicine. Steroid hormone therapy is one of such cases.
Another illustration of successful application of the similar treatment techniques for treatment of different disorders is stem cell therapy, a novel treatment method, which is still under development. Growing data suggests, that transplanted stem cell can successfully and for long period of time improve heart myocardial contractility and other heart functions after myocardial infarction, can support neoangiogenesis in areas of tissue infarction and damage, can replace several cell types in tissues, including β-cells in diabetes models, neurons, cardiomyocytes, hematopoetic cells of different lineages and so on, as well as be useful in the treatment of atherosclerosis [9].
The main principle of stem cell therapy is the idea of replacement of damaged and dead cells in injured tissues and organs with new healthy ones. It is known, that severe stress, tissue hypoxia and damage mobilizes some hematopoetic stem cells (HSCs) from bone marrow to peripheral bloodstream. After that HSCs can migrate towards hypoxic tissues and reach them. Finally they can start to proliferate to the cells types, typical for that damaged tissues. HSCs in the tissues are also able to produce several cytokines, chemokines, growthfactors, improve survival of damaged cells and limit apoptosis. As a result of some tissue regeneration, improvement in the function of a damaged organ can be achieved. Similar and even stronger regeneration and treatment effects can be displayed after transplantation of fetal or adult HSCs to recipient [10-12].
Low laser light irradiation is one other example of application of the same factor for the treatment of number of disorders, which, at first glance, have nothing or very little in common in their pathogenesis. Laser light can accelerate wound and burn healing, improve condition of patients after myocardial infarction and stroke, can support hematopoiesis of bone marrow after X-ray radiation or during cancer chemotherapy, can help for the treatment of diabetic angiopathy and neuropathy, as well as reduce atherosclerotic plaque formation. In cellular and tissue level LLLT exhibits positive effects for the treatment of disorders, having in common failure of blood supply with development of acute or chronic tissue hypoxia, different level of destruction of tissues, following with decreased regenerative abilities of cells, as well as altered cell metabolism [6, 7, 13, 14].
One can see that the therapeutic applications of LLLT and stem cell therapy are very close. So, earlier we proposed the hypotheses that one of the mechanisms of light therapy includes acceleration of tissue repair due to better mobilization of stem cells to the spot of injury after laser light irradiation [15]. That process should include several phases, including activation of stem cell migration towards area of tissue damage and hypoxia.
Stem cells are being investigated for their potential use in regenerative medicine. Stem cells share the following two defining characteristics: the capacity to differentiate into a spectrum of different cell types and the capacity to renew themselves [16]. The biological principle that underlies stem cell therapy is tissue-directed differentiation. For example, adult stem cells isolated from liver tissue and re-injected into liver become hepatocytes, whereas the same cells injected into myocardium become myocytes. [17] Stem cells have been engrafted into a broad spectrum of tissues, including regenerating bone, neural tissue, dystrophic skeletal muscle, and injured skeletal muscle. [18]. Myocardial regeneration is perhaps the most widely studied and debated example of stem cell plasticity. The most promising results have been obtained after transplantation and mobilization of bone marrow cells to the area of infarction.
The three clinically important steps in this natural process are mobilization of stem cells from the bone marrow, homing of these cells to the site of injury, and differentiation of the stem cell into a functional cell of the injured tissue [19].
Stem cell repair of cardiac and vascular tissue is a naturally occurring process after injury [20, 21] Circulating CD34+ mononuclear cell counts and plasma levels of endothelial growth factor are significantly increased in patients with acute myocardial infarction, peaking on day 7 after onset [22]. Due to limitations of the naturally occurring repair process after myocardium infarction and other injuries or pathologies several stem cell transplantation strategies were proposed and tested.
At present, however, enthusiasm for the therapeutic potential of strategies of stem cell transplantation is limited by certain practical considerations. For example, the number of stem cells, required for injection for the treatment of myocardial infarction, can be harvested approximately from 6 l of donor blood [23].
Other important limitation for autologous bone marrow stem/progenitor cell mobilization is a recent finding, that circulating endothelial progenitor cells in patients with coronary heart disease are impaired with respect to number and functional activity. Moreover, Heeschen et al [24] reported that regeneration and functional ability of bone marrow-derived mononuclear cells (BM-MNCs) in patients with chronic ischemic cardiomyopathy (ICMP) are also limited. In spite of the fact that, the number of BM-MNCs isolated from bone marrow aspirates of 18 patients with ICMP and 8 healthy subjects s did not differ, the colony-forming capacity of BM-MNCs from patients with ICMP was significantly lower compared with BM-MNCs from healthy controls. Likewise, the migratory response to SDF-1 and vascular endothelial growth factor (VEGF) was significantly reduced in BM-MNCs derived from patients with ICMP compared with BM-MNCs from healthy controls. The reduced neovascularization capacity in vivo of BM-MNCs derived from patients with ICMP closely correlated with the in vitro assessment of SDF-1-induced migration and colony-forming capacity.
The need for development of new methods for mobilization, as well as for homing of stem cells to the site of injury is therefore evident.
Several growth factors, chemokines and cytokines are involved in the regulation of stem cell mobilization, homing and differentiation. Stromal cell-derived factor-1 (SDF-1) is one of them. SDF-1 is a chemokine playing an important role in the trafficking of hematopoietic stem cells. SDF-1 is expressed on stromal cells of various tissues. CXCR4 is the only known receptor for SDF-1 [25]. SDF-1/CXCR4 interaction is reported to play an important physiological role during embryogenesis in hematopoiesis, vascular development, cardiogenesis, and cerebellar development [26-28].
Recently, several investigators have reported that CD34+ cells, classically considered to be hematopoietic stem cells, expressed CXCR4, and that SDF-1 could induce CD34+ cell migration in vitro [29]. Accordingly, SDF-1 is considered as one of the key regulators of hematopoietic stem cell trafficking between the peripheral circulation and bone marrow. SDF-1 has also been shown to effect CD34+ cell proliferation and mobilization and to induce angiogenesis in vivo [30 -32].
Hattori et al [31] reported that plasma elevation of SDF-1 induced mobilization of mature and immature hematopoietic progenitors and stem cells, including endothelial progenitor cells (EPCs). However, application of granulocyte colony-stimulating factor (G-CSF) for stem cell mobilization is widely accepted nowadays.
Yamaguchi et al [23] studied the effects of SDF-1 on migration and accumulation of EPCs. SDF-1 induced EPCs migration in a dose dependent manner in vitro. The magnitude of migration was similar to that induced by VEGF. Authors also reported that locally (in hind-limb ischemic muscle of experimental animals) administered SDF-1 could augment the local accumulation of transplanted EPCs from peripheral blood, thereby resulting in enhanced neovascularization. As a result, cell transplantation not only improved neovascularization but also reduced adverse biological consequences such as limb necrosis and auto-amputation in the mouse ischemic hind-limb model. These studies also disclosed that systemic EPCs transplantation improved myocardial neovascularization and cardiac function corresponding to reduced left ventricular scarring. Authors concluded that, at least under the experimental conditions used in the study, the effect of SDF-1 on neovascularization appears to result primarily from its ability to enhance the recruitment and incorporation of transplanted EPCs.
Damas at al. [33] reported that SDF-1α, at least in high concentrations, may mediate anti-inflammatory and matrix-stabilizing effects in unstable angina. These effects may promote plaque stabilization, and therapeutic intervention that enhances SDF-1 α activity could potentially be beneficial in acute coronary syndromes. Authors demonstrated significantly altered SDF-1/CXCR4 expression in patients with angina, with particularly marked changes in those with unstable disease, with low SDF-1 levels in plasma and altered expression of its corresponding receptor on peripheral blood mononuclear cells (PBMC). In contrast to the raised plasma levels of inflammatory chemokines in patients with angina plasma levels of SDF-1 and the surface expression of its corresponding receptor (CXCR4) on PBMC appear to be down-regulated in these patients. Thus, although persistent inflammation may involve up-regulation of inflammatory chemokines, recent studies suggest that inflammatory cytokines (eg, TNF-α and IL-1) may decrease the expression of SDF-1 and CXCR4.
Future progress of stem therapy techniques probably will include development of incubation methods for enhancement stem cell mobility and homing ability, as well as for faster proliferation into desire tissue cells. Increasing migration abilities will help to achieve better and faster results.
The ability of laser light to activate migration and mobility of different cells is well known. It was noticed, that irradiation of sperm cells in vitro can increase their mobility and fertility [34]. Moreover, this effect is more pronounced in case of damaged cells with low mobility rate. This gives a ground to assume that laser light irradiation in certain dosage and condition can improve functional abilities of cells. Future experiments are required to ascertain if stem cells respond to the laser light the same way.
The main finding on this study is that red and IR laser light can stimulate stem cell migration in vitro, and especially increase migration towards SDF-1α gradient. Stem cell ability to migrate towards tissues with higher SDF-1 concentration is one of the key mechanisms of stem cell homing. These results are giving ground to speculate that activation of stem cell migration can be one of the mechanisms of low level laser therapy. Taking into consideration that the combined of SDF-1 and laser irradiation had the strongest effect on stem cell homing, it would be reasonable to assume that this combination could be used in not only increasing the activity of stem cells but also in determining the main area of stem cell mobilization and homing. The current study did not aim to study the mechanisms of increased migration ability, which will be study in the future. But it is possible to suggest following explanation: laser irradiation can change the metabolism of stem cells, increase ATP production and so increase the migration, as well as up-regulate CXCR4 receptor expression or syntheses de novo. More studies are required to test if the laser light irradiation in vivo is able to make homing of transplanted stem cells to the area of damage more efficient, to check the influence of laser light on the mobilization rate of stem cells from bone marrow, to investigate if laser light can enhance functional abilities of stem cells. These studies would be desirable for better understanding of the mechanisms of laser therapy and for development of more effective methods of stem cell therapy.
References
1. Tuner J. and Hode L. Low Level Laser Therapy: Clinical Practice and Scientific Background, Prima Books, Grängesberg, Sweden, 1999.
2. Karu T. The Science of Low Power Laser Therapy, Gordon & Breach, London, 1998.
3. Baxter G.D. Therapeutic Lasers: Theory and Practice, Churchill Livingstone, London, 1994.
4. Simunovic Z., Ed. Lasers in Medicine and Dentistry, Vitgraf, Rijeka (Croatia), 2000.
5. Zhukov B.N. and Lysov N.A. Laser irradiation in experimental and clinical angiology (in Russian), Samara (Russia), 1996.
6. Kozlov V.I., et al. Bases of laser physio- and reflexo-therapy (in Russian), Zdorovje, Samara (Russia), 1993.
7. Paleev N.R. Ed. Phototherapy (in Russian), Meditsina, Moscow (Russia), 2001.
8. Skobelkin O. K. Ed. Application of low-intensive lasers in clinical practice (in Russian). Moscow, 1997.
9. Forrester J, Price M, Makkar R. Stem Cell Repair of Infarcted Myocardium. An Overview for Clinicians. Circulation. 2003;108:1139–1145.
10. Orlic D., Hill J., Arai A. Stem Cells for Myocardial Regeneration Circulation Research. 2002;91:1092.
11. Hodgson D., Behfar A., Zingman L.V., Kane G.C., Perez-Terzic C., Alekseev A.E., Puceat M., and Terzic A. Stable benefit of embryonic stem cell therapy in myocardial infarction. Am J Physiol Heart Circ Physiol, August 1, 2004; 287(2): H471 - H479.
12. Ozbaran M., Omay S. B., Nalbantgil S., Kultursay H., Kumanlioglu K., Nart D., and Pektok E. Autologous peripheral stem cell transplantation in patients with congestive heart failure due to ischemic heart disease. Eur. J. Cardiothorac. Surg., March 1, 2004; 25(3): 342 - 350.
13. Brill A.G., Shenkman B., Brill G.E. et al. Blood irradiation by He-Ne laser induces a decrease in platelet responses to physiological agonists and an increase in platelet cyclic GMP. Platelets. 2000. Vol. 11. P. 87-93.
14. Mester A. Biostimulative effect in wound healing by continuous wave 820 nm laser diode. Lasers in Med Science, abstract issue July 1988, No. 289.
15. Gasparyan L.V. Stem cells and therapeutic effect of light irradiation (in Russian). Collection of abstracts of the 10th International Conference of Quantum Medicine, Moscow, 2003, pp. 43-44.
16. Graf T. Differentiation plasticity of hematopoietic cells. Blood. 2002;99:3089–3101.
17. Malouf NN, Coleman WB, Girsham JW, et al. Adult-derived stem cells from the liver become myocytes in the heart in vivo. Am J Pathol. 2001;158:1929–1935.
18. Donovan PJ, Gearhart J. The end of the beginning for pluripotent stem cells. Nature. 2001;414:92–97.
19. Forrester J, Price M, Makkar R. Stem Cell Repair of Infarcted Myocardium. An Overview for Clinicians. Circulation. 2003;108:1139–1145.
20. Beltrami AP, Urbanek K, Kajstura J, et al. Evidence that human cardiac myocytes divide after myocardial infarction. N Engl J Med. 2001;344:1750–1757.
21. Gill M, Dias S, Hattori K, et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res. 2001;88:167–174.
22. Shintani S, Murohara T, Ikeda H, et al. Mobilization of endothelial progenitor cells in patients with acute myocardial infarction. Circulation. 2001;103:2776–2779.
23. Yamaguchi J, Kusano K, Masuo O, at al. Stromal Cell–Derived Factor-1 Effects on Ex Vivo Expanded Endothelial Progenitor Cell Recruitment for Ischemic Neovascularization. Circulation. 2003;107: 1322–1328.
24. Heeschen C, Lehmann R, Honold J, Assmus B, Aicher A, Walter DH, Martin H, Zeiher AM, Dimmeler S. Profoundly reduced neovascularization capacity of bone marrow mononuclear cells derived from patients with chronic ischemic heart disease. Circulation. 2004;109(13):1615-22.
25. Bleul CC, Farzan M, Choe H, et al. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/fusin and blocks HIV-1 entry. Nature. 1996;382:829–833.
26. Nagasawa T, Hirota S, Tachibana K, et al. Defects of B-cell lymphopoiesis and bone-marrow myelopoiesis in mice lacking the CXC chemokine PBSF/SDF-1. Nature. 1996;382:635–638.
27. Tachibana K, Hirota S, Iizasa H, et al. The chemokine receptor CXCR4 is essential for vascularization of the gastrointestinal tract. Nature. 1998; 393:591–594.
28. Zou YR, Kottmann AH, Kuroda M, et al. Function of the chemokine receptor CXCR4 in haematopoiesis and in cerebellar development. Nature. 1998;393:595–599.
29. Mohle R, Bautz F, Rafii S, et al. The chemokine receptor CXCR-4 is expressed on CD34+ hematopoietic progenitors and leukemic cells and mediates transendothelial migration induced by stromal cell-derived factor-1. Blood. 1998;91:4523–4530.
30. Lataillade JJ, Clay D, Dupuy C, et al. Chemokine SDF-1 enhances circulating CD34+ cell proliferation in synergy with cytokines: possible role in progenitor survival. Blood. 2000;95:756–768.
31. Hattori K, Heissig B, Tashiro K, et al. Plasma elevation of stromal cell-derived factor-1 induces mobilization of mature and immature hematopoietic progenitor and stem cells. Blood. 2001;97:3354–3360.
32. Salcedo R, Wasserman K, Young HA, et al. Vascular endothelial growth factor and basic fibroblast growth factor induce expression of CXCR4 on human endothelial cells: in vivo neovascularization induced by stromal derived factor-1α. Am J Pathol. 1999;154:1125–1135.
33. Damas J, Wæhre T, Yndestad A, et al. Stromal Cell–Derived Factor-1a in Unstable Angina. Circulation. 2002;106:36-42.
34. Pyrikova S.I et al. Effect of laser exposure on human seminal fluid (in Russian). Clinical and laboratory diagnosis. 1998;5:15-16.
Laser-induced regeneration of cartilage
Emil N. Sobol, Olga I. Baum, Anatoliy B. Shekhter, Anna Guller, Andrey V. Baskov - J. of Biomedical Optics, 16(8), 080902 (2011) (Publication) 4501This article shows results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine
Abstract
Laser radiation provides a means to control the fields of temperature and thermo mechanical stress, mass transfer, and modification of fine structure of the cartilage matrix. The aim of this outlook paper is to review physical and biological aspects of laser-induced regeneration of cartilage and to discuss the possibilities and prospects of its clinical applications. The problems and the pathways of tissue regeneration, the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches for cartilage repair; consider possible mechanisms of laser-induced regeneration. Finally, we present the results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine.
1.
