Start New Query
Laser-induced regeneration of cartilage
Emil N. Sobol, Olga I. Baum, Anatoliy B. Shekhter, Anna Guller, Andrey V. Baskov - J. of Biomedical Optics, 16(8), 080902 (2011) (Publication) 4501
This article shows results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine
View Resource
Abstract
Laser radiation provides a means to control the fields of temperature and thermo mechanical stress, mass transfer, and modification of fine structure of the cartilage matrix. The aim of this outlook paper is to review physical and biological aspects of laser-induced regeneration of cartilage and to discuss the possibilities and prospects of its clinical applications. The problems and the pathways of tissue regeneration, the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches for cartilage repair; consider possible mechanisms of laser-induced regeneration. Finally, we present the results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine.
1.
Introduction
Cartilage is a kind of highly specialized connective tissue. The structural variety of the cartilage provides its unique biomechanical capacity to bear different kinds of static and dynamic loads over a wide range of intensity. Biological role of cartilage structures stems from their critical significance for growth and development as well as for all kinds of body movements. The exceptional importance of cartilage elements for individual survival is, probably, due to mechanisms of natural selection, resulting in limited reparative potential of this tissue. Scanty cellular sources and low metabolic rate along with avascularity of cartilage contribute to its decreased regeneration ability. As a result of these strong limitations, the injuries of cartilage caused by inflammation, traumas, degeneration, and aging usually become chronic and recalcitrant to any kind of medical treatment. In the USA, according to tentative estimations, the prevalence of all forms of arthritis has been calculated in order of 40 million people; and the annual medical care costs were about 65 billion USD.1Degenerative spine diseases are a major cause of back pain that deteriorates the quality of life of patients and often leads to disability. Direct and indirect medical expenses are estimated as more than 90 billion per year.2
High prevalence and incidence, as well as the social and economic significance of cartilage pathology, attract great interest to this problem. Considerable efforts have been devoted to study various approaches to restore cartilage structures and to stimulate intrinsic capabilities of the tissue to regeneration. There are several treatment modalities of cartilage restoration suggested for clinical use (see Ref. 3 and referred literature): 1. surgical techniques; 2. controllable cell delivery to the lesion; and 3. tissue engineering applications of biodegradable materials (scaffolds) with cell-seeding and modification of cartilage reparative response by different growth factors and cytokines. Although there is a wealth of information regarding the substitution of lost cartilage by the mentioned approaches, the problem of cartilage repair is still unsolved. The long term results show no completed cartilage regeneration; in many cases, the new growing tissue materially differs from the well organized original cartilage. The reasons of insufficient cartilage reparation are connected with its structural and functional organization and with the difficulties of the precise control of the external physical and chemical effects.4, 5 Regeneration of cartilage may be realized in accordance with the natural genetic program of the cells. The efficacy of any approach aimed to control the regeneration process depends on the solution of three tasks: 1. the ability to reproduce the normal cell differentiation sequence from the progenitor cells to mature chondrocytes, 2. stimulation of the specific subpopulations of the resident cells to proliferation and/or new matrix production, and (c) achievement of adequate spatial organization of the new growing tissue. Probably, the most important feature of the laser-based treatment is the involvement and activation of the intrinsic mechanisms of cartilage repair. Many papers are devoted to the effect of low-intensive lasers on cartilage functional state and reparative ability. However, the effectiveness, as well as the placebo-versus-treatment ratio for low level laser therapy, is still under considerable dispute. A more detailed discussion of this issue may be found elsewhere.6 This paper is mainly limited with a consideration of the effect of nonablative laser radiation on the cartilaginous cells through their matrix microenvironment to provide natural and optimal conditions for regeneration. Wide ranges of wavelengths, precise localization of the irradiated area, and temporal and spatial modulation of laser radiation are the main advantages of the laser technologies, which may result in specific tissue response. In particular, the laser-induced modification of the cartilage extracellular matrix (ECM) seems to be of great significance in view of some new data on the developmental roles of the matrix molecules and mechanical loads. Although the evidence of laser irradiation morphogenetic effects is still largely circumstantial, we consider the available observations to address some possible perspectives of the controlled regeneration of cartilage using nonablative laser treatment. So, the aim of this paper is to review physical and biological aspects of laser-induced regeneration of cartilage, to discuss the possibilities and prospects of its clinical applications. The problems and the ways of tissue regeneration and the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches to cartilage repair, consider possible mechanisms of laser-induced regeneration, present the results in laser regeneration of joints and spine disks cartilages, and finally, discuss some future medical applications of laser regeneration.
2.