Introduction
Cartilage is a kind of highly specialized connective tissue. The structural variety of the cartilage provides its unique biomechanical capacity to bear different kinds of static and dynamic loads over a wide range of intensity. Biological role of cartilage structures stems from their critical significance for growth and development as well as for all kinds of body movements. The exceptional importance of cartilage elements for individual survival is, probably, due to mechanisms of natural selection, resulting in limited reparative potential of this tissue. Scanty cellular sources and low metabolic rate along with avascularity of cartilage contribute to its decreased regeneration ability. As a result of these strong limitations, the injuries of cartilage caused by inflammation, traumas, degeneration, and aging usually become chronic and recalcitrant to any kind of medical treatment. In the USA, according to tentative estimations, the prevalence of all forms of arthritis has been calculated in order of 40 million people; and the annual medical care costs were about 65 billion USD.1Degenerative spine diseases are a major cause of back pain that deteriorates the quality of life of patients and often leads to disability. Direct and indirect medical expenses are estimated as more than 90 billion per year.2
High prevalence and incidence, as well as the social and economic significance of cartilage pathology, attract great interest to this problem. Considerable efforts have been devoted to study various approaches to restore cartilage structures and to stimulate intrinsic capabilities of the tissue to regeneration. There are several treatment modalities of cartilage restoration suggested for clinical use (see Ref. 3 and referred literature): 1. surgical techniques; 2. controllable cell delivery to the lesion; and 3. tissue engineering applications of biodegradable materials (scaffolds) with cell-seeding and modification of cartilage reparative response by different growth factors and cytokines. Although there is a wealth of information regarding the substitution of lost cartilage by the mentioned approaches, the problem of cartilage repair is still unsolved. The long term results show no completed cartilage regeneration; in many cases, the new growing tissue materially differs from the well organized original cartilage. The reasons of insufficient cartilage reparation are connected with its structural and functional organization and with the difficulties of the precise control of the external physical and chemical effects.4, 5 Regeneration of cartilage may be realized in accordance with the natural genetic program of the cells. The efficacy of any approach aimed to control the regeneration process depends on the solution of three tasks: 1. the ability to reproduce the normal cell differentiation sequence from the progenitor cells to mature chondrocytes, 2. stimulation of the specific subpopulations of the resident cells to proliferation and/or new matrix production, and (c) achievement of adequate spatial organization of the new growing tissue. Probably, the most important feature of the laser-based treatment is the involvement and activation of the intrinsic mechanisms of cartilage repair. Many papers are devoted to the effect of low-intensive lasers on cartilage functional state and reparative ability. However, the effectiveness, as well as the placebo-versus-treatment ratio for low level laser therapy, is still under considerable dispute. A more detailed discussion of this issue may be found elsewhere.6 This paper is mainly limited with a consideration of the effect of nonablative laser radiation on the cartilaginous cells through their matrix microenvironment to provide natural and optimal conditions for regeneration. Wide ranges of wavelengths, precise localization of the irradiated area, and temporal and spatial modulation of laser radiation are the main advantages of the laser technologies, which may result in specific tissue response. In particular, the laser-induced modification of the cartilage extracellular matrix (ECM) seems to be of great significance in view of some new data on the developmental roles of the matrix molecules and mechanical loads. Although the evidence of laser irradiation morphogenetic effects is still largely circumstantial, we consider the available observations to address some possible perspectives of the controlled regeneration of cartilage using nonablative laser treatment. So, the aim of this paper is to review physical and biological aspects of laser-induced regeneration of cartilage, to discuss the possibilities and prospects of its clinical applications. The problems and the ways of tissue regeneration and the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches to cartilage repair, consider possible mechanisms of laser-induced regeneration, present the results in laser regeneration of joints and spine disks cartilages, and finally, discuss some future medical applications of laser regeneration.
2.
Cartilage as a Subject of Regeneration
There are a number of detailed reviews describing the structure and vital functions of cartilages.3, 7, 8 The main components of cartilage are cells (chondrocytes) and ECM consisting of water (70 to 80%), collagens, proteoglycans (PGs), hyaluronic acid (HA), and glycoproteins (GP). The PGs consist of glycosaminoglycanes (chondroitin sulphate and keratan sulphate) linked to the core-protein, which, in turn, is bound with HA threads interweaving between collagen fibrils (Fig. 1). PGs have a lot of negative charged groups; and the electrical neutrality of cartilage is due to the presence of positive ions (K+, Na+, H+, Ca2+, Mg2+). There are three types of cartilage tissue: hyaline cartilage (costal, nasal septum, articular cartilage of the joints), fibrous cartilage (annulus fibrosis of the spine disks, Eustachian tube), and elastic cartilage (auricle, epiglottis). Hyaline cartilage first forms in embryos and later transforms into other types of cartilage and bone tissues. The distinguishing features of the ECM of hyaline cartilage are having a very high content of glycosaminoglycanes and the prevalence of collagen type II fibrils.9, 10, 11, 12, 13 Fibrous cartilage is characterized by predominance of collagen type I.14, 15, 16 Matrix of the elastic cartilage possesses elastic fibers. Nasal and some other cartilages are covered with a perichondrium playing an important role in nutrition and growth of the avascular tissue. Articular cartilage has no perichondrium; it gets nutrition from synovial liquid and subchondral bone. An articular cartilage surface is covered by a cell-free lamina splendens (LS) consisting mainly of the HA and phospholipids.17 An important structural and metabolic unit of articular cartilage is a chondron.11 It includes a chondrocyte and its pericellular matrix (PM) bordered with a pericellular capsule (PC). The chondron is surrounded by territorial and interterritorial matrices. The chondrons and their matrix environment have different mechanical properties.3, 11 The PM is enriched with HA, sulphated PGs, biglycan, and GPs, including link protein and laminin. The PC is predominantly composed of compact thin fibrils of collagen type VI and fibronectin. It is suggested that the PM and PC provide hydrodynamic protection for the chondrocyte against pressure loading and take a part in control of spatial and temporal distribution of newly synthesized macromolecules as well as in the cell-matrix interaction.11 Territorial and interterritorial matrices are characterized by different degrees of the PGs maturity and with a different proportion of the chondroitin sulphate and keratan sulphate. The heteropolymeric fibrils of collagen types II, IX, and XI (HCF) emerging in the territorial matrice become the major load-bearing element in the interterritorial matrice.16 These fibrils are in charge of the tissue protection against multidirectional tensions.
Fig. 1
Cartilage components and structure. PG –proteolycanes; HA – hyaluronic acid, GP – glycoproteins; M – morphogenes; R -molecular receptors of chondrocyte's membrane; (+) ions (K+, Na+, H+, Ca2+, Mg2+).
A number of molecules that possess signal roles in morphogenetic processes, including chondrogenesis from embryonic development to regeneration, may interact with the receptors of the cellular membrane of chondrocyte. Binding of such morphogenes to the membrane receptors triggers various intracellular signaling cascades to result in regulation of the expression of genes. Hydrostatic pressures and fluid flows as well as multidirectional tensions contribute to tissue water displacement leading to changes of local concentrations of ions and morphogens. The GP molecules (integrins, fibronectin, laminin, etc.) distributing over the ECM serve as important mediators of the signaling molecules. They play an important role in the cell-matrix interactions and operate on the growth of cartilage tissue.
Hyaline cartilage has a zonal structure:11, 18, 19 the superficial layer contains fibroblast-like chondrocytes of type I. It is characterized by a decreased level of the PG aggregates (aggrecanes) and by a high content of small leucine-rich PGs (decorin and biglycan). The cells in the middle layer are chondrocytes of type II. They form multicellular clones and keep a certain ability of proliferation. A smaller subpopulation of the middle layer cells is presented by the chondrocytes type III covered with lacunas. These nonproliferating cells are also presented in the deep layer of cartilage. Type IV cells belong to a degrading cell group. Chondrocytes synthesize and degrade all components of cartilage matrix through specialized enzymes (prolyl hydroxylase, lysil oxidase, collagenases, aggrecanases etc.).16, 20 Metabolic activity of the chondrocytes in cartilage is controlled by hormones, various cytokines, growth factors, and vitamins (A, C, and D).21, 22, 23, 24 Ultimately, the biosynthetic and catabolic activities of cartilage cells, as well as the kinetics of the cellular population are governed by the local concentrations of the humoral and insoluble morphogens near the external membranes of chondrocytes.
The main mechanism of cartilage nutrition is diffusion of water carrying low-molecular substances (ions, glucose, amino acids, etc.). As the chondrocytes kinetics are under conditions of hypoxia, their metabolism is generally realized by the anaerobic glycolysis pathway. That, in combination with the chondrocytes paucity, determines a low level of cartilage metabolism. Half life period is three or four years for aggrecans, and about 10 years for collagen.25 All types of cartilage, especially articular cartage and intervertebral disks, have low repair potential. There is a lot of literature on this topic. 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37 Extra-articular cartilage is usually repaired by the means of proliferation and chondrogenic differentiation of the perichondrial cells. The defects of hyaline cartilage and the extensive defects of costal and auricular cartilages are usually filled up with fibrous connective tissue or fibrous cartilage, which both do not have adequate functional properties; that determines persistent attempts to find new possibilities for cartilage regeneration. The healing of cartilage defects can be improved with mechanical stimulation, intra-articular application of HA, hormone therapy,38, 39, 40, 41 and also with the use of osteochondral or cartilaginous implants, in particular together with cultivated chondrocytes.42, 43 One of the current leading approaches is in vitro growth of the tissue engineering constructs followed with their implantation into cartilage lesion. Autologous chondrocyte implantation (ACI) resulted in the formation of hyaline-like tissue with a quite stable clinical outcome.44, 45 But according to the histological data, only 39% of the defects treated with ACI were filled with hyaline cartilage, while 43% were filled with fibrocartilage, and 18% did not show any healing response at all.46
Regeneration process is associated with embryonic chondrogenesis mechanisms and partial dedifferentiation of mature cells. Figure 2 shows possible pathways of regeneration-related dedifferentiation of the cells in cartilage. Mesenchymal stem cells (MSC) can differentiate into cartilage cells of various types, including immature and mature chondrocytes, and notochordal and chondrocyte-like cells of the intervertebral disks. These processes are under multilevel control of signaling molecules and mechanical factors. Our main hypothesis is that differentiation and dedifferentiation of cartilage cells, as well as their metabolic activity, may be controlled by direct action of laser radiation on the cells and through laser-induced modification of the ECM.
Fig. 2
Differentiation of cartilage cells and possible pathways of their regeneration-related dedifferentiation. 1 – MSC, 2 – pre-chondrocytes, 3 – early chondrocytes (chondroblasts), 4 – columnar chondrocytes, 5 – hypertrophic chondrocytes, 6 – chondrocytes of fibrous cartilage, 7 – chondrocytes of hyaline cartilage, 8 – differentiation pathways, 9 – pathways of limited dedifferentiation, 10 – additional pathways of cellular differentiation (following the enchondral osteogenesis differentiation mechanism).
3.
Targets for Laser Effect. Possible Types of Cartilage Response on Laser Radiation
To discuss possible ways of using lasers for cartilage regeneration, it is important to know what effect laser parameters have on (a) different types of the cells; (b) different components of the ECM; (c) signaling molecules produced by the cells and accumulated in the ECM; (d) intercellular and cell-matrix interactions; (e) differentiation and dedifferentiation of the cells, their migration and biosynthesis activity. Feasible pathways promoting cartilage regeneration include: 1. additional cellular supply from bone marrow and blood; 2. biosynthesis amplification of the ECM components, 3. stimulation of the motility of mature chondrocytes, and 4. activation of resident adult stem cells toward their proliferation, differentiation, and ECM production. The main reasons of the low regeneration potential of cartilage are advanced differentiation of the resident chondrocytes and relatively slow metabolism of the tissue. The nonablative laser radiation may provide controllable thermal and mechanical effects (as on the cells, as on the matrix) resulting in activation of the cellular biosynthesis. In particular, nonuniform laser heating of cartilage induces heterogeneous thermal expansion, stress, and also the movement of the interstitial water and ions (see Fig. 3 and Sec. 4).
Fig. 3
Targets and mechanisms of the laser-induced regeneration of cartilage. Thin straight arrows show the direct laser influence on the components of cartilage. White thick arrows represent the most important biological responses to laser action. Dark thick arrows show the processes leading directly to regeneration.
One of the major obstacles for regeneration of cartilage, including partial-thickness defect of articular cartilage, is its avascularity, which hampers the progenitor cell movement from the blood and marrow to the damaged areas of the tissue. Preventing an entry of unspecialized cells and diminishing the rate of cartilage repair that slow regeneration, nevertheless, may have its good point, as it may potentially result in the growth of well organized tissue of the hyaline cartilage. Rapid repair of the full-thickness cartilage defects usually leads to undesirable growth of fibrous connective tissue or fibrous cartilage due to the impact of blood and bone-marrow-derived cells. It can be better understood by the following analogy. It is known that skin wound healing resulting in a fibrous scar is going through emergency regeneration due to swift proliferation of unspecialized fibroblasts. Their sources are the precursor cells coming into the wound via blood. These cells have nonspecific genetic program and form scar.47 In a similar manner, the bone-marrow cells coming to the full-thickness defect of articular cartilage differentiate into the fibroblasts of nonspecific connective tissue or into the chondrocytes of the fibrous cartilage. This provides quick filling of the defect, but fails in functionality of the novel tissue. One of the possible ways to promote growth of the hyaline cartilage in the full-thickness defects of articular cartilage plates can be laser-induced coagulation of the bottom of the defect. This may prevent access of unspecialized precursor cells from the blood or bone-marrow in order to develop more specific, i.e., hyaline cartilage.
It is known that in the course of embryogenesis, the hyaline cartilage forms in the zones undergoing compression load (articular cartilage), whereas, the fibrous cartilage (meniscus, annulus fibrosis of the intervertebral disk) usually develops in the stretched or torsioned zones. Spatial and temporal modulation of laser radiation allows controlling the actual distribution of stretched and compressed zones in cartilage. The mechanical loads are important factors governing an orchestra of chondrogenesis, including the processes of cellular differentiation. Therefore, the nonablative laser treatment may play a triggering role in the differentiation of immature cartilage cells. Laser radiation may probably be responsible for the reverse process of dedifferentiation of the mature chondrocytes leading to the recovery of their ability to divide. Existing natural pathways of cells dedifferentiation (see Fig. 2) open possibilities for tissue correction, in particular, replacement of abnormally grown fibrous tissue by hyaline cartilage possessing adequate mechanical and functional properties (Fig. 3).
Laser radiation can also be used to stimulate proliferation and acquiring the specialized phenotype by resident stem cells or MSC coming through synovial liquid in order to promote their transformation into mature hyaline-like chondrocytes. This approach is critically significant for healing of the partial-thickness defects of articular cartilage. At the same time, as the cellular population in full-thickness cartilage defect is highly heterogenic, laser irradiation may effect the proliferation of different kinds of cells. Thereafter, the additional controlling factor of the ECM architecture should be taken into account. Laser modification of the fine structure of ECM does not change its general organization. This provides natural environments for chondrocytes and promotes restoration of the hyaline type cartilage. One of the important factors is cell movement velocity, which correlates with the alignment of the matrix fibrillar components.48, 49 Nonablative laser irradiation allows structure modification and diffusion properties of ECM. This may support cell movement and favor tissue regeneration.
Laser-induced growth of hyaline cartilage in elastic cartilage was established in the course of in vivoexperiments on laser reshaping of porcine ears.50 The effects of laser irradiation on gene expression of chondrocytes and collagen of ECM have been studied for rabbit septal cartilage using laser settings typical for laser reshaping procedure.51 It was shown that laser irradiation of cartilage does not result in the detection of collagen type I. Only collagen type II was observed after laser irradiation in the corresponding cell culture in vitro. This fact indicates that cartilage cellular response to nonablative laser irradiation differs from the reaction of conventional wound healing. Laser irradiation of cartilage can leave intact collagen and preserve general matrix architecture, which favors chondrocyte survival and promotes new tissue growth. Evidence of hyaline cartilage development in laser-irradiated intervertebral disks was revealed in the animal experiments (see Sec. 5.2). The advantage of the laser effect on chondrocytes proliferation compared to other thermal, mechanical, and chemical effects was demonstrated in Ref. 52. No evidence of chondrocyte DNA replication was observed in tissues heated using nonlaser methods, grown in TGF-β-contained media, or mechanically traumatized. In contrast, for laser irradiated chondrocytes, flow cytometry provided evidence that laser irradiation causes a proliferative response in cho...
Original Source: https://www.spiedigitallibrary.org/journals/journal-of-biomedical-optics/volume-16/issue-08/080902/Laser-induced-regeneration-of-cartilage/10.1117/1.3614565.full?SSO=1
Lasers, stem cells, and COPD
Feng Lin†, Steven F Josephs†, Doru T Alexandrescu†, Famela Ramos, Vladimir Bogin, Vincent Gammill, Constantin A Dasanu, Rosalia De Necochea-Campion, Amit N Patel, Ewa Carrier and David R Koos - 2010 (Publication) 4469This well annotated paper looks at the biphasic response to try to find the appropriate dosage for several applications including COPD.
Lasers, stem cells, and COPD
· Feng Lin†,
· Steven F Josephs†,
· Doru T Alexandrescu†,
· Famela Ramos,
· Vladimir Bogin,
· Vincent Gammill,
· Constantin A Dasanu,
· Rosalia De Necochea-Campion,
· Amit N Patel,
· Ewa Carrier and
· David R KoosEmail author
†Contributed equally
Journal of Translational Medicine20108:16
https://doi.org/10.1186/1479-5876-8-16
© Lin et al; licensee BioMed Central Ltd. 2010
Received: 7 January 2010
Accepted: 16 February 2010
Published: 16 February 2010
Abstract
The medical use of low level laser (LLL) irradiation has been occurring for decades, primarily in the area of tissue healing and inflammatory conditions. Despite little mechanistic knowledge, the concept of a non-invasive, non-thermal intervention that has the potential to modulate regenerative processes is worthy of attention when searching for novel methods of augmenting stem cell-based therapies. Here we discuss the use of LLL irradiation as a "photoceutical" for enhancing production of stem cell growth/chemoattractant factors, stimulation of angiogenesis, and directly augmenting proliferation of stem cells. The combination of LLL together with allogeneic and autologous stem cells, as well as post-mobilization directing of stem cells will be discussed.
Introduction (Personal Perspective)
We came upon the field of low level laser (LLL) therapy by accident. One of our advisors read a press release about a company using this novel technology of specific light wavelengths to treat stroke. Given the possible role of stem cells in post-stroke regeneration, we decided to cautiously investigate. As a background, it should be said that our scientific team has been focusing on the area of cord blood banking and manufacturing of disposables for processing of adipose stem cells for the past 3 years. Our board has been interested in strategically refocusing the company from services-oriented into a more research-focused model. An unbiased exploration into the various degenerative conditions that may be addressed by our existing know-how led us to explore the condition of chronic obstructive pulmonary disease (COPD), an umbrella term covering chronic bronchitis and emphysema, which is the 4th largest cause of death in the United States. As a means of increasing our probability of success in treatment of this condition, the decision was made to develop an adjuvant therapy that would augment stem cell activity. The field of LLL therapy attracted us because it appeared to be relatively unexplored scientific territory for which large amounts of clinical experience exist. Unfortunately, it was difficult to obtain the cohesive "state-of-the-art" description of the molecular/cellular mechanisms of this therapy in reviews that we have searched. Therefore we sought in this mini-review to discuss what we believe to be relevant to investigators attracted by the concept of "regenerative photoceuticals". Before presenting our synthesis of the field, we will begin by describing our rationale for approaching COPD with the autologous stem cell based approaches we are developing.
COPD as an Indication for Stem Cell Therapy
COPD possesses several features making it ideal for stem cell based interventions: a) the quality of life and lack of progress demands the ethical exploration of novel approaches. For example, bone marrow stem cells have been used in over a thousand cardiac patients with some indication of efficacy [1, 2]. Adipose-based stem cell therapies have been successfully used in thousands of race-horses and companion animals without adverse effects [3], as well as numerous clinical trials are ongoing and published human data reports no adverse effects (reviewed in ref [4]). Unfortunately, evaluation of stem cell therapy in COPD has lagged behind other areas of regenerative investigation; b) the underlying cause of COPD appears to be inflammatory and/or immunologically mediated. The destruction of alveolar tissue is associated with T cell reactivity [5, 6], pathological pulmonary macrophage activation [7], and auto-antibody production [8]. Mesenchymal stem cells have been demonstrated to potently suppress autoreactive T cells [9, 10], inhibit macrophage activation [11], and autoantibody responses [12]. Additionally, mesenchymal stem cells can be purified in high concentrations from adipose stromal vascular tissue together with high concentrations of T regulatory cells [4], which in animal models are approximately 100 more potent than peripheral T cells at secreting cytokines therapeutic for COPD such as IL-10 [13, 14]. Additionally, use of adipose derived cells has yielded promising clinical results in autoimmune conditions such as multiple sclerosis [4]; and c) Pulmonary stem cells capable of regenerating damaged parenchymal tissue have been reported [15]. Administration of mesenchymal stem cells into neonatal oxygen-damaged lungs, which results in COPD-like alveoli dysplasia, has been demonstrated to yield improvements in two recent publications [16, 17].
Based on the above rationale for stem cell-based COPD treatments, we began our exploration into this area by performing several preliminary experiments and filing patents covering combination uses of stem cells with various pharmacologically available antiinflammatories, as well as methods of immune modulation. These have served as the basis for two of our pipeline candidates, ENT-111, and ENT-894. As a commercially-oriented organization, we needed to develop a therapeutic candidate that not only has a great potential for efficacy, but also can be easily implemented as part of the standard of care. Our search led us to the area of low level laser (LLL) therapy. From our initial perception as neophytes to this field, the area of LLL therapy has been somewhat of a medical mystery. A pubmed search for "low level laser therapy" yields more than 1700 results, yet before stumbling across this concept, none of us, or our advisors, have ever heard of this area of medicine.
On face value, this field appeared to be somewhat of a panacea: clinical trials claiming efficacy for conditions ranging from alcoholism [18], to sinusitis [19], to ischemic heart disease [20]. Further confusing was that many of the studies used different types of LLL-generating devices, with different parameters, in different model systems, making comparison of data almost impossible. Despite this initial impression, the possibility that a simple, non-invasive methodology could exist that augments regenerative potential in a tissue-focused manner became very enticing to us. Specific uses envisioned, for which intellectual property was filed included using light to concentrate stem cells to an area of need, to modulate effects of stem cells once they are in that specific area, or even to use light together with other agents to modulate endogenous stem cells.
The purpose of the current manuscript is to overview some of the previous work performed in this area that was of great interest to our ongoing work in regenerative medicine. We believe that greater integration of the area of LLL with current advancements in molecular and cellular biology will accelerate medical progress. Unfortunately, in our impression to date, this has been a very slow process.
What is Low Level Laser Irradiation?
Lasers (Light amplification by stimulated emission of radiation) are devices that typically generate electromagnetic radiation which is relatively uniform in wavelength, phase, and polarization, originally described by Theodore Maiman in 1960 in the form of a ruby laser [21]. These properties have allowed for numerous medical applications including uses in surgery, activation of photodynamic agents, and various ablative therapies in cosmetics that are based on heat/tissue destruction generated by the laser beam [22, 23, 24]. These applications of lasers are considered "high energy" because of their intensity, which ranges from about 10-100 Watts. The subject of the current paper will be another type of laser approach called low level lasers (LLL) that elicits effects through non-thermal means. This area of investigation started with the work of Mester et al who in 1967 reported non-thermal effects of lasers on mouse hair growth [25]. In a subsequent study [26], the same group reported acceleration of wound healing and improvement in regenerative ability of muscle fibers post wounding using a 1 J/cm2 ruby laser. Since those early days, numerous in vitro and in vivo studies have been reported demonstrating a wide variety of therapeutic effects involving LLL, a selected sample of which will be discussed below. In order to narrow our focus of discussion, it is important to first begin by establishing the current definition of LLL therapy. According to Posten et al [27], there are several parameters of importance: a) Power output of laser being 10-3 to 10-1 Watts; b) Wavelength in the range of 300-10,600 nm; c) Pulse rate from 0, meaning continuous to 5000 Hertz (cycles per second); d) intensity of 10-2-10 W/cm(2) and dose of 0.01 to 100 J/cm2. Most common methods of administering LLL radiation include lasers such as ruby (694 nm), Ar (488 and 514 nm), He-Ne (632.8 nm), Krypton (521, 530, 568, and 647 nm), Ga-Al-As (805 or 650 nm), and Ga-As (904 nm). Perhaps one of the most distinguishing features of LLL therapy as compared to other photoceutical modalities is that effects are mediated not through induction of thermal effects but rather through a process that is still not clearly defined called "photobiostimulation". It appears that this effect of LLL is not depend on coherence, and therefore allows for use of non-laser light generating devices such as inexpensive Light Emitting Diode (LED) technology [28].
To date several mechanisms of biological action have been proposed, although none are clearly established. These include augmentation of cellular ATP levels [29], manipulation of inducible nitric oxide synthase (iNOS) activity [30, 31], suppression of inflammatory cytokines such as TNF-alpha, IL-1beta, IL-6 and IL-8 [32, 33, 34, 35, 36], upregulation of growth factor production such as PDGF, IGF-1, NGF and FGF-2 [36, 37, 38, 39], alteration of mitochondrial membrane potential [29, 40, 41, 42] due to chromophores found in the mitochondrial respiratory chain [43, 44] as reviewed in [45], stimulation of protein kinase C (PKC) activation [46], manipulation of NF-κB activation [47], direct bacteriotoxic effect mediated by induction of reactive oxygen species (ROS) [48], modification of extracellular matrix components [49], inhibition of apoptosis [29], stimulation of mast cell degranulation [50], and upregulation of heat shock proteins [51]. Unfortunately these effects have been demonstrated using a variety of LLL devices in non-comparable models. To add to confusion, dose-dependency seems to be confined to such a narrow range or does not seem to exist in that numerous systems therapeutic effects disappear with increased dose.
In vitro studies of LLL
In areas of potential phenomenology, it is important to begin by assessing in vitro studies reported in the literature in which reproducibility can be attained with some degree of confidence, and mechanistic dissection is simpler as compared with in vivo systems. In 1983, one of the first studies to demonstrate in vitro effects of LLL was published. The investigators used a helium neon (He-Ne) laser to generate a visible red light at 632.8 nm for treatment of porcine granulosa cells. The paper described upregulation of metabolic and hormone-producing activity of the cells when exposed for 60 seconds to pulsating low power (2.8 mW) irradiation [52]. The possibility of modulating biologically-relevant signaling proteins by LLL was further assessed in a study using an energy dose of 1.5 J/cm2 in cultured keratinocytes. Administration of He-Ne laser emitted light resulted in upregulated gene expression of IL-1 and IL-8 [53]. Production of various growth factors in vitro suggests the possibility of enhanced cellular mitogenesis and mobility as a result of LLL treatment. Using a diode-based method to generate a similar wavelength to the He-Ne laser (363 nm), Mvula et al reported in two papers that irradiation at 5 J/cm2 of adipose derived mesenchymal stem cells resulted in enhanced proliferation, viability and expression of the adhesion molecule beta-1 integrin as compared to control [54, 55]. In agreement with possible regenerative activity based on activation of stem cells, other studies have used an in vitro injury model to examine possible therapeutic effects. Migration of fibroblasts was demonstrated to be enhanced in a "wound assay" in which cell monolayers are scraped with a pipette tip and amount of time needed to restore the monolayer is used as an indicator of "healing". The cells exposed to 5 J/cm2 generated by an He-Ne laser migrated rapidly across the wound margin indicating a stimulatory or positive influence of phototherapy. Higher doses (10 and 16 J/cm2) caused a decrease in cell viability and proliferation with a significant amount of damage to the cell membrane and DNA [56]. In order to examine whether LLL may positively affect healing under non-optimal conditions that mimic clinical situations treatment of fibroblasts from diabetic animals was performed. It was demonstrated that with the He-Ne laser dosage of 5 J/cm2 fibroblasts exhibited an enhanced migration activity, however at 16 J/cm2 activity was negated and cellular damage observed [57]. Thus from these studies it appears that energy doses from 1.5 J/cm2 to 5 J/cm2 are capable of eliciting "biostimulatory effects" in vitro in the He-Ne-based laser for adherent cells that may be useful in regeneration such as fibroblasts and mesenchymal stem cells.
Studies have also been performed in vitro on immunological cells. High intensity He-Ne irradiation at 28 and 112 J/cm2 of human peripheral blood mononuclear cells, a heterogeneous population of T cells, B cells, NK cells, and monocytes has been described to induce chromatin relaxation and to augment proliferative response to the T cell mitogen phytohemaglutin [58]. In human peripheral blood mononuclear cells (PBMC), another group reported in two papers that interleukin-1 alpha (IL-1 alpha), tumor necrosis factor-alpha (TNF-alpha), interleukin-2 (IL-2), and interferon-gamma (IFN-gamma) at a protein and gene level in PBMC was increased after He-Ne irradiation at 18.9 J/cm2 and decreased with 37.8 J/cm2 [59, 60]. Stimulation of human PBMC proliferation and murine splenic lymphocytes was also reported with He-Ne LLL [61, 62]. In terms of innate immune cells, enhanced phagocytic activity of murine macrophages have been reported with energy densities ranging from 100 to 600 J/cm2, with an optimal dose of 200 J/cm2 [63]. Furthermore, LLL has been demonstrated to augment human monocyte killing mycobacterial cells at similar densities, providing a functional correlation [64].
Thus from the selected in vitro studies discussed, it appears that modulation of proliferation and soluble factor production by LLL can be reliably reproduced. However the data may be to some extent contradictory. For example, the over-arching clinical rationale for use of LLL in conditions such as sinusitis [65], arthritis [66, 67], or wound healing [68] is that treatment is associated with anti-inflammatory effects. However the in vitro studies described above suggested LLL stimulates proinflammatory agents such as TNF-alpha or IL-1 [59, 60]. This suggests the in vivo effects of LLL may be very complex, which to some extent should not be surprising. Factors affecting LLL in vivo actions would include degree of energy penetration through the tissue, the various absorption ability of cells in the various tissues, and complex chemical changes that maybe occurring in paracrine/autocrine manner. Perhaps an analogy to the possible discrepancy between LLL effects in vitro versus in vivo may be made with the medical practice of extracorporeal ozonation of blood. This practice is similar to LLL therapy given that it is used in treatment of conditions such as atherosclerosis, non-healing ulcers, and various degenerative conditions, despite no clear mechanistic understanding [69, 70, 71]. In vitro studies have demonstrated that ozone is a potent oxidant and inducer of cell apoptosis and inflammatory signaling [72, 73, 74]. In contrast, in vivo systemic changes subsequent to administration of ozone or ozonized blood in animal models and patients are quite the opposite. Numerous investigators have published enhanced anti-oxidant enzyme activity such as elevations in Mg-SOD and glutathione-peroxidase levels, as well as diminishment of inflammation-associated pathology [75, 76, 77, 78]. Regardless of the complexity of in vivo situations, the fact that reproducible, in vitro experiments, demonstrate a biological effect provided support for us that there is some basis for LLL and it is not strictly an area of phenomenology.
Animal Studies with LLL
As early as 1983, Surinchak et al reported in a rat skin incision healing model that wounds exposed He-Ne radiation of fluency 2.2 J/cm2 for 3 min twice daily for 14 days demonstrated a 55% increase in breaking strength over control rats. Interestingly, higher doses yielded poorer healing [79]. This application of laser light was performed directly on shaved skin. In a contradictory experiment, it was reported that rats irradiated for 12 days with four levels of laser light (0.0, 0.47, 0.93, and 1.73 J/cm2) a possible strengthening of wounds tension was observed at the highest levels of irradiation (1.73 J/cm2), however it did not reach significance when analyzed by resampling statistics [80]. In another wound-healing study Ghamsari et al reported accelerated healing in the cranial surface of teats in dairy cows by administration of He-Ne irradiation at 3.64 J/cm2 dose of low-level laser, using a helium-neon system with an output of 8.5 mW, continuous wave [81]. Collagen fibers in LLL groups were denser, thicker, better arranged and more continuous with existing collagen fibers than those in non-LLL groups. The mean tensile strength was significantly greater in LLL groups than in non-LLL groups [82]. In the random skin flap model, the use of He-Ne laser irradiation with 3 J/cm2 energy density immediately after the surgery and for the four subsequent days was evaluated in 4 experimental groups: Group 1 (control) sham irradiation with He-Ne laser; Group 2 irradiation by punctual contact technique on the skin flap surface; Group 3 laser irradiation surrounding the skin flap; and Group 4 laser irradiation both on the skin flap surface and around it. The percentage of necrotic area of the four groups was determined on day 7-post injury. The control group had an average necrotic area of 48.86%; the group irradiated on the skin flap surface alone had 38.67%; the group irradiated around the skin flap had 35.34%; and the group irradiated one the skin flap surface and around it had 22.61%. All experimental groups reached statistically significant values when compared to control [83]. Quite striking results were obtained in an alloxan-induced diabetes wound healing model in which a circular 4 cm2 excisional wound was created on the dorsum of the diabetic rats. Treatment with He-Ne irradiation at 4.8 J/cm2 was performed 5 days a week until the wound healed completely and compared to sham irradiated animals. The laser-treated group healed on average by the 18th day whereas, the control group healed on average by the 59th day [84].
In addition to mechanically-induced wounds, beneficial effects of LLL have been obtained in burn-wounds in which deep second-degree burn wounds were induced in rats and the effects of daily He-Ne irradiation at 1.2 and 2.4 J/cm2 were assessed in comparison to 0.2% nitrofurazone cream. The number of macrophages at day 16, and the depth of new epidermis at day 30, was significantly less in the laser treated groups in comparison with control and nitrofurazone treated groups. Additionally, infections with S. epidermidis and S. aureus were significantly reduced [85].