Cartilage as a Subject of Regeneration
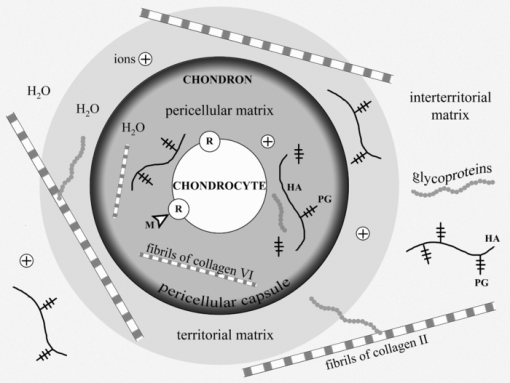
There are a number of detailed reviews describing the structure and vital functions of cartilages.3, 7, 8 The main components of cartilage are cells (chondrocytes) and ECM consisting of water (70 to 80%), collagens, proteoglycans (PGs), hyaluronic acid (HA), and glycoproteins (GP). The PGs consist of glycosaminoglycanes (chondroitin sulphate and keratan sulphate) linked to the core-protein, which, in turn, is bound with HA threads interweaving between collagen fibrils (Fig. 1). PGs have a lot of negative charged groups; and the electrical neutrality of cartilage is due to the presence of positive ions (K+, Na+, H+, Ca2+, Mg2+). There are three types of cartilage tissue: hyaline cartilage (costal, nasal septum, articular cartilage of the joints), fibrous cartilage (annulus fibrosis of the spine disks, Eustachian tube), and elastic cartilage (auricle, epiglottis). Hyaline cartilage first forms in embryos and later transforms into other types of cartilage and bone tissues. The distinguishing features of the ECM of hyaline cartilage are having a very high content of glycosaminoglycanes and the prevalence of collagen type II fibrils.9, 10, 11, 12, 13 Fibrous cartilage is characterized by predominance of collagen type I.14, 15, 16 Matrix of the elastic cartilage possesses elastic fibers. Nasal and some other cartilages are covered with a perichondrium playing an important role in nutrition and growth of the avascular tissue. Articular cartilage has no perichondrium; it gets nutrition from synovial liquid and subchondral bone. An articular cartilage surface is covered by a cell-free lamina splendens (LS) consisting mainly of the HA and phospholipids.17 An important structural and metabolic unit of articular cartilage is a chondron.11 It includes a chondrocyte and its pericellular matrix (PM) bordered with a pericellular capsule (PC). The chondron is surrounded by territorial and interterritorial matrices. The chondrons and their matrix environment have different mechanical properties.3, 11 The PM is enriched with HA, sulphated PGs, biglycan, and GPs, including link protein and laminin. The PC is predominantly composed of compact thin fibrils of collagen type VI and fibronectin. It is suggested that the PM and PC provide hydrodynamic protection for the chondrocyte against pressure loading and take a part in control of spatial and temporal distribution of newly synthesized macromolecules as well as in the cell-matrix interaction.11 Territorial and interterritorial matrices are characterized by different degrees of the PGs maturity and with a different proportion of the chondroitin sulphate and keratan sulphate. The heteropolymeric fibrils of collagen types II, IX, and XI (HCF) emerging in the territorial matrice become the major load-bearing element in the interterritorial matrice.16 These fibrils are in charge of the tissue protection against multidirectional tensions.
Fig. 1
Cartilage components and structure. PG –proteolycanes; HA – hyaluronic acid, GP – glycoproteins; M – morphogenes; R -molecular receptors of chondrocyte's membrane; (+) ions (K+, Na+, H+, Ca2+, Mg2+).
A number of molecules that possess signal roles in morphogenetic processes, including chondrogenesis from embryonic development to regeneration, may interact with the receptors of the cellular membrane of chondrocyte. Binding of such morphogenes to the membrane receptors triggers various intracellular signaling cascades to result in regulation of the expression of genes. Hydrostatic pressures and fluid flows as well as multidirectional tensions contribute to tissue water displacement leading to changes of local concentrations of ions and morphogens. The GP molecules (integrins, fibronectin, laminin, etc.) distributing over the ECM serve as important mediators of the signaling molecules. They play an important role in the cell-matrix interactions and operate on the growth of cartilage tissue.
Hyaline cartilage has a zonal structure:11, 18, 19 the superficial layer contains fibroblast-like chondrocytes of type I. It is characterized by a decreased level of the PG aggregates (aggrecanes) and by a high content of small leucine-rich PGs (decorin and biglycan). The cells in the middle layer are chondrocytes of type II. They form multicellular clones and keep a certain ability of proliferation. A smaller subpopulation of the middle layer cells is presented by the chondrocytes type III covered with lacunas. These nonproliferating cells are also presented in the deep layer of cartilage. Type IV cells belong to a degrading cell group. Chondrocytes synthesize and degrade all components of cartilage matrix through specialized enzymes (prolyl hydroxylase, lysil oxidase, collagenases, aggrecanases etc.).16, 20 Metabolic activity of the chondrocytes in cartilage is controlled by hormones, various cytokines, growth factors, and vitamins (A, C, and D).21, 22, 23, 24 Ultimately, the biosynthetic and catabolic activities of cartilage cells, as well as the kinetics of the cellular population are governed by the local concentrations of the humoral and insoluble morphogens near the external membranes of chondrocytes.
The main mechanism of cartilage nutrition is diffusion of water carrying low-molecular substances (ions, glucose, amino acids, etc.). As the chondrocytes kinetics are under conditions of hypoxia, their metabolism is generally realized by the anaerobic glycolysis pathway. That, in combination with the chondrocytes paucity, determines a low level of cartilage metabolism. Half life period is three or four years for aggrecans, and about 10 years for collagen.25 All types of cartilage, especially articular cartage and intervertebral disks, have low repair potential. There is a lot of literature on this topic. 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37 Extra-articular cartilage is usually repaired by the means of proliferation and chondrogenic differentiation of the perichondrial cells. The defects of hyaline cartilage and the extensive defects of costal and auricular cartilages are usually filled up with fibrous connective tissue or fibrous cartilage, which both do not have adequate functional properties; that determines persistent attempts to find new possibilities for cartilage regeneration. The healing of cartilage defects can be improved with mechanical stimulation, intra-articular application of HA, hormone therapy,38, 39, 40, 41 and also with the use of osteochondral or cartilaginous implants, in particular together with cultivated chondrocytes.42, 43 One of the current leading approaches is in vitro growth of the tissue engineering constructs followed with their implantation into cartilage lesion. Autologous chondrocyte implantation (ACI) resulted in the formation of hyaline-like tissue with a quite stable clinical outcome.44, 45 But according to the histological data, only 39% of the defects treated with ACI were filled with hyaline cartilage, while 43% were filled with fibrocartilage, and 18% did not show any healing response at all.46
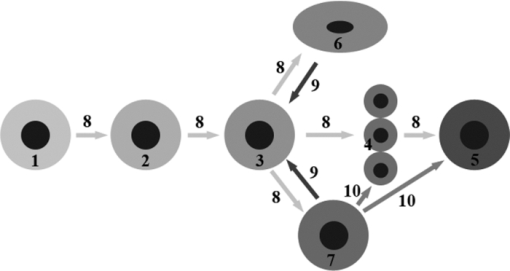
Regeneration process is associated with embryonic chondrogenesis mechanisms and partial dedifferentiation of mature cells. Figure 2 shows possible pathways of regeneration-related dedifferentiation of the cells in cartilage. Mesenchymal stem cells (MSC) can differentiate into cartilage cells of various types, including immature and mature chondrocytes, and notochordal and chondrocyte-like cells of the intervertebral disks. These processes are under multilevel control of signaling molecules and mechanical factors. Our main hypothesis is that differentiation and dedifferentiation of cartilage cells, as well as their metabolic activity, may be controlled by direct action of laser radiation on the cells and through laser-induced modification of the ECM.