While numerous studies have examined dermatological applications of LLL, which may conceptually be easier to perform due to ability to topically apply light, extensive investigation has also been made in the area of orthopedic applications. Healing acceleration has been observed in regeneration of the rat mid-cortical diaphysis of the tibiae, which is a model of post-injury bone healing. A small hole was surgically made with a dentistry burr in the tibia and the injured area and LLL was administered over a 7 or 14 day course transcutaneously starting 24 h from surgery. Incident energy density dosages of 31.5 and 94.5 J/cm2 were applied during the period of the tibia wound healing. Increased angiogenesis was observed after 7 days irradiation at an energy density of 94.5 J/cm2, but significantly decreased the number of vessels in the 14-day irradiated tibiae, independent of the dosage [86]. In an osteoarthritis model treatment with He-Ne resulted in augmentation of heat shock proteins and pathohistological improvement of arthritic cartilage [87]. The possibility that a type of preconditioning response is occurring, which would involve induction of genes such as hemoxygenase-1 [88], remains to be investigated. Effects of LLL therapy on articular cartilage were confirmed by another group. The experiment consisted of 42 young Wistar rats whose hind limbs were operated on in order to immobilize the knee joint. One week after operation they were assigned to three groups; irradiance 3.9 W/cm2, 5.8 W/cm2, and sham treatment. After 6 times of treatment for another 2 weeks significantpreservation of articular cartilage stiffness with 3.9 and 5.8 W/cm2therapy was observed [89].
Muscle regeneration by LLL was demonstrated in a rat model of disuse atrophy in which eight-week-old rats were subjected to hindlimb suspension for 2 weeks, after which they were released and recovered. During the recovery period, rats underwent daily LLL irradiation (Ga-Al-As laser; 830 nm; 60 mW; total, 180 s) to the right gastrocnemius muscle through the skin. After 2-weeks the number of capillaries and fibroblast growth factor levels exhibited significant elevation relative to those of the LLL-untreated muscles. LLL treatment induced proliferation in satellite cells as detected by BRdU [90].
Other animal studies of LLL have demonstrated effects in areas that appear unrelated such as suppression of snake venom induced muscle death [91], decreasing histamine-induced vasospasms [92], inhibition of post-injury restenosis [93], and immune stimulation by thymic irradiation [94].
Clinical Studies Using LLL
Growth factor secretion by LLL and its apparent regenerative activities have stimulated studies in radiation-induced mucositis. A 30 patient randomized trial of carcinoma patients treated by radiotherapy alone (65 Gy at a rate of 2 Gy/fraction, 5 fractions per week) without prior surgery or concomitant chemotherapy suffering from radiation-induced mucositis was performed using a He-Ne 60 mW laser. Grade 3 mucositis occured with a frequency of 35.2% in controls and at 7.6% of treated patients. Furthermore, a decrease in "severe pain" (grade 3) was observed in that 23.8% in the control group experienced this level of pain, as compared to 1.9% in the treatment group [95]. A subsequent study reported similar effects [96].
Healing ability of lasers was also observed in a study of patients with gingival flap incisions. Fifty-eight extraction patients had one of two gingival flap incisions lased with a 1.4 mW He-Ne (670 nm) at 0.34 J/cm2. Healing rates were evaluated clinically and photographically. Sixty-nine percent of the irradiated incisions healed faster than the control incisions. No significant difference in healing was noted when patients were compared by age, gender, race, and anatomic location of the incision [97]. Another study evaluating healing effects of LLL in dental practice examined 48 patients subjected to surgical removal of their lower third molars. Treated patients were administered Ga-Al-As diode generated 808 nm at a dose of 12 J. The study demonstrated that extraoral LLL is more effective than intraoral LLL, which was more effective than control for the reduction of postoperative trismus and swelling after extraction of the lower third molar [98].
Given the predominance of data supporting fibroblast proliferative ability and animal wound healing effects of LLL therapy, a clinical trial was performed on healing of ulcers. In a double-blinded fashion 23 diabetic leg ulcers from 14 patients were divided into two groups. Phototherapy was applied (<1.0 J/cm2) twice per week, using a Dynatron Solaris 705(R) LED device that concurrently emits 660 and 890 nm energies. At days 15, 30, 45, 60, 75, and 90 mean ulcer granulation and healing rates were significantly higher for the treatment group as compared to control. By day 90, 58.3% of the ulcers in the LLL treated group were fully healed and 75% achieved 90-100% healing. In the placebo group only one ulcer healed fully [68].
As previously mentioned, LLL appears to have some angiogenic activity. One of the major problems in coronary artery disease is lack of collateralization. In a 39 patient study advanced CAD, two sessions of irradiation of low-energy laser light on skin in the chest area from helium-neon B1 lasers. The time of irradiation was 15 minutes while operations were performed 6 days a week for one month. Reduction in Canadian Cardiology Society (CCS) score, increased exercise capacity and time, less frequent angina symptoms during the treadmill test, longer distance of 6-minute walk test and a trend towards less frequent 1 mm ST depression lasting 1 min during Holter recordings was noted after therapy [99].
Perhaps one of the largest clinical trials with LLL was the NEST trial performed by Photothera. In this double blind trial 660 stroke patients were recruited and randomized: 331 received LLL and 327 received sham. No prespecified test achieved significance, but a post hoc analysis of patients with a baseline National Institutes of Health Stroke Scale score of <16 showed a favorable outcome at 90 days on the primary end point (P < 0.044) [100]. Currently Photothera is in the process of repeating this trial with modified parameters.
Relevance of LLL to COPD
A therapeutic intervention in COPD would require addressing the issues of inflammation and regeneration. Although approaches such as administration of bone marrow stem cells, or fat derived cellular components have both regenerative and anti-inflammatory activity in animal models, the need to enhance their potency for clinical applications can be seen in the recent Osiris's COPD trial interim data which reported no significant improvement in pulmonary function [101]. Accordingly, we sought to develop a possible rationale for how LLL may be useful as an adjunct to autologous stem cell therapy.
Table 1 depicts some of the properties of LLL that provide a rationale for the combined use with stem cells. One of the basic properties of LLL seems to be ability to inhibit inflammation at the level of innate immune activation. Representative studies showed that LLL was capable of suppressing inflammatory genes and/or pathology after administration of lipopolysaccharide (LPS) as a stimulator of monocytes [102] and bronchial cells [34], in vitro, and leukocyte infiltration in vivo [103, 104]. Inflammation induced by other stimulators such as zymosan, carrageenan, and TNF-alpha was also inhibited by LLL [32, 105, 106]. Growth factor stimulating activity of LLL was demonstrated in both in vitro and in vivo experiments in which augmentation of FGF-2, PDGF and IGF-1 was observed [36, 37, 107]. Endogenous production of these growth factors may be useful in regeneration based on activation of endogenous pulmonary stem cells [108, 109]. Another aspect of LLL activities of relevance is ability to stimulate angiogenesis. In COPD, the constriction of blood vessels as a result of poor oxygen uptake is results in a feedback loop culminating in pulmonary hypertension. Administration of angiogenic factors has been demonstrated to be beneficial in several animal models of pulmonary pathology [110, 111]. The ability of LLL to directly induce proliferation of HUVEC cells [112], as well as to augment production of angiogenic factors such as VEGF [113], supports the possibility of creation of an environment hospitable to neoangiogenesis which is optimal for stem cell growth. In fact, a study demonstrated in vivo induction of neocapillary formation subsequent to LLL administration in a hindlimb ischemia model [114]. The critical importance of angiogenesis in stem cell mediated regeneration has previously been demonstrated in the stroke model, where the major therapeutic activity of exogenous stem cells has been attributed to angiogenic as opposed to transdifferentiation effects [115].
Table 1 Examples of LLL Properties Relevant to COPD COPD Property LLL Experiment
Original Source: https://translational-medicine.biomedcentral.com/articles/10.1186/1479-5876-8-16
Abstract
The medical use of low level laser (LLL) irradiation has been occurring for decades, primarily in the area of tissue
healing and inflammatory conditions. Despite little mechanistic knowledge, the concept of a non-invasive, nonthermal
intervention that has the potential to modulate regenerative processes is worthy of attention when searching
for novel methods of augmenting stem cell-based therapies. Here we discuss the use of LLL irradiation as a
“photoceutical” for enhancing production of stem cell growth/chemoattractant factors, stimulation of angiogenesis,
and directly augmenting proliferation of stem cells. The combination of LLL together with allogeneic and autologous
stem cells, as well as post-mobilization directing of stem cells will be discussed.
Introduction (Personal Perspective)
We came upon the field of low level laser (LLL) therapy
by accident. One of our advisors read a press release
about a company using this novel technology of specific
light wavelengths to treat stroke. Given the possible role
of stem cells in post-stroke regeneration, we decided to
cautiously investigate. As a background, it should be
said that our scientific team has been focusing on the
area of cord blood banking and manufacturing of disposables
for processing of adipose stem cells for the past 3
years. Our board has been interested in strategically
refocusing the company from services-oriented into a
more research-focused model. An unbiased exploration
into the various degenerative conditions that may be
addressed by our existing know-how led us to explore
the condition of chronic obstructive pulmonary disease
(COPD), an umbrella term covering chronic bronchitis
and emphysema, which is the 4th largest cause of death
in the United States. As a means of increasing our probability
of success in treatment of this condition, the
decision was made to develop an adjuvant therapy that
would augment stem cell activity. The field of LLL therapy
attracted us because it appeared to be relatively
unexplored scientific territory for which large amounts
of clinical experience exist. Unfortunately, it was difficult
to obtain the cohesive “state-of-the-art” description of
the molecular/cellular mechanisms of this therapy in
reviews that we have searched. Therefore we sought in
this mini-review to discuss what we believe to be relevant
to investigators attracted by the concept of “regenerative
photoceuticals”. Before presenting our synthesis
of the field, we will begin by describing our rationale for
approaching COPD with the autologous stem cell based
approaches we are developing.
COPD as an Indication for Stem Cell Therapy
COPD possesses several features making it ideal for
stem cell based interventions: a) the quality of life and
lack of progress demands the ethical exploration of
novel approaches. For example, bone marrow stem cells
have been used in over a thousand cardiac patients with
some indication of efficacy [1,2]. Adipose-based stem
cell therapies have been successfully used in thousands
of race-horses and companion animals without adverse
effects [3], as well as numerous clinical trials are
ongoing and published human data reports no adverse
effects (reviewed in ref [4]). Unfortunately, evaluation of
stem cell therapy in COPD has lagged behind other
areas of regenerative investigation; b) the underlying
cause of COPD appears to be inflammatory and/or
immunologically mediated. The destruction of alveolar
tissue is associated with T cell reactivity [5,6], pathological
pulmonary macrophage activation [7], and auto-antibody
production [8]. Mesenchymal stem cells have been
demonstrated to potently suppress autoreactive T cells
[9,10], inhibit macrophage activation [11], and autoantibody
responses [12]. Additionally, mesenchymal stem
cells can be purified in high concentrations from adipose
stromal vascular tissue together with high concentrations of T regulatory cells [4], which in animal
models are approximately 100 more potent than peripheral
T cells at secreting cytokines therapeutic for COPD
such as IL-10 [13,14]. Additionally, use of adipose
derived cells has yielded promising clinical results in
autoimmune conditions such as multiple sclerosis [4];
and c) Pulmonary stem cells capable of regenerating
damaged parenchymal tissue have been reported [15].
Administration of mesenchymal stem cells into neonatal
oxygen-damaged lungs, which results in COPD-like
alveoli dysplasia, has been demonstrated to yield
improvements in two recent publications [16,17].
Based on the above rationale for stem cell-based
COPD treatments, we began our exploration into this
area by performing several preliminary experiments and
filing patents covering combination uses of stem cells
with various pharmacologically available antiinflammatories,
as well as methods of immune modulation. These
have served as the basis for two of our pipeline candidates,
ENT-111, and ENT-894. As a commerciallyoriented
organization, we needed to develop a therapeutic
candidate that not only has a great potential for efficacy,
but also can be easily implemented as part of the
standard of care. Our search led us to the area of low
level laser (LLL) therapy. From our initial perception as
neophytes to this field, the area of LLL therapy has been
somewhat of a medical mystery. A pubmed search for
“low level laser therapy” yields more than 1700 results,
yet before stumbling across this concept, none of us, or
our advisors, have ever heard of this area of medicine.
On face value, this field appeared to be somewhat of a
panacea: clinical trials claiming efficacy for conditions
ranging from alcoholism [18], to sinusitis [19], to
ischemic heart disease [20]. Further confusing was that
many of the studies used different types of LLL-generating
devices, with different parameters, in different model
systems, making comparison of data almost impossible.
Despite this initial impression, the possibility that a simple,
non-invasive methodology could exist that augments
regenerative potential in a tissue-focused manner
became very enticing to us. Specific uses envisioned, for
which intellectual property was filed included using light
to concentrate stem cells to an area of need, to modulate
effects of stem cells once they are in that specific
area, or even to use light together with other agents to
modulate endogenous stem cells.
The purpose of the current manuscript is to overview
some of the previous work performed in this area that was
of great interest to our ongoing work in regenerative medicine.
We believe that greater integration of the area of
LLL with current advancements in molecular and cellular
biology will accelerate medical progress. Unfortunately, in
our impression to date, this has been a very slow process.
What is Low Level Laser Irradiation?
Lasers (Light amplification by stimulated emission of
radiation) are devices that typically generate electromagnetic
radiation which is relatively uniform in wavelength,
phase, and polarization, originally described by Theodore
Maiman in 1960 in the form of a ruby laser [21]. These
properties have allowed for numerous medical applications
including uses in surgery, activation of photodynamic
agents, and various ablative therapies in cosmetics that are
based on heat/tissue destruction generated by the laser
beam [22-24]. These applications of lasers are considered
“high energy” because of their intensity, which ranges
from about 10-100 Watts. The subject of the current
paper will be another type of laser approach called low
level lasers (LLL) that elicits effects through non-thermal
means. This area of investigation started with the work of
Mester et al who in 1967 reported non-thermal effects of
lasers on mouse hair growth [25]. In a subsequent study
[26], the same group reported acceleration of wound healing
and improvement in regenerative ability of muscle
fibers post wounding using a 1 J/cm2 ruby laser. Since
those early days, numerous in vitro and in vivo studies
have been reported demonstrating a wide variety of therapeutic
effects involving LLL, a selected sample of which
will be discussed below. In order to narrow our focus of
discussion, it is important to first begin by establishing the
current definition of LLL therapy. According to Posten et
al [27], there are several parameters of importance: a)
Power output of laser being 10-3 to 10-1 Watts; b) Wavelength
in the range of 300-10,600 nm; c) Pulse rate from 0,
meaning continuous to 5000 Hertz (cycles per second); d)
intensity of 10-2-10 W/cm(2) and dose of 0.01 to 100 J/
cm2. Most common methods of administering LLL radiation
include lasers such as ruby (694 nm), Ar (488 and 514
nm), He-Ne (632.8 nm), Krypton (521, 530, 568, and 647
nm), Ga-Al-As (805 or 650 nm), and Ga-As (904 nm).
Perhaps one of the most distinguishing features of LLL
therapy as compared to other photoceutical modalities is
that effects are mediated not through induction of thermal
effects but rather through a process that is still not clearly
defined called “photobiostimulation”. It appears that this
effect of LLL is not depend on coherence, and therefore
allows for use of non-laser light generating devices such as
inexpensive Light Emitting Diode (LED) technology [28].
To date several mechanisms of biological action have
been proposed, although none are clearly established.
These include augmentation of cellular ATP levels [29],
manipulation of inducible nitric oxide synthase (iNOS)
activity [30,31], suppression of inflammatory cytokines
such as TNF-alpha, IL-1beta, IL-6 and IL-8 [32-36],
upregulation of growth factor production such as PDGF,
IGF-1, NGF and FGF-2 [36-39], alteration of mitochondrial
membrane potential [29,40-42] due to chromophores found in the mitochondrial respiratory
chain [43,44] as reviewed in [45], stimulation of protein
kinase C (PKC) activation [46], manipulation of NF-!B
activation [47], direct bacteriotoxic effect mediated by
induction of reactive oxygen species (ROS) [48], modification
of extracellular matrix components [49], inhibition
of apoptosis [29], stimulation of mast cell
degranulation [50], and upregulation of heat shock proteins
[51]. Unfortunately these effects have been demonstrated
using a variety of LLL devices in noncomparable
models. To add to confusion, dose-dependency
seems to be confined to such a narrow range or
does not seem to exist in that numerous systems therapeutic
effects disappear with increased dose.