Fig. 2
Differentiation of cartilage cells and possible pathways of their regeneration-related dedifferentiation. 1 – MSC, 2 – pre-chondrocytes, 3 – early chondrocytes (chondroblasts), 4 – columnar chondrocytes, 5 – hypertrophic chondrocytes, 6 – chondrocytes of fibrous cartilage, 7 – chondrocytes of hyaline cartilage, 8 – differentiation pathways, 9 – pathways of limited dedifferentiation, 10 – additional pathways of cellular differentiation (following the enchondral osteogenesis differentiation mechanism).
3.
Targets for Laser Effect. Possible Types of Cartilage Response on Laser Radiation
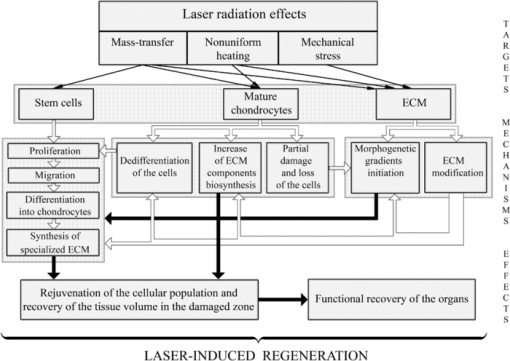
To discuss possible ways of using lasers for cartilage regeneration, it is important to know what effect laser parameters have on (a) different types of the cells; (b) different components of the ECM; (c) signaling molecules produced by the cells and accumulated in the ECM; (d) intercellular and cell-matrix interactions; (e) differentiation and dedifferentiation of the cells, their migration and biosynthesis activity. Feasible pathways promoting cartilage regeneration include: 1. additional cellular supply from bone marrow and blood; 2. biosynthesis amplification of the ECM components, 3. stimulation of the motility of mature chondrocytes, and 4. activation of resident adult stem cells toward their proliferation, differentiation, and ECM production. The main reasons of the low regeneration potential of cartilage are advanced differentiation of the resident chondrocytes and relatively slow metabolism of the tissue. The nonablative laser radiation may provide controllable thermal and mechanical effects (as on the cells, as on the matrix) resulting in activation of the cellular biosynthesis. In particular, nonuniform laser heating of cartilage induces heterogeneous thermal expansion, stress, and also the movement of the interstitial water and ions (see Fig. 3 and Sec. 4).
Fig. 3
Targets and mechanisms of the laser-induced regeneration of cartilage. Thin straight arrows show the direct laser influence on the components of cartilage. White thick arrows represent the most important biological responses to laser action. Dark thick arrows show the processes leading directly to regeneration.
One of the major obstacles for regeneration of cartilage, including partial-thickness defect of articular cartilage, is its avascularity, which hampers the progenitor cell movement from the blood and marrow to the damaged areas of the tissue. Preventing an entry of unspecialized cells and diminishing the rate of cartilage repair that slow regeneration, nevertheless, may have its good point, as it may potentially result in the growth of well organized tissue of the hyaline cartilage. Rapid repair of the full-thickness cartilage defects usually leads to undesirable growth of fibrous connective tissue or fibrous cartilage due to the impact of blood and bone-marrow-derived cells. It can be better understood by the following analogy. It is known that skin wound healing resulting in a fibrous scar is going through emergency regeneration due to swift proliferation of unspecialized fibroblasts. Their sources are the precursor cells coming into the wound via blood. These cells have nonspecific genetic program and form scar.47 In a similar manner, the bone-marrow cells coming to the full-thickness defect of articular cartilage differentiate into the fibroblasts of nonspecific connective tissue or into the chondrocytes of the fibrous cartilage. This provides quick filling of the defect, but fails in functionality of the novel tissue. One of the possible ways to promote growth of the hyaline cartilage in the full-thickness defects of articular cartilage plates can be laser-induced coagulation of the bottom of the defect. This may prevent access of unspecialized precursor cells from the blood or bone-marrow in order to develop more specific, i.e., hyaline cartilage.
It is known that in the course of embryogenesis, the hyaline cartilage forms in the zones undergoing compression load (articular cartilage), whereas, the fibrous cartilage (meniscus, annulus fibrosis of the intervertebral disk) usually develops in the stretched or torsioned zones. Spatial and temporal modulation of laser radiation allows controlling the actual distribution of stretched and compressed zones in cartilage. The mechanical loads are important factors governing an orchestra of chondrogenesis, including the processes of cellular differentiation. Therefore, the nonablative laser treatment may play a triggering role in the differentiation of immature cartilage cells. Laser radiation may probably be responsible for the reverse process of dedifferentiation of the mature chondrocytes leading to the recovery of their ability to divide. Existing natural pathways of cells dedifferentiation (see Fig. 2) open possibilities for tissue correction, in particular, replacement of abnormally grown fibrous tissue by hyaline cartilage possessing adequate mechanical and functional properties (Fig. 3).