In vitro studies of LLL
In areas of potential phenomenology, it is important to
begin by assessing in vitro studies reported in the literature
in which reproducibility can be attained with some
degree of confidence, and mechanistic dissection is simpler
as compared with in vivo systems. In 1983, one of
the first studies to demonstrate in vitro effects of LLL
was published. The investigators used a helium neon
(He-Ne) laser to generate a visible red light at 632.8 nm
for treatment of porcine granulosa cells. The paper
described upregulation of metabolic and hormone-producing
activity of the cells when exposed for 60 seconds
to pulsating low power (2.8 mW) irradiation [52]. The
possibility of modulating biologically-relevant signaling
proteins by LLL was further assessed in a study using an
energy dose of 1.5 J/cm2 in cultured keratinocytes.
Administration of He-Ne laser emitted light resulted in
upregulated gene expression of IL-1 and IL-8 [53]. Production
of various growth factors in vitro suggests the
possibility of enhanced cellular mitogenesis and mobility
as a result of LLL treatment. Using a diode-based
method to generate a similar wavelength to the He-Ne
laser (363 nm), Mvula et al reported in two papers that
irradiation at 5 J/cm2 of adipose derived mesenchymal
stem cells resulted in enhanced proliferation, viability
and expression of the adhesion molecule beta-1 integrin
as compared to control [54,55]. In agreement with possible
regenerative activity based on activation of stem
cells, other studies have used an in vitro injury model to
examine possible therapeutic effects. Migration of fibroblasts
was demonstrated to be enhanced in a “wound
assay” in which cell monolayers are scraped with a pipette
tip and amount of time needed to restore the
monolayer is used as an indicator of “healing”. The cells
exposed to 5 J/cm2 generated by an He-Ne laser
migrated rapidly across the wound margin indicating a
stimulatory or positive influence of phototherapy.
Higher doses (10 and 16 J/cm2) caused a decrease in
cell viability and proliferation with a significant amount
of damage to the cell membrane and DNA [56]. In
order to examine whether LLL may positively affect
healing under non-optimal conditions that mimic clinical
situations treatment of fibroblasts from diabetic animals
was performed. It was demonstrated that with the
He-Ne laser dosage of 5 J/cm2 fibroblasts exhibited an
enhanced migration activity, however at 16 J/cm2 activity
was negated and cellular damage observed [57]. Thus
from these studies it appears that energy doses from 1.5
J/cm2 to 5 J/cm2 are capable of eliciting “biostimulatory
effects” in vitro in the He-Ne-based laser for adherent
cells that may be useful in regeneration such as fibroblasts
and mesenchymal stem cells.
Studies have also been performed in vitro on immunological
cells. High intensity He-Ne irradiation at 28
and 112 J/cm2 of human peripheral blood mononuclear
cells, a heterogeneous population of T cells, B cells, NK
cells, and monocytes has been described to induce chromatin
relaxation and to augment proliferative response
to the T cell mitogen phytohemaglutin [58]. In human
peripheral blood mononuclear cells (PBMC), another
group reported in two papers that interleukin-1 alpha
(IL-1 alpha), tumor necrosis factor-alpha (TNF-alpha),
interleukin-2 (IL-2), and interferon-gamma (IFNgamma)
at a protein and gene level in PBMC was
increased after He-Ne irradiation at 18.9 J/cm2 and
decreased with 37.8 J/cm2 [59,60]. Stimulation of human
PBMC proliferation and murine splenic lymphocytes
was also reported with He-Ne LLL [61,62]. In terms of
innate immune cells, enhanced phagocytic activity of
murine macrophages have been reported with energy
densities ranging from 100 to 600 J/cm2, with an optimal
dose of 200 J/cm2 [63]. Furthermore, LLL has been
demonstrated to augment human monocyte killing
mycobacterial cells at similar densities, providing a functional
correlation [64].
Thus from the selected in vitro studies discussed, it
appears that modulation of proliferation and soluble factor
production by LLL can be reliably reproduced. However
the data may be to some extent contradictory. For
example, the over-arching clinical rationale for use of
LLL in conditions such as sinusitis [65], arthritis [66,67],
or wound healing [68] is that treatment is associated
with anti-inflammatory effects. However the in vitro studies
described above suggested LLL stimulates proinflammatory
agents such as TNF-alpha or IL-1 [59,60].
This suggests the in vivo effects of LLL may be very
complex, which to some extent should not be surprising.
Factors affecting LLL in vivo actions would include
degree of energy penetration through the tissue, the various
absorption ability of cells in the various tissues, and
complex chemical changes that maybe occurring in
paracrine/autocrine manner. Perhaps an analogy to the
possible discrepancy between LLL effects in vitro versus in vivo may be made with the medical practice of extracorporeal
ozonation of blood. This practice is similar to
LLL therapy given that it is used in treatment of conditions
such as atherosclerosis, non-healing ulcers, and
various degenerative conditions, despite no clear
mechanistic understanding [69-71]. In vitro studies have
demonstrated that ozone is a potent oxidant and inducer
of cell apoptosis and inflammatory signaling [72-74].
In contrast, in vivo systemic changes subsequent to
administration of ozone or ozonized blood in animal
models and patients are quite the opposite. Numerous
investigators have published enhanced anti-oxidant
enzyme activity such as elevations in Mg-SOD and glutathione-
peroxidase levels, as well as diminishment of
inflammation-associated pathology [75-78]. Regardless
of the complexity of in vivo situations, the fact that
reproducible, in vitro experiments, demonstrate a biological
effect provided support for us that there is some
basis for LLL and it is not strictly an area of
phenomenology.
Animal Studies with LLL
As early as 1983, Surinchak et al reported in a rat skin
incision healing model that wounds exposed He-Ne
radiation of fluency 2.2 J/cm2 for 3 min twice daily for
14 days demonstrated a 55% increase in breaking
strength over control rats. Interestingly, higher doses
yielded poorer healing [79]. This application of laser
light was performed directly on shaved skin. In a contradictory
experiment, it was reported that rats irradiated
for 12 days with four levels of laser light (0.0, 0.47, 0.93,
and 1.73 J/cm2) a possible strengthening of wounds tension
was observed at the highest levels of irradiation
(1.73 J/cm2), however it did not reach significance when
analyzed by resampling statistics [80]. In another
wound-healing study Ghamsari et al reported accelerated
healing in the cranial surface of teats in dairy cows
by administration of He-Ne irradiation at 3.64 J/cm2
dose of low-level laser, using a helium-neon system with
an output of 8.5 mW, continuous wave [81]. Collagen
fibers in LLL groups were denser, thicker, better
arranged and more continuous with existing collagen
fibers than those in non-LLL groups. The mean tensile
strength was significantly greater in LLL groups than in
non-LLL groups [82]. In the random skin flap model,
the use of He-Ne laser irradiation with 3 J/cm2 energy
density immediately after the surgery and for the four
subsequent days was evaluated in 4 experimental
groups: Group 1 (control) sham irradiation with He-Ne
laser; Group 2 irradiation by punctual contact technique
on the skin flap surface; Group 3 laser irradiation surrounding
the skin flap; and Group 4 laser irradiation
both on the skin flap surface and around it. The percentage
of necrotic area of the four groups was determined
on day 7-post injury. The control group had an average
necrotic area of 48.86%; the group irradiated on the skin
flap surface alone had 38.67%; the group irradiated
around the skin flap had 35.34%; and the group irradiated
one the skin flap surface and around it had
22.61%. All experimental groups reached statistically significant
values when compared to control [83]. Quite
striking results were obtained in an alloxan-induced diabetes
wound healing model in which a circular 4 cm2
excisional wound was created on the dorsum of the diabetic
rats. Treatment with He-Ne irradiation at 4.8 J/
cm2 was performed 5 days a week until the wound
healed completely and compared to sham irradiated animals.
The laser-treated group healed on average by the
18th day whereas, the control group healed on average
by the 59th day [84].
In addition to mechanically-induced wounds, beneficial
effects of LLL have been obtained in burn-wounds
in which deep second-degree burn wounds were
induced in rats and the effects of daily He-Ne irradiation
at 1.2 and 2.4 J/cm2 were assessed in comparison to
0.2% nitrofurazone cream. The number of macrophages
at day 16, and the depth of new epidermis at day 30,
was significantly less in the laser treated groups in comparison
with control and nitrofurazone treated groups.
Additionally, infections with S. epidermidis and S. aureus
were significantly reduced [85].
While numerous studies have examined dermatological
applications of LLL, which may conceptually be
easier to perform due to ability to topically apply light,
extensive investigation has also been made in the area
of orthopedic applications. Healing acceleration has
been observed in regeneration of the rat mid-cortical
diaphysis of the tibiae, which is a model of post-injury
bone healing. A small hole was surgically made with a
dentistry burr in the tibia and the injured area and LLL
was administered over a 7 or 14 day course transcutaneously
starting 24 h from surgery. Incident energy density
dosages of 31.5 and 94.5 J/cm2 were applied during
the period of the tibia wound healing. Increased angiogenesis
was observed after 7 days irradiation at an
energy density of 94.5 J/cm2, but significantly decreased
the number of vessels in the 14-day irradiated tibiae,
independent of the dosage [86]. In an osteoarthritis
model treatment with He-Ne resulted in augmentation
of heat shock proteins and pathohistological improvement
of arthritic cartilage [87]. The possibility that a
type of preconditioning response is occurring, which
would involve induction of genes such as hemoxygenase-
1 [88], remains to be investigated. Effects of LLL
therapy on articular cartilage were confirmed by another
group. The experiment consisted of 42 young Wistar
rats whose hind limbs were operated on in order to
immobilize the knee joint. One week after operation they were assigned to three groups; irradiance 3.9 W/
cm2, 5.8 W/cm2, and sham treatment. After 6 times of
treatment for another 2 weeks significantpreservation of
articular cartilage stiffness with 3.9 and 5.8 W/cm2 therapy
was observed [89].
Muscle regeneration by LLL was demonstrated in a rat
model of disuse atrophy in which eight-week-old rats
were subjected to hindlimb suspension for 2 weeks,
after which they were released and recovered. During
the recovery period, rats underwent daily LLL irradiation
(Ga-Al-As laser; 830 nm; 60 mW; total, 180 s) to
the right gastrocnemius muscle through the skin. After
2-weeks the number of capillaries and fibroblast growth
factor levels exhibited significant elevation relative to
those of the LLL-untreated muscles. LLL treatment
induced proliferation in satellite cells as detected by
BRdU [90].
Other animal studies of LLL have demonstrated
effects in areas that appear unrelated such as suppression
of snake venom induced muscle death [91],
decreasing histamine-induced vasospasms [92], inhibition
of post-injury restenosis [93], and immune stimulation
by thymic irradiation [94].
Clinical Studies Using LLL
Growth factor secretion by LLL and its apparent regenerative
activities have stimulated studies in radiationinduced
mucositis. A 30 patient randomized trial of carcinoma
patients treated by radiotherapy alone (65 Gy at
a rate of 2 Gy/fraction, 5 fractions per week) without
prior surgery or concomitant chemotherapy suffering
from radiation-induced mucositis was performed using a
He-Ne 60 mW laser. Grade 3 mucositis occured with a
frequency of 35.2% in controls and at 7.6% of treated
patients. Furthermore, a decrease in “severe pain” (grade
3) was observed in that 23.8% in the control group
experienced this level of pain, as compared to 1.9% in
the treatment group [95]. A subsequent study reported
similar effects [96].
Healing ability of lasers was also observed in a study
of patients with gingival flap incisions. Fifty-eight extraction
patients had one of two gingival flap incisions lased
with a 1.4 mW He-Ne (670 nm) at 0.34 J/cm2. Healing
rates were evaluated clinically and photographically.
Sixty-nine percent of the irradiated incisions healed faster
than the control incisions. No significant difference
in healing was noted when patients were compared by
age, gender, race, and anatomic location of the incision
[97]. Another study evaluating healing effects of LLL in
dental practice examined 48 patients subjected to surgical
removal of their lower third molars. Treated patients
were administered Ga-Al-As diode generated 808 nm at
a dose of 12 J. The study demonstrated that extraoral
LLL is more effective than intraoral LLL, which was
more effective than control for the reduction of postoperative
trismus and swelling after extraction of the
lower third molar [98].
Given the predominance of data supporting fibroblast
proliferative ability and animal wound healing effects of
LLL therapy, a clinical trial was performed on healing of
ulcers. In a double-blinded fashion 23 diabetic leg ulcers
from 14 patients were divided into two groups. Phototherapy
was applied (<1.0 J/cm2) twice per week, using a
Dynatron Solaris 705(R) LED device that concurrently
emits 660 and 890 nm energies. At days 15, 30, 45, 60,
75, and 90 mean ulcer granulation and healing rates
were significantly higher for the treatment group as
compared to control. By day 90, 58.3% of the ulcers in
the LLL treated group were fully healed and 75%
achieved 90-100% healing. In the placebo group only
one ulcer healed fully [68].
As previously mentioned, LLL appears to have some
angiogenic activity. One of the major problems in coronary
artery disease is lack of collateralization. In a 39
patient study advanced CAD, two sessions of irradiation
of low-energy laser light on skin in the chest area from
helium-neon B1 lasers. The time of irradiation was 15
minutes while operations were performed 6 days a week
for one month. Reduction in Canadian Cardiology
Society (CCS) score, increased exercise capacity and
time, less frequent angina symptoms during the treadmill
test, longer distance of 6-minute walk test and a
trend towards less frequent 1 mm ST depression lasting
1 min during Holter recordings was noted after therapy
[99].
Perhaps one of the largest clinical trials with LLL was
the NEST trial performed by Photothera. In this double
blind trial 660 stroke patients were recruited and randomized:
331 received LLL and 327 received sham. No
prespecified test achieved significance, but a post hoc
analysis of patients with a baseline National Institutes of
Health Stroke Scale score of <16 showed a favorable
outcome at 90 days on the primary end point (P <
0.044) [100]. Currently Photothera is in the process of
repeating this trial with modified parameters.
Relevance of LLL to COPD
A therapeutic intervention in COPD would require
addressing the issues of inflammation and regeneration.
Although approaches such as administration of bone marrow
stem cells, or fat derived cellular components have
both regenerative and anti-inflammatory activity in animal
models, the need to enhance their potency for clinical
applications can be seen in the recent Osiris’s COPD trial
interim data which reported no significant improvement
in pulmonary function [101]. Accordingly, we sought to
develop a possible rationale for how LLL may be useful as
an adjunct to autologous stem cell therapy.
Table 1 Examples of LLL Properties Relevant to COPD
COPD
Property
LLL Experiment LLL Details Ref
Inflammation In vivo. Decreased joint inflammation in zymosan-induced
arthritis
Semiconductor laser (685 nm and 830 nm) at (2.5 J/cm2)
In vitro. Suppression of LPS-induced bronchial inflammation and
TNF-alpha.
655 nm at of 2.6 J/cm2
In vivo. Carrageenan-induced pleurisy had decreased leukocyte
infiltration and cytokine (TNF-alpha, IL-6, and MCP)
660 nm at 2.1 J/cm2
In vitro. LPS stimulated Raw 264.7 monocytes had reduced gene
expression of MCP-1, IL-1 and IL-6
780 nm diode laser at 2.2 J/cm2)
In vivo. Suppression of LPS-stimulated neutrophil influx,
myeloperoxidase activity and IL-1beta in bronchoalveolar lavage
fluid.
660 nm diode laser at 7.5 J/cm2
In vitro. Inhibition of TNF-alpha induced IL-1, IL-8 and TNF-alpha
mRNA in human synoviocytes
810 nm (5 J/cm2) suppressed IL-1 and TNF, (25 J/cm2) also
suppressed IL-8
In vivo. Reduction of TNF-alpha in diaphragm muscle after
intravenous LPS injection.
4 sessions in 24 h with diode Ga-AsI-Al laser of 650 nm and
a total dose of 5.2 J/cm2
In vivo. Inhibition of LPS induced peritonitis and neutrophil influx 3 J/cm2 and 7.5 J/cm2
Growth Factor Production
In vivo. Upregulation of TGF-b and PDGF in rat gingiva after
incision.
He-Ne laser (632.8 nm) at a dose of 7.5 J/cm2
In vitro. Osteoblast-like cells were isolated from fetal rat calvariae
had increased IGF-1
Ga-Al-As laser (830 nm) at (3.75 J/cm2).
In vitro. Upregulated production of IGF-1 and FGF-2 in human
gingival fibroblasts.
685 nm, for 140 s, 2 J/cm2
Angiogenesis
In vivo. Increased fiber to capillary ratio in rabbits with ligated
femoral arteries.
Gallium-aluminum-arsenide (Ga-Al-As) diode laser, 904 nm
and power of 10 mW
In vitro. Stimulation of HUVEC proliferation by conditioned media
from LLL-treated T cells
820 nm at 1.2 and 3.6 J/cm2.
In vitro. 7-fold increased production of VEGF by cardiomyocytes,
1.6-fold increase by smooth muscle cells (SMC) and fibroblasts.
Supernatant of SMC had increased HUVEC-stimulating potential.