Laser radiation can also be used to stimulate proliferation and acquiring the specialized phenotype by resident stem cells or MSC coming through synovial liquid in order to promote their transformation into mature hyaline-like chondrocytes. This approach is critically significant for healing of the partial-thickness defects of articular cartilage. At the same time, as the cellular population in full-thickness cartilage defect is highly heterogenic, laser irradiation may effect the proliferation of different kinds of cells. Thereafter, the additional controlling factor of the ECM architecture should be taken into account. Laser modification of the fine structure of ECM does not change its general organization. This provides natural environments for chondrocytes and promotes restoration of the hyaline type cartilage. One of the important factors is cell movement velocity, which correlates with the alignment of the matrix fibrillar components.48, 49 Nonablative laser irradiation allows structure modification and diffusion properties of ECM. This may support cell movement and favor tissue regeneration.
Laser-induced growth of hyaline cartilage in elastic cartilage was established in the course of in vivoexperiments on laser reshaping of porcine ears.50 The effects of laser irradiation on gene expression of chondrocytes and collagen of ECM have been studied for rabbit septal cartilage using laser settings typical for laser reshaping procedure.51 It was shown that laser irradiation of cartilage does not result in the detection of collagen type I. Only collagen type II was observed after laser irradiation in the corresponding cell culture in vitro. This fact indicates that cartilage cellular response to nonablative laser irradiation differs from the reaction of conventional wound healing. Laser irradiation of cartilage can leave intact collagen and preserve general matrix architecture, which favors chondrocyte survival and promotes new tissue growth. Evidence of hyaline cartilage development in laser-irradiated intervertebral disks was revealed in the animal experiments (see Sec. 5.2). The advantage of the laser effect on chondrocytes proliferation compared to other thermal, mechanical, and chemical effects was demonstrated in Ref. 52. No evidence of chondrocyte DNA replication was observed in tissues heated using nonlaser methods, grown in TGF-β-contained media, or mechanically traumatized. In contrast, for laser irradiated chondrocytes, flow cytometry provided evidence that laser irradiation causes a proliferative response in cho...
Original Source: https://www.spiedigitallibrary.org/journals/journal-of-biomedical-optics/volume-16/issue-08/080902/Laser-induced-regeneration-of-cartilage/10.1117/1.3614565.full?SSO=1
Home Search Introduction
Ken Teegardin - (Website) 4361
View Resource
This tool is a searchable collection of technical publications, books, videos and other resources about the use of lasers and light for PhotoBioModulation (PBM). Enter a keyword above or see some of our favorite queries below.
Here are some of our favorite queries:
- Information for Beginners
- Best wavelength for PBM (980nm vs. 810nm vs 660nm...)
- Compare Lasers and LEDs
- Pulsing versus Continuous Wave (CW)
- Dosage: The best practices for dosage
- How NOT to promote laser therapy (by Turner and Hode)
- Great summary of positive double blind studies
- Whole Body Systems
- See all videos
- See all books about laser therapy and cold lasers
- Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing Athletic Performance
- Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Intro to laser acupuncture
- Traumatic brain injury research
- Soft Tissue
- Autoimmune
- Lyme's Disease
- Parkinson's research
- Alzheimer's research
- Hashimoto's research
- Covid
- Contraindications
- LLLT and Cancer
All the resources include links to the original source so we are not making any statement about the use of lasers for treating non-FDA cleared application, we are simple summarizing what others have said.
Where every possible, we have included a link to the orginal publication.
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- Where ever possible, the detailed section about the resource will link to the sources.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic lasers. It does include some resources on weight loss and smoking cessation.
The results of the search are sorted based on 3 quality factors on a scale of 1 to 10 with 10 being the best score. Originally all the resources were given a 5-5-5 until they could be individually evaluated. These scores are purely opinion and are only used to simplify the rank of the results from more valuable to least valuable. This should not be considered a critique of any work. This system was created to help researchers (including ourselves) find the most usable resources for any cold laser therapy research. The resources are assigned values based on the following 3 factors:
- Efficacy: The resource (especially research papers) should show a significant improvement in the condition being treated. Resources that show better results are given a higher quality score.
- Detail: The source must give enough information that the results can be duplicated. If a resource lacks too many details that it cannot be recreated, it is given a lower detail score.
- Lack of Bias: Many resources are created to try and show that one device is superior to its competition. Many manufacturers have staff that crank out biased papers on a regular basis on the hope that this will make their product look superior. If the author of the resource is paid by a manufacturer of the resource appears to be biased towards one device and not one technology, the resource has much less value.
Over the past few years of working with research, we found that a majority of the published resources are lacking in one of these three ranking factors.
The original goal of this research tool was to tie published resources to the protocols in the laser-therapy.us library. This connection allows users to trace each protocol back to a list of resources so the protocol can be researched and improved.
General Comments
POWER
When many of the first research papers were published, the most power laser available for therapy were less than 100mW and many systems had to be pulsed to keep the laser from burning out too quickly. Today, system are available that will deliver up to 60,000mW of continuous output. Because of these power limitation, many early studies were limited to extremely low dosages by today’s standards. It takes a 50mW system 17 minutes to deliver 50 joules at the surface of the skin. If this was spread over a large area of damage or was treating a deeper problem, the actual dosages were much less than 1J/cm2. Today, we know that these dosages typically produce very little or no results.
WAVELENGTH
About 80% of the resources in this database are in the near infrared wavelength. There is also some interest in the red wavelength (600 to 660nm) . Other wavelengths like blue, purple, and green have very little scientific research behind them and have not gotten much traction in the core therapy market with the exception of some fringe consumer products.
Legal Disclaimer
This research tool is free to use but we make no claims about the accuracy of the information. It is an aggregation of existing published resources and it is up to the user to determine if the source of the resources has any value. The information provided through this web site should not be used for diagnosing or treating a health problem or disease. If you have or suspect you may have a health problem, you should consult your local health care provider.
Biophtonica Introduction
Biophotonica - (Website) 4523
View Resource
Welcome to the BioPhotonica Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
- Fertility and Reproductive Health
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only PBM related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
Light House Health Introduction
LightHouse - (Website) 4515
View Resource
Welcome to the Lighthouse Health Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
The query result(s) can be shared using the following direct link. Anyone who clicks on this link in an email or on a web site will be shown the current results for the query.
https://www.laser-therapy.us/research/index.cfm?researchinput=softtissue
Laser-induced regeneration of cartilage
Emil N. Sobol, Olga I. Baum, Anatoliy B. Shekhter, Anna Guller, Andrey V. Baskov - J. of Biomedical Optics, 16(8), 080902 (2011) (Publication) 4501This article shows results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine
Abstract
Laser radiation provides a means to control the fields of temperature and thermo mechanical stress, mass transfer, and modification of fine structure of the cartilage matrix. The aim of this outlook paper is to review physical and biological aspects of laser-induced regeneration of cartilage and to discuss the possibilities and prospects of its clinical applications. The problems and the pathways of tissue regeneration, the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches for cartilage repair; consider possible mechanisms of laser-induced regeneration. Finally, we present the results in laser regeneration of joints and spine disks cartilages and discuss some future applications of lasers in regenerative medicine.
1.
Introduction
Cartilage is a kind of highly specialized connective tissue. The structural variety of the cartilage provides its unique biomechanical capacity to bear different kinds of static and dynamic loads over a wide range of intensity. Biological role of cartilage structures stems from their critical significance for growth and development as well as for all kinds of body movements. The exceptional importance of cartilage elements for individual survival is, probably, due to mechanisms of natural selection, resulting in limited reparative potential of this tissue. Scanty cellular sources and low metabolic rate along with avascularity of cartilage contribute to its decreased regeneration ability. As a result of these strong limitations, the injuries of cartilage caused by inflammation, traumas, degeneration, and aging usually become chronic and recalcitrant to any kind of medical treatment. In the USA, according to tentative estimations, the prevalence of all forms of arthritis has been calculated in order of 40 million people; and the annual medical care costs were about 65 billion USD.1Degenerative spine diseases are a major cause of back pain that deteriorates the quality of life of patients and often leads to disability. Direct and indirect medical expenses are estimated as more than 90 billion per year.2
High prevalence and incidence, as well as the social and economic significance of cartilage pathology, attract great interest to this problem. Considerable efforts have been devoted to study various approaches to restore cartilage structures and to stimulate intrinsic capabilities of the tissue to regeneration. There are several treatment modalities of cartilage restoration suggested for clinical use (see Ref. 3 and referred literature): 1. surgical techniques; 2. controllable cell delivery to the lesion; and 3. tissue engineering applications of biodegradable materials (scaffolds) with cell-seeding and modification of cartilage reparative response by different growth factors and cytokines. Although there is a wealth of information regarding the substitution of lost cartilage by the mentioned approaches, the problem of cartilage repair is still unsolved. The long term results show no completed cartilage regeneration; in many cases, the new growing tissue materially differs from the well organized original cartilage. The reasons of insufficient cartilage reparation are connected with its structural and functional organization and with the difficulties of the precise control of the external physical and chemical effects.4, 5 Regeneration of cartilage may be realized in accordance with the natural genetic program of the cells. The efficacy of any approach aimed to control the regeneration process depends on the solution of three tasks: 1. the ability to reproduce the normal cell differentiation sequence from the progenitor cells to mature chondrocytes, 2. stimulation of the specific subpopulations of the resident cells to proliferation and/or new matrix production, and (c) achievement of adequate spatial organization of the new growing tissue. Probably, the most important feature of the laser-based treatment is the involvement and activation of the intrinsic mechanisms of cartilage repair. Many papers are devoted to the effect of low-intensive lasers on cartilage functional state and reparative ability. However, the effectiveness, as well as the placebo-versus-treatment ratio for low level laser therapy, is still under considerable dispute. A more detailed discussion of this issue may be found elsewhere.6 This paper is mainly limited with a consideration of the effect of nonablative laser radiation on the cartilaginous cells through their matrix microenvironment to provide natural and optimal conditions for regeneration. Wide ranges of wavelengths, precise localization of the irradiated area, and temporal and spatial modulation of laser radiation are the main advantages of the laser technologies, which may result in specific tissue response. In particular, the laser-induced modification of the cartilage extracellular matrix (ECM) seems to be of great significance in view of some new data on the developmental roles of the matrix molecules and mechanical loads. Although the evidence of laser irradiation morphogenetic effects is still largely circumstantial, we consider the available observations to address some possible perspectives of the controlled regeneration of cartilage using nonablative laser treatment. So, the aim of this paper is to review physical and biological aspects of laser-induced regeneration of cartilage, to discuss the possibilities and prospects of its clinical applications. The problems and the ways of tissue regeneration and the types and features of cartilage will be introduced first. Then we will review various actual and prospective approaches to cartilage repair, consider possible mechanisms of laser-induced regeneration, present the results in laser regeneration of joints and spine disks cartilages, and finally, discuss some future medical applications of laser regeneration.