He:Ne continuous wave laser (632 nm). 0.5 J/cm2 for SMC,
2.1 J/cm2 for fibroblasts and 1.05 J/cm2 for cardiomyocytes.
In vitro. Direct stimulation of HUVEC proliferation 670 nm diode device at 2 and 8 J/cm2
Direct Stem Cell Effects
In vivo. LLL precondition significantly enhanced early cell survival
rate by 2-fold, decreased the apoptotic percentage of implanted
BMSCs in infarcted myocardium and increased the number of
newly formed capillaries.
635 nm at 0.96 J/cm2
In vitro. LLL stimulated MSC proliferation, VEGF and NGF
production, and myogenic differentiation after 5-aza induction.
635 nm diode laser at 0.5 J/cm2 for MSC proliferation, 5 J/
cm2 for VEGF and NGF production and for augmentation of
induced myogenic differentiation
In vitro. Increased proliferation of rat MSC. Red light LED 630 nm at 2 and 4 J/cm(2)
In vitro. Augmented proliferation of bone marrow and cardiac
specific stem cells.
GA-Al-As 810 nm at 1 and 3 J/cm2
In vitro/In vivo. Administration of LLL-treated MSC resulted 53%
reduction in infarct size, 5- and 6.3-fold significant increase in cell
density that positively immunoreacted to BrdU and c-kit,
respectively, and 1.4- and 2-fold higher level of angiogenesis and
vascular endothelial growth factor, respectively, when compared
to non-laser-treated implanted cells
Ga-Al-As laser (810 nm wavelength), 1 J/cm2
In vitro. Enhanced proliferation of adipose derived MSC in
presence of EGF.
636 nm diode, 5 J/cm2
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
Table 1: Examples of LLL Properties Relevant to COPD (Continued)
In vitro. Enhanced proliferation and beta-1 integrin expression of
adipose derived MSC.
635 nm diode laser, at 5 J/cm2
Clinical. 660 stroke patients: 331 received LLL and 327 received
sham. No prespecified test achieved significance, but a post hoc
analysis of patients with a baseline National Institutes of Health
Stroke Scale score of <16 showed a favorable outcome at 90
days on the primary end point (P < 0.044).
808 nm. No density disclosed.
Table 1 depicts some of the properties of LLL that provide
a rationale for the combined use with stem cells. One
of the basic properties of LLL seems to be ability to inhibit
inflammation at the level of innate immune activation.
Representative studies showed that LLL was capable of
suppressing inflammatory genes and/or pathology after
administration of lipopolysaccharide (LPS) as a stimulator
of monocytes [102] and bronchial cells [34], in vitro, and
leukocyte infiltration in vivo [103,104]. Inflammation
induced by other stimulators such as zymosan, carrageenan,
and TNF-alpha was also inhibited by LLL
[32,105,106]. Growth factor stimulating activity of LLL
was demonstrated in both in vitro and in vivo experiments
in which augmentation of FGF-2, PDGF and IGF-1 was
observed [36,37,107]. Endogenous production of these
growth factors may be useful in regeneration based on
activation of endogenous pulmonary stem cells [108,109].
Another aspect of LLL activities of relevance is ability to
stimulate angiogenesis. In COPD, the constriction of
blood vessels as a result of poor oxygen uptake is results
in a feedback loop culminating in pulmonary hypertension.
Administration of angiogenic factors has been
demonstrated to be beneficial in several animal models of
pulmonary pathology [110,111]. The ability of LLL to
directly induce proliferation of HUVEC cells [112], as well
as to augment production of angiogenic factors such as
VEGF [113], supports the possibility of creation of an
environment hospitable to neoangiogenesis which is optimal
for stem cell growth. In fact, a study demonstrated in
vivo induction of neocapillary formation subsequent to
LLL administration in a hindlimb ischemia model [114].
The critical importance of angiogenesis in stem cell
mediated regeneration has previously been demonstrated
in the stroke model, where the major therapeutic activity
of exogenous stem cells has been attributed to angiogenic
as opposed to transdifferentiation effects [115].
Direct evidence of LLL stimulating stem cells has been
obtained using mesenchymal stem cells derived both
from the bone marrow and from the adipose tissue
[116,117]. Interestingly in vivo administration of LLL stimulated
MSC has resulted in 50% decrease in cardiac
infarct size [118]. Clinical translation of LLL has been
performed in the area of stroke, in which a 660 patient
trial demonstrated statistically significant effects in post
trial subset analysis [100].
Conclusions
Despite clinical use of LLL for decades, the field is still
in its infancy. As is obvious from the wide variety of
LLL sources, frequencies, and intensities used, no standard
protocols exist. The ability of LLL to induce
growth factor production, inhibition of inflammation,
stimulation of angiogenesis, and direct effects on stem
cells suggests the urgent need for combining this modality
with regenerative medicine, giving birth to the new
field of “regenerative photoceuticals”. Development of a
regenerative treatment for COPD as well as for other
degenerative diseases would be of considerable benefit.
Regarding COPD, such treatment would be life-saving/
life extending for thousands of affected individuals.
Ceasing smoking or not starting to smoke would considerably
impact this disease.
Acknowledgements
The authors thank Victoria Dardov and Matthew Gandjian for critical
discussions and input.
Author details
1Entest BioMedical, San Diego, CA, USA. 2Georgetown Dermatology,
Washington DC, USA. 3Cromos Pharma Services, Longview, WA, USA. 4Center
for the Study of Natural Oncology, Del Mar, CA, USA. 5Department of
Hematology and Medical Oncology, St Francis Hospital and Medical Center,
Hartford, CT, USA. 6Moores Cancer Center, University of California San Diego,
CA, USA. 7Department of Cardiothoracic Surgery, University of Utah, Salt
Lake City, UT, USA.
Authors’ contributions
FL, SFJ, DTA, FR, VB, VG, CAD, RDNC, ANP, EC, DRK contributed to literature
review, analysis and discussion, synthesis of concepts, writing of the
manuscript and proof-reading of the final draft.
Competing interests
David R Koos is a shareholder, as well as Chairman and CEO of Entest Bio.
Feng Lin is research director of Entest Bio. All other authors declare no
competing interest.
Received: 7 January 2010
Accepted: 16 February 2010 Published: 16 February 2010
References
1. Abdel-Latif A, Bolli R, Tleyjeh IM, Montori VM, Perin EC, Hornung CA, Zuba-
Surma EK, Al-Mallah M, Dawn B: Adult bone marrow-derived cells for
cardiac repair: a systematic review and meta-analysis. Arch Intern Med
2007, 167:989-997.
2. Martin-Rendon E, Brunskill SJ, Hyde CJ, Stanworth SJ, Mathur A, Watt SM:
Autologous bone marrow stem cells to treat acute myocardial infarction:
a systematic review. Eur Heart J 2008, 29:1807-1818.
3. Vet-Stem Regenerative Veterinary Medicine. http://www.vet-stem.com.
4. Riordan NH, Ichim TE, Min WP, Wang H, Solano F, Lara F, Alfaro M,
Rodriguez JP, Harman RJ, Patel AN, Murphy MP, Lee RR, Minev B: Nonexpanded
adipose stromal vascular fraction cell therapy for multiple
sclerosis. J Transl Med 2009, 7:29.
5. Motz GT, Eppert BL, Sun G, Wesselkamper SC, Linke MJ, Deka R,
Borchers MT: Persistence of lung CD8 T cell oligoclonal expansions upon
smoking cessation in a mouse model of cigarette smoke-induced
emphysema. J Immunol 2008, 181:8036-8043.
6. Maeno T, Houghton AM, Quintero PA, Grumelli S, Owen CA, Shapiro SD:
CD8+ T Cells are required for inflammation and destruction in cigarette
smoke-induced emphysema in mice. J Immunol 2007, 178:8090-8096.
7. Woodruff PG, Koth LL, Yang YH, Rodriguez MW, Favoreto S, Dolganov GM,
Paquet AC, Erle DJ: A distinctive alveolar macrophage activation state
induced by cigarette smoking. Am J Respir Crit Care Med 2005,
172:1383-1392.
8. Stefanska AM, Walsh PT: Chronic obstructive pulmonary disease: evidence
for an autoimmune component. Cell Mol Immunol 2009, 6:81-86.
9. Gonzalez-Rey E, Gonzalez MA, Varela N, O’Valle F, Hernandez-Cortes P,
Rico L, Buscher D, Delgado M: Human adipose-derived mesenchymal
stem cells reduce inflammatory and T cell responses and induce
regulatory T cells in vitro in rheumatoid arthritis. Ann Rheum Dis
69:241-248.
10. Lepelletier Y, Lecourt S, Arnulf B, Vanneaux V, Fermand JP, Menasche P,
Domet T, Marolleau JP, Hermine O, Larghero J: Galectin-1 and
Semaphorin-3A are two soluble factors conferring T cell
immunosuppression to bone marrow mesenchymal stem cell. Stem Cells
Dev 2009.
11. Tsyb AF, Petrov VN, Konoplyannikov AG, Saypina EV, Lepechina LA,
Kalsina S, Semenkova IV, Agaeva EV: In vitro inhibitory effect of
mesenchymal stem cells on zymosan-induced production of reactive
oxygen species. Bull Exp Biol Med 2008, 146:158-164.
12. Sun L, Akiyama K, Zhang H, Yamaza T, Hou Y, Zhao S, Xu T, Le A, Shi S:
Mesenchymal stem cell transplantation reverses multiorgan dysfunction
in systemic lupus erythematosus mice and humans. Stem Cells 2009,
27:1421-1432.
13. Feuerer M, Herrero L, Cipolletta D, Naaz A, Wong J, Nayer A, Lee J,
Goldfine AB, Benoist C, Shoelson S, Mathis D: Lean, but not obese, fat is
enriched for a unique population of regulatory T cells that affect
metabolic parameters. Nat Med 2009, 15:930-939.
14. Ogawa Y, Duru EA, Ameredes BT: Role of IL-10 in the resolution of airway
inflammation. Curr Mol Med 2008, 8:437-445.
15. Serrano-Mollar A, Nacher M, Gay-Jordi G, Closa D, Xaubet A, Bulbena O:
Intratracheal transplantation of alveolar type II cells reverses bleomycininduced
lung fibrosis. Am J Respir Crit Care Med 2007, 176:1261-1268.
16. Aslam M, Baveja R, Liang OD, Fernandez-Gonzalez A, Lee C, Mitsialis SA,
Kourembanas S: Bone marrow stromal cells attenuate lung injury in a
murine model of neonatal chronic lung disease. Am J Respir Crit Care Med
2009, 180:1122-1130.
17. van Haaften T, Byrne R, Bonnet S, Rochefort GY, Akabutu J, Bouchentouf M,
Rey-Parra GJ, Galipeau J, Haromy A, Eaton F, Chen M, Hashimoto K,
Abley D, Korbutt G, Archer SL, Thébaud B: Airway delivery of
mesenchymal stem cells prevents arrested alveolar growth in neonatal
lung injury in rats. Am J Respir Crit Care Med 2009, 180:1131-1142.
18. Zalewska-Kaszubska J, Obzejta D: Use of low-energy laser as adjunct
treatment of alcohol addiction. Lasers Med Sci 2004, 19:100-104.
19. Nikitin AV, Esaulenko IE, Shatalova OL: [Effectiveness of laser puncture in
elderly patients with bronchial asthma accompanied by chronic
rhinosinusitis]. Adv Gerontol 2008, 21:424-426.
20. Vasil’ev AP, Strel’tsova NN, Iu Senatorov N: [Laser irradiation in the
treatment of ischemic heart disease]. Vopr Kurortol Fizioter Lech Fiz Kult
2001, 10-13.
21. Maiman TH: Stimulated optical radiation in Ruby. Nature 187:493.
22. Roy D: Ablative facial resurfacing. Dermatol Clin 2005, 23:549-559, viii.
23. Brown MC: An evidence-based approach to patient selection for laser
vision correction. J Refract Surg 2009, 25:S661-667.
24. Brancaleon L, Moseley H: Laser and non-laser light sources for
photodynamic therapy. Lasers Med Sci 2002, 17:173-186.
25. Mester ESB, Tota JG: “Effect of laser on hair growth of mice”. Kiserl
Orvostud 1967, 19:628-631.
26. Mester E, Korenyi-Both A, Spiry T, Tisza S: The effect of laser irradiation on
the regeneration of muscle fibers (preliminary report). Z Exp Chir 1975,
8:258-262.
27. Posten W, Wrone DA, Dover JS, Arndt KA, Silapunt S, Alam M: Low-level
laser therapy for wound healing: mechanism and efficacy. Dermatol Surg
2005, 31:334-340.
28. Vladimirov YA, Osipov AN, Klebanov GI: Photobiological principles of
therapeutic applications of laser radiation. Biochemistry (Mosc) 2004,
69:81-90.
29. Hu WP, Wang JJ, Yu CL, Lan CC, Chen GS, Yu HS: Helium-neon laser
irradiation stimulates cell proliferation through photostimulatory effects
in mitochondria. J Invest Dermatol 2007, 127:2048-2057.
30. Moriyama Y, Nguyen J, Akens M, Moriyama EH, Lilge L: In vivo effects of
low level laser therapy on inducible nitric oxide synthase. Lasers Surg
Med 2009, 41:227-231.
31. Samoilova KA, Zhevago NA, Petrishchev NN, Zimin AA: Role of nitric oxide
in the visible light-induced rapid increase of human skin
microcirculation at the local and systemic levels: II. healthy volunteers.
Photomed Laser Surg 2008, 26:443-449.
32. Yamaura M, Yao M, Yaroslavsky I, Cohen R, Smotrich M, Kochevar IE: Low
level light effects on inflammatory cytokine production by rheumatoid
arthritis synoviocytes. Lasers Surg Med 2009, 41:282-290.
33. Shiba H, Tsuda H, Kajiya M, Fujita T, Takeda K, Hino T, Kawaguchi H,
Kurihara H: Neodymium-doped yttrium-aluminium-garnet laser
irradiation abolishes the increase in interleukin-6 levels caused by
peptidoglycan through the p38 mitogen-activated protein kinase
pathway in human pulp cells. J Endod 2009, 35:373-376.
34. de Lima Mafra F, Costa MS, Albertini R, Silva JA Jr, Aimbire F: Low level
laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-
adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat
bronchi segments in E. coli lipopolysaccharide-induced airway
inflammation by a NF-kappaB dependent mechanism. Lasers Surg Med
2009, 41:68-74.
35. Aimbire F, de Oliveira Ligeiro AP, Albertini R, Correa JC, de Campos
Ladeira CB, Lyon JP, Silva JA Jr, Costa MS: Low level laser therapy (LLLT)
decreases pulmonary microvascular leakage, neutrophil influx and IL-
1beta levels in airway and lung from rat subjected to LPS-induced
inflammation. Inflammation 2008, 31:189-197.
36. Safavi SM, Kazemi B, Esmaeili M, Fallah A, Modarresi A, Mir M: Effects of
low-level He-Ne laser irradiation on the gene expression of IL-1beta,
TNF-alpha, IFN-gamma, TGF-beta, bFGF, and PDGF in rat’s gingiva. Lasers
Med Sci 2008, 23:331-335.
37. Saygun I, Karacay S, Serdar M, Ural AU, Sencimen M, Kurtis B: Effects of
laser irradiation on the release of basic fibroblast growth factor (bFGF),
insulin like growth factor-1 (IGF-1), and receptor of IGF-1 (IGFBP3) from
gingival fibroblasts. Lasers Med Sci 2008, 23:211-215.
38. Schwartz F, Brodie C, Appel E, Kazimirsky G, Shainberg A: Effect of helium/
neon laser irradiation on nerve growth factor synthesis and secretion in
skeletal muscle cultures. J Photochem Photobiol B 2002, 66:195-200.
39. Yu W, Naim JO, Lanzafame RJ: The effect of laser irradiation on the
release of bFGF from 3T3 fibroblasts. Photochem Photobiol 1994,
59:167-170.
40. Zungu IL, Hawkins Evans D, Abrahamse H: Mitochondrial responses of
normal and injured human skin fibroblasts following low level laser
irradiation–an in vitro study. Photochem Photobiol 2009, 85:987-996.
41. Wu S, Xing D, Gao X, Chen WR: High fluence low-power laser irradiation
induces mitochondrial permeability transition mediated by reactive
oxygen species. J Cell Physiol 2009, 218:603-611.
42. Lan CC, Wu CS, Chiou MH, Chiang TY, Yu HS: Low-energy helium-neon
laser induces melanocyte proliferation via interaction with type IV
collagen: visible light as a therapeutic option for vitiligo. Br J Dermatol
2009, 161:273-280.
43. Karu T: Photobiology of low-power laser effects. Health Phys 1989,
56:691-704.
44. Tiphlova O, Karu T: Role of primary photoacceptors in low-power laser
effects: action of He-Ne laser radiation on bacteriophage T4-Escherichia
coli interaction. Lasers Surg Med 1989, 9:67-69.
45. Karu TI: Mitochondrial signaling in mammalian cells activated by red and
near-IR radiation. Photochem Photobiol 2008, 84:1091-1099.