2.
Cartilage as a Subject of Regeneration
There are a number of detailed reviews describing the structure and vital functions of cartilages.3, 7, 8 The main components of cartilage are cells (chondrocytes) and ECM consisting of water (70 to 80%), collagens, proteoglycans (PGs), hyaluronic acid (HA), and glycoproteins (GP). The PGs consist of glycosaminoglycanes (chondroitin sulphate and keratan sulphate) linked to the core-protein, which, in turn, is bound with HA threads interweaving between collagen fibrils (Fig. 1). PGs have a lot of negative charged groups; and the electrical neutrality of cartilage is due to the presence of positive ions (K+, Na+, H+, Ca2+, Mg2+). There are three types of cartilage tissue: hyaline cartilage (costal, nasal septum, articular cartilage of the joints), fibrous cartilage (annulus fibrosis of the spine disks, Eustachian tube), and elastic cartilage (auricle, epiglottis). Hyaline cartilage first forms in embryos and later transforms into other types of cartilage and bone tissues. The distinguishing features of the ECM of hyaline cartilage are having a very high content of glycosaminoglycanes and the prevalence of collagen type II fibrils.9, 10, 11, 12, 13 Fibrous cartilage is characterized by predominance of collagen type I.14, 15, 16 Matrix of the elastic cartilage possesses elastic fibers. Nasal and some other cartilages are covered with a perichondrium playing an important role in nutrition and growth of the avascular tissue. Articular cartilage has no perichondrium; it gets nutrition from synovial liquid and subchondral bone. An articular cartilage surface is covered by a cell-free lamina splendens (LS) consisting mainly of the HA and phospholipids.17 An important structural and metabolic unit of articular cartilage is a chondron.11 It includes a chondrocyte and its pericellular matrix (PM) bordered with a pericellular capsule (PC). The chondron is surrounded by territorial and interterritorial matrices. The chondrons and their matrix environment have different mechanical properties.3, 11 The PM is enriched with HA, sulphated PGs, biglycan, and GPs, including link protein and laminin. The PC is predominantly composed of compact thin fibrils of collagen type VI and fibronectin. It is suggested that the PM and PC provide hydrodynamic protection for the chondrocyte against pressure loading and take a part in control of spatial and temporal distribution of newly synthesized macromolecules as well as in the cell-matrix interaction.11 Territorial and interterritorial matrices are characterized by different degrees of the PGs maturity and with a different proportion of the chondroitin sulphate and keratan sulphate. The heteropolymeric fibrils of collagen types II, IX, and XI (HCF) emerging in the territorial matrice become the major load-bearing element in the interterritorial matrice.16 These fibrils are in charge of the tissue protection against multidirectional tensions.
Fig. 1
Cartilage components and structure. PG –proteolycanes; HA – hyaluronic acid, GP – glycoproteins; M – morphogenes; R -molecular receptors of chondrocyte's membrane; (+) ions (K+, Na+, H+, Ca2+, Mg2+).
A number of molecules that possess signal roles in morphogenetic processes, including chondrogenesis from embryonic development to regeneration, may interact with the receptors of the cellular membrane of chondrocyte. Binding of such morphogenes to the membrane receptors triggers various intracellular signaling cascades to result in regulation of the expression of genes. Hydrostatic pressures and fluid flows as well as multidirectional tensions contribute to tissue water displacement leading to changes of local concentrations of ions and morphogens. The GP molecules (integrins, fibronectin, laminin, etc.) distributing over the ECM serve as important mediators of the signaling molecules. They play an important role in the cell-matrix interactions and operate on the growth of cartilage tissue.
Hyaline cartilage has a zonal structure:11, 18, 19 the superficial layer contains fibroblast-like chondrocytes of type I. It is characterized by a decreased level of the PG aggregates (aggrecanes) and by a high content of small leucine-rich PGs (decorin and biglycan). The cells in the middle layer are chondrocytes of type II. They form multicellular clones and keep a certain ability of proliferation. A smaller subpopulation of the middle layer cells is presented by the chondrocytes type III covered with lacunas. These nonproliferating cells are also presented in the deep layer of cartilage. Type IV cells belong to a degrading cell group. Chondrocytes synthesize and degrade all components of cartilage matrix through specialized enzymes (prolyl hydroxylase, lysil oxidase, collagenases, aggrecanases etc.).16, 20 Metabolic activity of the chondrocytes in cartilage is controlled by hormones, various cytokines, growth factors, and vitamins (A, C, and D).21, 22, 23, 24 Ultimately, the biosynthetic and catabolic activities of cartilage cells, as well as the kinetics of the cellular population are governed by the local concentrations of the humoral and insoluble morphogens near the external membranes of chondrocytes.