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
46. Zhang L, Xing D, Zhu D, Chen Q: Low-power laser irradiation inhibiting
Abeta25-35-induced PC12 cell apoptosis via PKC activation. Cell Physiol
Biochem 2008, 22:215-222.
47. Aimbire F, Santos FV, Albertini R, Castro-Faria-Neto HC, Mittmann J,
Pacheco-Soares C: Low-level laser therapy decreases levels of lung
neutrophils anti-apoptotic factors by a NF-kappaB dependent
mechanism. Int Immunopharmacol 2008, 8:603-605.
48. Lipovsky A, Nitzan Y, Lubart R: A possible mechanism for visible lightinduced
wound healing. Lasers Surg Med 2008, 40:509-514.
49. Ignatieva N, Zakharkina O, Andreeva I, Sobol E, Kamensky V, Lunin V: Effects
of laser irradiation on collagen organization in chemically induced
degenerative annulus fibrosus of lumbar intervertebral disc. Lasers Surg
Med 2008, 40:422-432.
50. Silveira LB, Prates RA, Novelli MD, Marigo HA, Garrocho AA, Amorim JC,
Sousa GR, Pinotti M, Ribeiro MS: Investigation of mast cells in human
gingiva following low-intensity laser irradiation. Photomed Laser Surg
2008, 26:315-321.
51. Coombe AR, Ho CT, Darendeliler MA, Hunter N, Philips JR, Chapple CC,
Yum LW: The effects of low level laser irradiation on osteoblastic cells.
Clin Orthod Res 2001, 4:3-14.
52. Gregoraszczuk E, Dobrowolski JW, Galas J: Effect of low intensity laser
beam on steroid dehydrogenase activity and steroid hormone
production in cultured porcine granulosa cells. Folia Histochem Cytochem
(Krakow) 1983, 21:87-92.
53. Yu HS, Chang KL, Yu CL, Chen JW, Chen GS: Low-energy helium-neon
laser irradiation stimulates interleukin-1 alpha and interleukin-8 release
from cultured human keratinocytes. J Invest Dermatol 1996, 107:593-596.
54. Mvula B, Mathope T, Moore T, Abrahamse H: The effect of low level laser
irradiation on adult human adipose derived stem cells. Lasers Med Sci
2008, 23:277-282.
55. Mvula B, Moore TJ, Abrahamse H: Effect of low-level laser irradiation and
epidermal growth factor on adult human adipose-derived stem cells.
Lasers Med Sci 25:33-39.
56. Hawkins DH, Abrahamse H: The role of laser fluence in cell viability,
proliferation, and membrane integrity of wounded human skin
fibroblasts following helium-neon laser irradiation. Lasers Surg Med 2006,
38:74-83.
57. Houreld N, Abrahamse H: In vitro exposure of wounded diabetic
fibroblast cells to a helium-neon laser at 5 and 16 J/cm2. Photomed Laser
Surg 2007, 25:78-84.
58. Smol’yaninova NK, Karu TI, Fedoseeva GE, Zelenin AV: Effects of He-Ne
laser irradiation on chromatin properties and synthesis of nucleic acids
in human peripheral blood lymphocytes. Biomed Sci 1991, 2:121-126.
59. Funk JO, Kruse A, Neustock P, Kirchner H: Helium-neon laser irradiation
induces effects on cytokine production at the protein and the mRNA
level. Exp Dermatol 1993, 2:75-83.
60. Funk JO, Kruse A, Kirchner H: Cytokine production after helium-neon laser
irradiation in cultures of human peripheral blood mononuclear cells. J
Photochem Photobiol B 1992, 16:347-355.
61. Gulsoy M, Ozer GH, Bozkulak O, Tabakoglu HO, Aktas E, Deniz G, Ertan C:
The biological effects of 632.8-nm low energy He-Ne laser on peripheral
blood mononuclear cells in vitro. J Photochem Photobiol B 2006,
82:199-202.
62. Novoselova EG, Cherenkov DA, Glushkova OV, Novoselova TV,
Chudnovskii VM, Iusupov VI, Fesenko EE: [Effect of low-intensity laser
radiation (632.8 nm) on immune cells isolated from mice]. Biofizika 2006,
51:509-518.
63. Dube A, Bansal H, Gupta PK: Modulation of macrophage structure and
function by low level He-Ne laser irradiation. Photochem Photobiol Sci
2003, 2:851-855.
64. Hemvani N, Chitnis DS, Bhagwanani NS: Helium-neon and nitrogen laser
irradiation accelerates the phagocytic activity of human monocytes.
Photomed Laser Surg 2005, 23:571-574.
65. Moustsen PA, Vinter N, Aas-Andersen L, Kragstrup J: [Laser treatment of
sinusitis in general practice assessed by a double-blind controlled
study]. Ugeskr Laeger 1991, 153:2232-2234.
66. Shen X, Zhao L, Ding G, Tan M, Gao J, Wang L, Lao L: Effect of combined
laser acupuncture on knee osteoarthritis: a pilot study. Lasers Med Sci
2009, 24:129-136.
67. Ekim A, Armagan O, Tascioglu F, Oner C, Colak M: Effect of low level laser
therapy in rheumatoid arthritis patients with carpal tunnel syndrome.
Swiss Med Wkly 2007, 137:347-352.
68. Minatel DG, Frade MA, Franca SC, Enwemeka CS: Phototherapy promotes
healing of chronic diabetic leg ulcers that failed to respond to other
therapies. Lasers Surg Med 2009, 41:433-441.
69. Bocci V, Travagli V, Zanardi I: May oxygen-ozone therapy improves
cardiovascular disorders?. Cardiovasc Hematol Disord Drug Targets 2009,
9:78-85.
70. Bocci V, Borrelli E, Travagli V, Zanardi I: The ozone paradox: ozone is a
strong oxidant as well as a medical drug. Med Res Rev 2009, 29:646-682.
71. Re L, Mawsouf MN, Menendez S, Leon OS, Sanchez GM, Hernandez F:
Ozone therapy: clinical and basic evidence of its therapeutic potential.
Arch Med Res 2008, 39:17-26.
72. Damera G, Zhao H, Wang M, Smith M, Kirby C, Jester WF, Lawson JA,
Panettieri RA Jr: Ozone modulates IL-6 secretion in human airway
epithelial and smooth muscle cells. Am J Physiol Lung Cell Mol Physiol
2009, 296:L674-683.
73. Manzer R, Dinarello CA, McConville G, Mason RJ: Ozone exposure of
macrophages induces an alveolar epithelial chemokine response
through IL-1alpha. Am J Respir Cell Mol Biol 2008, 38:318-323.
74. McDonald RJ, Usachencko J: Neutrophils injure bronchial epithelium after
ozone exposure. Inflammation 1999, 23:63-73.
75. Rodriguez ZZ, Guanche D, Alvarez RG, Rosales FH, Alonso Y, Schulz S:
Preconditioning with ozone/oxygen mixture induces reversion of some
indicators of oxidative stress and prevents organic damage in rats with
fecal peritonitis. Inflamm Res 2009.
76. Zamora ZB, Borrego A, Lopez OY, Delgado R, Gonzalez R, Menendez S,
Hernandez F, Schulz S: Effects of ozone oxidative preconditioning on
TNF-alpha release and antioxidant-prooxidant intracellular balance in
mice during endotoxic shock. Mediators Inflamm 2005, 2005:16-22.
77. Borrego A, Zamora ZB, Gonzalez R, Romay C, Menendez S, Hernandez F,
Montero T, Rojas E: Protection by ozone preconditioning is mediated by
the antioxidant system in cisplatin-induced nephrotoxicity in rats.
Mediators Inflamm 2004, 13:13-19.
78. Martinez-Sanchez G, Al-Dalain SM, Menendez S, Re L, Giuliani A, Candelario-
Jalil E, Alvarez H, Fernandez-Montequin JI, Leon OS: Therapeutic efficacy of
ozone in patients with diabetic foot. Eur J Pharmacol 2005, 523:151-161.
79. Surinchak JS, Alago ML, Bellamy RF, Stuck BE, Belkin M: Effects of low-level
energy lasers on the healing of full-thickness skin defects. Lasers Surg
Med 1983, 2:267-274.
80. Broadley C, Broadley KN, Disimone G, Riensch L, Davidson JM: Low-energy
helium-neon laser irradiation and the tensile strength of incisional
wounds in the rat. Wound Repair Regen 1995, 3:512-517.
81. Ghamsari SM, Taguchi K, Abe N, Acorda JA, Yamada H: Histopathological
effect of low-level laser therapy on sutured wounds of the teat in dairy
cattle. Vet Q 1996, 18:17-21.
82. Ghamsari SM, Taguchi K, Abe N, Acorda JA, Sato M, Yamada H: Evaluation
of low level laser therapy on primary healing of experimentally induced
full thickness teat wounds in dairy cattle. Vet Surg 1997, 26:114-120.
83. Pinfildi CE, Liebano RE, Hochman BS, Ferreira LM: Helium-neon laser in
viability of random skin flap in rats. Lasers Surg Med 2005, 37:74-77.
84. Maiya GA, Kumar P, Rao L: Effect of low intensity helium-neon (He-Ne)
laser irradiation on diabetic wound healing dynamics. Photomed Laser
Surg 2005, 23:187-190.
85. Bayat M, Vasheghani MM, Razavi N, Taheri S, Rakhshan M: Effect of lowlevel
laser therapy on the healing of second-degree burns in rats: a
histological and microbiological study. J Photochem Photobiol B 2005,
78:171-177.
86. Garavello I, Baranauskas V, da Cruz-Hofling MA: The effects of low laser
irradiation on angiogenesis in injured rat tibiae. Histol Histopathol 2004,
19:43-48.
87. Lin YS, Huang MH, Chai CY, Yang RC: Effects of helium-neon laser on
levels of stress protein and arthritic histopathology in experimental
osteoarthritis. Am J Phys Med Rehabil 2004, 83:758-765.
88. Jamieson RW, Friend PJ: Organ reperfusion and preservation. Front Biosci
2008, 13:221-235.
89. Akai M, Usuba M, Maeshima T, Shirasaki Y, Yasuoka S: Laser’s effect on
bone and cartilage change induced by joint immobilization: an
experiment with animal model. Lasers Surg Med 1997, 21:480-484.
Lin et al. Journal of Translational Medicine 2010, 8:16
http://www.translational-medicine.com/content/8/1/16
90. Nakano J, Kataoka H, Sakamoto J, Origuchi T, Okita M, Yoshimura T: Lowlevel
laser irradiation promotes the recovery of atrophied gastrocnemius
skeletal muscle in rats. Exp Physiol 2009, 94:1005-1015.
91. Doin-Silva R, Baranauskas V, Rodrigues-Simioni L, da Cruz-Hofling MA: The
ability of low level laser therapy to prevent muscle tissue damage
induced by snake venom. Photochem Photobiol 2009, 85:63-69.
92. Gal D, Chokshi SK, Mosseri M, Clarke RH, Isner JM: Percutaneous delivery of
low-level laser energy reverses histamine-induced spasm in
atherosclerotic Yucatan microswine. Circulation 1992, 85:756-768.
93. Kipshidze N, Sahota H, Komorowski R, Nikolaychik V, Keelan MH Jr:
Photoremodeling of arterial wall reduces restenosis after balloon
angioplasty in an atherosclerotic rabbit model. J Am Coll Cardiol 1998,
31:1152-1157.
94. Novoselova EG, Glushkova OV, Cherenkov DA, Chudnovsky VM, Fesenko EE:
Effects of low-power laser radiation on mice immunity. Photodermatol
Photoimmunol Photomed 2006, 22:33-38.
95. Bensadoun RJ, Franquin JC, Ciais G, Darcourt V, Schubert MM, Viot M,
Dejou J, Tardieu C, Benezery K, Nguyen TD, Laudoyer Y, Dassonville O,
Poissonnet G, Vallicioni J, Thyss A, Hamdi M, Chauvel P, Demard F: Lowenergy
He/Ne laser in the prevention of radiation-induced mucositis. A
multicenter phase III randomized study in patients with head and neck
cancer. Support Care Cancer 1999, 7:244-252.
96. Arun Maiya G, Sagar MS, Fernandes D: Effect of low level helium-neon
(He-Ne) laser therapy in the prevention & treatment of radiation
induced mucositis in head & neck cancer patients. Indian J Med Res 2006,
124:399-402.
97. Neiburger EJ: Rapid healing of gingival incisions by the helium-neon
diode laser. J Mass Dent Soc 1999, 48:8-13, 40..
98. Aras MH, Gungormus M: Placebo-controlled randomized clinical trial of
the effect two different low-level laser therapies (LLLT)-intraoral and
extraoral-on trismus and facial swelling following surgical extraction of
the lower third molar. Lasers Med Sci 2009.
99. Zycinski P, Krzeminska-Pakula M, Peszynski-Drews C, Kierus A, Trzos E,
Rechcinski T, Figiel L, Kurpesa M, Plewka M, Chrzanowski L, Drozdz J: Laser
biostimulation in end-stage multivessel coronary artery disease–a
preliminary observational study. Kardiol Pol 2007, 65:13-21, discussion 22-
13..
100. Zivin JA, Albers GW, Bornstein N, Chippendale T, Dahlof B, Devlin T,
Fisher M, Hacke W, Holt W, Ilic S, Kasner S, Lew R, Nash M, Perez J,
Rymer M, Schellinger P, Schneider D, Schwab S, Veltkamp R, Walker M,
Streeter J, NeuroThera Effectiveness and Safety Trial-2 Investigators:
Effectiveness and safety of transcranial laser therapy for acute ischemic
stroke. Stroke 2009, 40:1359-1364.
101. Osiris Therapeutics Reports Interim Data for COPD Trial. http://www.
medicalnewstoday.com/articles/155267.php.
102. Gavish L, Perez LS, Reissman P, Gertz SD: Irradiation with 780 nm diode
laser attenuates inflammatory cytokines but upregulates nitric oxide in
lipopolysaccharide-stimulated macrophages: implications for the
prevention of aneurysm progression. Lasers Surg Med 2008, 40:371-378.
103. Correa F, Lopes Martins RA, Correa JC, Iversen VV, Joenson J, Bjordal JM:
Low-level laser therapy (GaAs lambda = 904 nm) reduces inflammatory
cell migration in mice with lipopolysaccharide-induced peritonitis.
Photomed Laser Surg 2007, 25:245-249.
104. Aimbire F, Lopes-Martins RA, Castro-Faria-Neto HC, Albertini R,
Chavantes MC, Pacheco MT, Leonardo PS, Iversen VV, Bjordal JM: Low-level
laser therapy can reduce lipopolysaccharide-induced contractile force
dysfunction and TNF-alpha levels in rat diaphragm muscle. Lasers Med
Sci 2006, 21:238-244.
105. de Morais NC, Barbosa AM, Vale ML, Villaverde AB, de Lima CJ, Cogo JC,
Zamuner SR: Anti-Inflammatory Effect of Low-Level Laser and Light-
Emitting Diode in Zymosan-Induced Arthritis. Photomed Laser Surg 2009.
106. Boschi ES, Leite CE, Saciura VC, Caberlon E, Lunardelli A, Bitencourt S,
Melo DA, Oliveira JR: Anti-Inflammatory effects of low-level laser therapy
(660 nm) in the early phase in carrageenan-induced pleurisy in rat.
Lasers Surg Med 2008, 40:500-508.
107. Shimizu N, Mayahara K, Kiyosaki T, Yamaguchi A, Ozawa Y, Abiko Y: Lowintensity
laser irradiation stimulates bone nodule formation via insulinlike
growth factor-I expression in rat calvarial cells. Lasers Surg Med 2007,
39:551-559.
108. Hackett TL, Shaheen F, Johnson A, Wadsworth S, Pechkovsky DV,
Jacoby DB, Kicic A, Stick SM, Knight DA: Characterization of side
population cells from human airway epithelium. Stem Cells 2008,
26:2576-2585.
109. Irwin D, Helm K, Campbell N, Imamura M, Fagan K, Harral J, Carr M,
Young KA, Klemm D, Gebb S, Dempsey EC, West J, Majka S: Neonatal lung
side population cells demonstrate endothelial potential and are altered
in response to hyperoxia-induced lung simplification. Am J Physiol Lung
Cell Mol Physiol 2007, 293:L941-951.
110. Thebaud B, Ladha F, Michelakis ED, Sawicka M, Thurston G, Eaton F,
Hashimoto K, Harry G, Haromy A, Korbutt G, Archer SL: Vascular
endothelial growth factor gene therapy increases survival, promotes
lung angiogenesis, and prevents alveolar damage in hyperoxia-induced
lung injury: evidence that angiogenesis participates in alveolarization.
Circulation 2005, 112:2477-2486.
111. Thebaud B: Angiogenesis in lung development, injury and repair:
implications for chronic lung disease of prematurity. Neonatology 2007,
91:291-297.