The main mechanism of cartilage nutrition is diffusion of water carrying low-molecular substances (ions, glucose, amino acids, etc.). As the chondrocytes kinetics are under conditions of hypoxia, their metabolism is generally realized by the anaerobic glycolysis pathway. That, in combination with the chondrocytes paucity, determines a low level of cartilage metabolism. Half life period is three or four years for aggrecans, and about 10 years for collagen.25 All types of cartilage, especially articular cartage and intervertebral disks, have low repair potential. There is a lot of literature on this topic. 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37 Extra-articular cartilage is usually repaired by the means of proliferation and chondrogenic differentiation of the perichondrial cells. The defects of hyaline cartilage and the extensive defects of costal and auricular cartilages are usually filled up with fibrous connective tissue or fibrous cartilage, which both do not have adequate functional properties; that determines persistent attempts to find new possibilities for cartilage regeneration. The healing of cartilage defects can be improved with mechanical stimulation, intra-articular application of HA, hormone therapy,38, 39, 40, 41 and also with the use of osteochondral or cartilaginous implants, in particular together with cultivated chondrocytes.42, 43 One of the current leading approaches is in vitro growth of the tissue engineering constructs followed with their implantation into cartilage lesion. Autologous chondrocyte implantation (ACI) resulted in the formation of hyaline-like tissue with a quite stable clinical outcome.44, 45 But according to the histological data, only 39% of the defects treated with ACI were filled with hyaline cartilage, while 43% were filled with fibrocartilage, and 18% did not show any healing response at all.46
Regeneration process is associated with embryonic chondrogenesis mechanisms and partial dedifferentiation of mature cells. Figure 2 shows possible pathways of regeneration-related dedifferentiation of the cells in cartilage. Mesenchymal stem cells (MSC) can differentiate into cartilage cells of various types, including immature and mature chondrocytes, and notochordal and chondrocyte-like cells of the intervertebral disks. These processes are under multilevel control of signaling molecules and mechanical factors. Our main hypothesis is that differentiation and dedifferentiation of cartilage cells, as well as their metabolic activity, may be controlled by direct action of laser radiation on the cells and through laser-induced modification of the ECM.
Fig. 2
Differentiation of cartilage cells and possible pathways of their regeneration-related dedifferentiation. 1 – MSC, 2 – pre-chondrocytes, 3 – early chondrocytes (chondroblasts), 4 – columnar chondrocytes, 5 – hypertrophic chondrocytes, 6 – chondrocytes of fibrous cartilage, 7 – chondrocytes of hyaline cartilage, 8 – differentiation pathways, 9 – pathways of limited dedifferentiation, 10 – additional pathways of cellular differentiation (following the enchondral osteogenesis differentiation mechanism).
3.
Targets for Laser Effect. Possible Types of Cartilage Response on Laser Radiation
To discuss possible ways of using lasers for cartilage regeneration, it is important to know what effect laser parameters have on (a) different types of the cells; (b) different components of the ECM; (c) signaling molecules produced by the cells and accumulated in the ECM; (d) intercellular and cell-matrix interactions; (e) differentiation and dedifferentiation of the cells, their migration and biosynthesis activity. Feasible pathways promoting cartilage regeneration include: 1. additional cellular supply from bone marrow and blood; 2. biosynthesis amplification of the ECM components, 3. stimulation of the motility of mature chondrocytes, and 4. activation of resident adult stem cells toward their proliferation, differentiation, and ECM production. The main reasons of the low regeneration potential of cartilage are advanced differentiation of the resident chondrocytes and relatively slow metabolism of the tissue. The nonablative laser radiation may provide controllable thermal and mechanical effects (as on the cells, as on the matrix) resulting in activation of the cellular biosynthesis. In particular, nonuniform laser heating of cartilage induces heterogeneous thermal expansion, stress, and also the movement of the interstitial water and ions (see Fig. 3 and Sec. 4).
Fig. 3
Targets and mechanisms of the laser-induced regeneration of cartilage. Thin straight arrows show the direct laser influence on the components of cartilage. White thick arrows represent the most important biological responses to laser action. Dark thick arrows show the processes leading directly to regeneration.
One of the major obstacles for regeneration of cartilage, including partial-thickness defect of articular cartilage, is its avascularity, which hampers the progenitor cell movement from the blood and marrow to the damaged areas of the tissue. Preventing an entry of unspecialized cells and diminishing the rate of cartilage repair that slow regeneration, nevertheless, may have its good point, as it may potentially result in the growth of well organized tissue of the hyaline cartilage. Rapid repair of the full-thickness cartilage defects usually leads to undesirable growth of fibrous connective tissue or fibrous cartilage due to the impact of blood and bone-marrow-derived cells. It can be better understood by the following analogy. It is known that skin wound healing resulting in a fibrous scar is going through emergency regeneration due to swift proliferation of unspecialized fibroblasts. Their sources are the precursor cells coming into the wound via blood. These cells have nonspecific genetic program and form scar.47 In a similar manner, the bone-marrow cells coming to the full-thickness defect of articular cartilage differentiate into the fibroblasts of nonspecific connective tissue or into the chondrocytes of the fibrous cartilage. This provides quick filling of the defect, but fails in functionality of the novel tissue. One of the possible ways to promote growth of the hyaline cartilage in the full-thickness defects of articular cartilage plates can be laser-induced coagulation of the bottom of the defect. This may prevent access of unspecialized precursor cells from the blood or bone-marrow in order to develop more specific, i.e., hyaline cartilage.
It is known that in the course of embryogenesis, the hyaline cartilage forms in the zones undergoing compression load (articular cartilage), whereas, the fibrous cartilage (meniscus, annulus fibrosis of the intervertebral disk) usually develops in the stretched or torsioned zones. Spatial and temporal modulation of laser radiation allows controlling the actual distribution of stretched and compressed zones in cartilage. The mechanical loads are important factors governing an orchestra of chondrogenesis, including the processes of cellular differentiation. Therefore, the nonablative laser treatment may play a triggering role in the differentiation of immature cartilage cells. Laser radiation may probably be responsible for the reverse process of dedifferentiation of the mature chondrocytes leading to the recovery of their ability to divide. Existing natural pathways of cells dedifferentiation (see Fig. 2) open possibilities for tissue correction, in particular, replacement of abnormally grown fibrous tissue by hyaline cartilage possessing adequate mechanical and functional properties (Fig. 3).