112. Schindl A, Merwald H, Schindl L, Kaun C, Wojta J: Direct stimulatory effect
of low-intensity 670 nm laser irradiation on human endothelial cell
proliferation. Br J Dermatol 2003, 148:334-336.
113. Kipshidze N, Nikolaychik V, Keelan MH, Shankar LR, Khanna A, Kornowski R,
Leon M, Moses J: Low-power helium: neon laser irradiation enhances
production of vascular endothelial growth factor and promotes growth
of endothelial cells in vitro. Lasers Surg Med 2001, 28:355-364.
114. Ihsan FR: Low-level laser therapy accelerates collateral circulation and
enhances microcirculation. Photomed Laser Surg 2005, 23:289-294.
115. Taguchi A, Soma T, Tanaka H, Kanda T, Nishimura H, Yoshikawa H,
Tsukamoto Y, Iso H, Fujimori Y, Stern DM, Naritomi H, Matsuyama T:
Administration of CD34+ cells after stroke enhances neurogenesis via
angiogenesis in a mouse model. J Clin Invest 2004, 114:330-338.
116. Li WT, Leu YC: Effects of low level red-light irradiation on the
proliferation of mesenchymal stem cells derived from rat bone marrow.
Conf Proc IEEE Eng Med Biol Soc 2007, 2007:5830-5833.
117. Tuby H, Maltz L, Oron U: Low-level laser irradiation (LLLI) promotes
proliferation of mesenchymal and cardiac stem cells in culture. Lasers
Surg Med 2007, 39:373-378.
118. Tuby H, Maltz L, Oron U: Implantation of low-level laser irradiated
mesenchymal stem cells into the infarcted rat heart is associated with
reduction in infarct size and enhanced angiogenesis. Photomed Laser
Surg 2009, 27:227-233.
119. Agaiby AD, Ghali LR, Wilson R, Dyson M: Laser modulation of angiogenic
factor production by T-lymphocytes. Lasers Surg Med 2000, 26:357-363.
120. Zhang H, Hou JF, Shen Y, Wang W, Wei YJ, Hu S: Low Level Laser
Irradiation Precondition to Create Friendly Milieu of Infarcted
Myocardium and Enhance Early Survival of Transplanted Bone Marrow
Cells. J Cell Mol Med 2009.
121. Hou JF, Zhang H, Yuan X, Li J, Wei YJ, Hu SS: In vitro effects of low-level
laser irradiation for bone marrow mesenchymal stem cells: proliferation,
growth factors secretion and myogenic differentiation. Lasers Surg Med
2008, 40:726-733.
doi:10.1186/1479-5876-8-16
Cite this article as: Lin et al.: Lasers, stem cells, and COPD. Journal of
Translational Medicine 2010 8:16.
Original Source: http://www.translational-medicine.com/content/8/1/16
Abstract
Regenerative capacity following injury or an ischemic event is confined to non mammalian vertebrates. Mammals have a limited capacity to restore organs
following injury to organs like the liver and skeletal muscles but practically no ability to regenerate organs like the heart or brain following an ischemic event
or injury. We tried a new approach in cell based therapy to improve regeneration in various organs following ischemic injury. Low-level laser therapy (LLLT)
which has photobiostimulating effects on cells was delivered to autologous bone marrow (BM) that is enriched with stem cells and various progenitor cells, in
order to induce the cells in the BM for the benefit of the injured /ischemic organs. In a model of induced myocardial infarction (MI) in rats laser application
to the BM caused a marked and significant decrease (79%) in infarct size (scarring) 3 weeks post-MI. It was also found that a significantly higher density of
c-kit positive cells (a marker of mesenchymal stem cells) in the myocardium of laser-treated rats relative to non-treated rat’s post-MI. The novel approach
presented in this study, of the use of stem cells for cell therapy to the infracted heart, avoids the need to isolate millions of stem cells, to grow them in vitro and
to inject them back into the patient. In the same line of rationale we tried to find whether LLLT to the BM could be beneficial also to kidney impairment after
ischemic reperfusion injury (IRI) to the rat kidney. C-kit positive cell density in kidneys post-IRI and laser-treatment was significantly (p=0.05) 2.4-fold higher
compared to the non laser treated group. Creatinine, blood urea nitrogen, and cystatin-C levels were significantly lower in the laser-treated rats as compared
to non-treated ones. The effect of LLLT delivery to BM was also tested on Alzheimer’s disease (AD) mice in their late stage of the disease. Mice were given
multiple (every 10 days) LLLT to BM from age 4 to 6 months. It was found that in the treated AD-mice neurological tests (Fear and Cognitive tests) revealed
a significantly (p<0.05) better neurological performance and cognitive capacity compared to the non-treated AD mice. Furthermore, concomitantly with the
improved neurological performance, ß-amyloid density in the hippocampal region of the brains was revealed to be significantly less in the laser-treated mice
as compared to control. In conclusion, a novel approach, of applying LLLT to autologeous BM in order to induce stem cells that are consequently recruited to the
injured/ischemic organ leading to a marked beneficial effect post-ischemic event or degenerative process is presented. This approach is novel in the respect
that it is stimulating the patient’s own abilities to initiate a regenerative response in an organ by the utilization of light. The possibility that this approach can
also be applied to other ischemic/injured organs or organs undergoing degenerative processes (i.e. neurodegenerative diseases), with consequent beneficial
effects, cannot be ruled out
CELL THERAPY FOR INJURED/ISCHEMIC ORGANS
Clinical trials have lately been implemented in a growing
abundance due to the extensive research and new approaches of
cell based therapies for the reconstruction of impaired organs.
Regenerative capacity following injury or an ischemic event
is confined to non mammalian vertebrates. In particular, fish
and primitive amphibians can regenerate organs like the heart,
brain and limbs. However, mammals have a limited capacity
to restore organs following injury to organs like the liver and
skeletal muscles but practically no ability to regenerate organs
like the heart or brain following an ischemic event or injury.
The mammalian heart, including the human heart, for example,
has a very limited capacity to regenerate following damage or
an acute ischemic event like myocardial infarction (MI). This is
due to the very low level of cardiomyocyte proliferation and the
limited number of cells expressing stem-cell marker proteins.
Stem-cell-based therapy was suggested as a potential solution
to the above situation. In recent years, cell-based therapy for
cardiac repair in particular has undergone a rapid transition
from basic science research to clinical reality [1-3]. The general
outcome of the clinical trials was that the procedures and longterm
outcome post-stem-cell implantation to the heart via the
coronary arteries are safe. However, improvement in long-term
functional performance of the heart was either not achieved or
was marginal [1-3].
There are several central issues pertaining to the use of cell
implantation in stem-cell therapy: the number of implanted stem
cells has to be high since there is massive cell death following
implantation or injection of cells into the heart or the blood
circulation. Another central issue in stem-cell implantation for
organ repair is the creation of a receptive cell environment in the
ischemic organ. Several factors (e.g. inhibition of inflammation
and apoptosis, secretion of cell growth factors etc.) are necessary
for optimal cell implantation [4]. The injected cells may have to
migrate from the circulating blood to the ischemic niche. They
can then remain active and secrete growth factors, exerting a paracrine effect on the ischemic tissue [5]. Alternatively, they
may stimulate the small population of stem cells in the ischemic
organ (such as the heart), to proliferate and differentiate so as to
enhance cardiac repair post-MI [6]. Another issue is the timing
of injection of the stem cells to the infarcted heart and effect of
MI (inflammatory phase) on the BM [7]. Photobiostimulation
of cells in the bone marrow (BM), that is enriched with various
progenitor cells, by low level laser therapy (LLLT) may suggest a
new approach that may overcome some of the above limitation.
This new approach will be discussed in the present mini review
[8-11].
LOW LEVEL LASER THERAPY FOR THE ISCHEMIC
HEART
In general LLLT has been found to modulate various biological
processes, such as increasing mitochondrial respiration and ATP
synthesis, facilitating wound healing, and promoting the process
of skeletal muscle regeneration and angiogenesis [12,13]. It
was previously shown that LLLT can enhance skeletal muscle
regeneration following partial excision in the rat hind limb
muscles when the laser was delivered directly to the injured
organ multiple times (for 2 min each time) following injury [14].
This phenomenon was even more prominent following cold
injury to the frog skeletal muscles indicating that enhancement
of regeneration by LLLT is probably a general phenomenon in
vertebrates and maybe more effective in cold blooded animals
which innately have a lower metabolic rate in their cells [13-15].
In an experimental model of the infarcted heart in rats and dogs, it
was demonstrated that LLLT (Diode –Ga-Al-As 810nm at a power
density of 5 mW/cm2 for 120sec duration of laser exposure
comprising 0.6 J/cm2), application directly to the infarcted area
in the heart at optimal power parameters significantly reduces
infarct size (scar tissue formation) [16,17]. This phenomenon
was partially attributed to a significant elevation in ATP content,
heat shock proteins, vascular endothelial growth factor (VEGF),
and angiogenesis in the ischemic zone of the laser-irradiated
rats, as compared to non-irradiated rats [16,17]. The mechanism
associated with the photobiostimulation by LLLT is not yet
clearly understood [12]. There is evidence that cytochrome c
oxidase and perhaps also plasma membranes in cells function as
photoacceptors of the photons, and thereafter a cascade of events
occur in the mitochondria, leading to effects on various processes
like ATP production, up-regulation of VEGF, etc [12].
The effect of photobiostimulation on stem cells or progenitor
cells has not been extensively studied [18-21]. It was previously
shown that laser application (Diode laser at 50mW/cm2 for
100sec, energy density 0.5 mW/cm2) to the mesenchymal stem
cells isolated from bone marrow or cardiac stem cells causes a
significant increase in their proliferation in vitro [20]. Based on
previous studies that showed an increase in cytoprotective effect
on the ischemic heart following LLLT, a new approach was taken
to apply laser irradiation to stem cells grown in culture prior
to their implantation to the infarcted heart as a cell therapy for
heart repair [21]. In that study it was demonstrated that MSCs
that were laser treated prior to their implantation to the rat
infarcted heart caused a significant reduction in infarct size
as compared to MSCs that were injected to the heart without
prior laser treatment. This phenomenon was also associated
with significant elevation of vascular endothelial growth factor
(VEGF) in the myocardium of the rats that received the lasertreated
MSCs. In a recent study [8] the possibility of recruiting
autologous stem cells stimulated by LLLT in the BM to the
infarcted heart was addressed. The rationale behind the attempt
to use LLLT to induce the “crude” BM in the bone was, and still is,
that one cannot significantly affect the complex process post-MI
or ischemic injury to the kidney with a single type of stem cell. The
native BM is known for its many types and subtypes of stem cells,
which are defined by their reactivity to various antibodies. The
BM also contains many progenitor cells (i.e. monocytes) that can
further differentiate, for example to macrophages. Macrophages
have been shown recently to have a crucial role in the scarring
process post-MI. Thus LLLT may induce concomitantly in the BM
various types of cells that will increase in number in the blood
circulation following their enhanced proliferation in the BM.
These cells will probably, eventually, and to a certain extent and
under certain circumstances, home in on the ischemic zone in the
ischemic organ (heart, kidney etc.). In this study [8] it was found
that when LLLT was applied in vivo to the BM, and MSCs were
isolated from that BM 3 and 6 weeks later and grown in vitro, they
grew at a higher rate of proliferation relative to MSCs isolated
from non-laser-treated BM. This indicated that the MSCs when
in the BM, following LLLT application in vivo can be induced to
proliferate to a higher rate than non-treated MSCs. Furthermore,
laser application (Diode laser 808nm at power density of 10mW/
cm2 for 100 sec comprising 1J/cm2 energy density) to the BM (at
about 20 min post-MI) caused a marked and significant decrease
(79%) in infarct size 3 weeks post-MI. This extent of infarct size
reduction was even more effective in reducing scarring than that
of laser application directly to the infarcted heart, as also found
in previous studies with infarcted rat and dog hearts [10]. Even
when laser was applied 4 hours post-MI to the BM of infarcted
rats, a marked and significant reduction in the infarcted area
was observed in the laser-treated rats compared to control.
We also found a significantly higher density of c-kit+ (a marker
of MSCs) cells in the myocardium of laser-treated rats relative
to non-treated rat’s post-MI. Moreover, it was demonstrated in
this study that c-kit+ cells post-laser application to the BM of MIinduced
rats, homed specifically in on the infarcted heart and
not on uninjured organs (i.e. liver, kidney) in the same rat [16].
It can be hypothesized that the increased number of c-kit+ cells
found in the myocardium came from proliferating MSCs in the
BM that had migrated to the circulating blood and homed onto
the infarcted heart. Another finding of this study was that of
the preferred homing of the recruited or endogenous c-kit+ cells
in on the infarcted area, rather than their random deposition
throughout the left ventricle in the heart. Indeed, at 3-weeks
post-MI the density of c-kit+ cells in the infarcted area was 27-
fold higher in the rats whose BM had been treated with LLLT as
compared to control rats. Similarly, Hatzistergos et al. [6] found
that endogenous c-kit+ cardiac stem cells increased by 20-fold
in the porcine infarcted heart as compared to control following
Home Search Introduction
Ken Teegardin - (Website) 4361This tool is a searchable collection of technical publications, books, videos and other resources about the use of lasers and light for PhotoBioModulation (PBM). Enter a keyword above or see some of our favorite queries below.
Here are some of our favorite queries:
- Information for Beginners
- Best wavelength for PBM (980nm vs. 810nm vs 660nm...)
- Compare Lasers and LEDs
- Pulsing versus Continuous Wave (CW)
- Dosage: The best practices for dosage
- How NOT to promote laser therapy (by Turner and Hode)
- Great summary of positive double blind studies
- Whole Body Systems
- See all videos
- See all books about laser therapy and cold lasers
- Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing Athletic Performance
- Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Intro to laser acupuncture
- Traumatic brain injury research
- Soft Tissue
- Autoimmune
- Lyme's Disease
- Parkinson's research
- Alzheimer's research
- Hashimoto's research
- Covid
- Contraindications
- LLLT and Cancer
All the resources include links to the original source so we are not making any statement about the use of lasers for treating non-FDA cleared application, we are simple summarizing what others have said.
Where every possible, we have included a link to the orginal publication.
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- Where ever possible, the detailed section about the resource will link to the sources.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic lasers. It does include some resources on weight loss and smoking cessation.
The results of the search are sorted based on 3 quality factors on a scale of 1 to 10 with 10 being the best score. Originally all the resources were given a 5-5-5 until they could be individually evaluated. These scores are purely opinion and are only used to simplify the rank of the results from more valuable to least valuable. This should not be considered a critique of any work. This system was created to help researchers (including ourselves) find the most usable resources for any cold laser therapy research. The resources are assigned values based on the following 3 factors:
- Efficacy: The resource (especially research papers) should show a significant improvement in the condition being treated. Resources that show better results are given a higher quality score.
- Detail: The source must give enough information that the results can be duplicated. If a resource lacks too many details that it cannot be recreated, it is given a lower detail score.
- Lack of Bias: Many resources are created to try and show that one device is superior to its competition. Many manufacturers have staff that crank out biased papers on a regular basis on the hope that this will make their product look superior. If the author of the resource is paid by a manufacturer of the resource appears to be biased towards one device and not one technology, the resource has much less value.
Over the past few years of working with research, we found that a majority of the published resources are lacking in one of these three ranking factors.
The original goal of this research tool was to tie published resources to the protocols in the laser-therapy.us library. This connection allows users to trace each protocol back to a list of resources so the protocol can be researched and improved.
General Comments
POWER
When many of the first research papers were published, the most power laser available for therapy were less than 100mW and many systems had to be pulsed to keep the laser from burning out too quickly. Today, system are available that will deliver up to 60,000mW of continuous output. Because of these power limitation, many early studies were limited to extremely low dosages by today’s standards. It takes a 50mW system 17 minutes to deliver 50 joules at the surface of the skin. If this was spread over a large area of damage or was treating a deeper problem, the actual dosages were much less than 1J/cm2. Today, we know that these dosages typically produce very little or no results.
WAVELENGTH
About 80% of the resources in this database are in the near infrared wavelength. There is also some interest in the red wavelength (600 to 660nm) . Other wavelengths like blue, purple, and green have very little scientific research behind them and have not gotten much traction in the core therapy market with the exception of some fringe consumer products.
Legal Disclaimer
This research tool is free to use but we make no claims about the accuracy of the information. It is an aggregation of existing published resources and it is up to the user to determine if the source of the resources has any value. The information provided through this web site should not be used for diagnosing or treating a health problem or disease. If you have or suspect you may have a health problem, you should consult your local health care provider.
Biophtonica Introduction
Biophotonica - (Website) 4523Welcome to the BioPhotonica Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
- Fertility and Reproductive Health
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only PBM related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
Light House Health Introduction
LightHouse - (Website) 4515Welcome to the Lighthouse Health Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
The query result(s) can be shared using the following direct link. Anyone who clicks on this link in an email or on a web site will be shown the current results for the query.
https://www.laser-therapy.us/research/index.cfm?researchinput=stemcell