Laser radiation can also be used to stimulate proliferation and acquiring the specialized phenotype by resident stem cells or MSC coming through synovial liquid in order to promote their transformation into mature hyaline-like chondrocytes. This approach is critically significant for healing of the partial-thickness defects of articular cartilage. At the same time, as the cellular population in full-thickness cartilage defect is highly heterogenic, laser irradiation may effect the proliferation of different kinds of cells. Thereafter, the additional controlling factor of the ECM architecture should be taken into account. Laser modification of the fine structure of ECM does not change its general organization. This provides natural environments for chondrocytes and promotes restoration of the hyaline type cartilage. One of the important factors is cell movement velocity, which correlates with the alignment of the matrix fibrillar components.48, 49 Nonablative laser irradiation allows structure modification and diffusion properties of ECM. This may support cell movement and favor tissue regeneration.
Laser-induced growth of hyaline cartilage in elastic cartilage was established in the course of in vivoexperiments on laser reshaping of porcine ears.50 The effects of laser irradiation on gene expression of chondrocytes and collagen of ECM have been studied for rabbit septal cartilage using laser settings typical for laser reshaping procedure.51 It was shown that laser irradiation of cartilage does not result in the detection of collagen type I. Only collagen type II was observed after laser irradiation in the corresponding cell culture in vitro. This fact indicates that cartilage cellular response to nonablative laser irradiation differs from the reaction of conventional wound healing. Laser irradiation of cartilage can leave intact collagen and preserve general matrix architecture, which favors chondrocyte survival and promotes new tissue growth. Evidence of hyaline cartilage development in laser-irradiated intervertebral disks was revealed in the animal experiments (see Sec. 5.2). The advantage of the laser effect on chondrocytes proliferation compared to other thermal, mechanical, and chemical effects was demonstrated in Ref. 52. No evidence of chondrocyte DNA replication was observed in tissues heated using nonlaser methods, grown in TGF-β-contained media, or mechanically traumatized. In contrast, for laser irradiated chondrocytes, flow cytometry provided evidence that laser irradiation causes a proliferative response in cho...
Original Source: https://www.spiedigitallibrary.org/journals/journal-of-biomedical-optics/volume-16/issue-08/080902/Laser-induced-regeneration-of-cartilage/10.1117/1.3614565.full?SSO=1
Home Search Introduction
Ken Teegardin - (Website) 4361This tool is a searchable collection of technical publications, books, videos and other resources about the use of lasers and light for PhotoBioModulation (PBM). Enter a keyword above or see some of our favorite queries below.
Here are some of our favorite queries:
- Information for Beginners
- Best wavelength for PBM (980nm vs. 810nm vs 660nm...)
- Compare Lasers and LEDs
- Pulsing versus Continuous Wave (CW)
- Dosage: The best practices for dosage
- How NOT to promote laser therapy (by Turner and Hode)
- Great summary of positive double blind studies
- Whole Body Systems
- See all videos
- See all books about laser therapy and cold lasers
- Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing Athletic Performance
- Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Intro to laser acupuncture
- Traumatic brain injury research
- Soft Tissue
- Autoimmune
- Lyme's Disease
- Parkinson's research
- Alzheimer's research
- Hashimoto's research
- Covid
- Contraindications
- LLLT and Cancer
All the resources include links to the original source so we are not making any statement about the use of lasers for treating non-FDA cleared application, we are simple summarizing what others have said.
Where every possible, we have included a link to the orginal publication.
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- Where ever possible, the detailed section about the resource will link to the sources.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic lasers. It does include some resources on weight loss and smoking cessation.
The results of the search are sorted based on 3 quality factors on a scale of 1 to 10 with 10 being the best score. Originally all the resources were given a 5-5-5 until they could be individually evaluated. These scores are purely opinion and are only used to simplify the rank of the results from more valuable to least valuable. This should not be considered a critique of any work. This system was created to help researchers (including ourselves) find the most usable resources for any cold laser therapy research. The resources are assigned values based on the following 3 factors:
- Efficacy: The resource (especially research papers) should show a significant improvement in the condition being treated. Resources that show better results are given a higher quality score.
- Detail: The source must give enough information that the results can be duplicated. If a resource lacks too many details that it cannot be recreated, it is given a lower detail score.
- Lack of Bias: Many resources are created to try and show that one device is superior to its competition. Many manufacturers have staff that crank out biased papers on a regular basis on the hope that this will make their product look superior. If the author of the resource is paid by a manufacturer of the resource appears to be biased towards one device and not one technology, the resource has much less value.
Over the past few years of working with research, we found that a majority of the published resources are lacking in one of these three ranking factors.
The original goal of this research tool was to tie published resources to the protocols in the laser-therapy.us library. This connection allows users to trace each protocol back to a list of resources so the protocol can be researched and improved.
General Comments
POWER
When many of the first research papers were published, the most power laser available for therapy were less than 100mW and many systems had to be pulsed to keep the laser from burning out too quickly. Today, system are available that will deliver up to 60,000mW of continuous output. Because of these power limitation, many early studies were limited to extremely low dosages by today’s standards. It takes a 50mW system 17 minutes to deliver 50 joules at the surface of the skin. If this was spread over a large area of damage or was treating a deeper problem, the actual dosages were much less than 1J/cm2. Today, we know that these dosages typically produce very little or no results.
WAVELENGTH
About 80% of the resources in this database are in the near infrared wavelength. There is also some interest in the red wavelength (600 to 660nm) . Other wavelengths like blue, purple, and green have very little scientific research behind them and have not gotten much traction in the core therapy market with the exception of some fringe consumer products.
Legal Disclaimer
This research tool is free to use but we make no claims about the accuracy of the information. It is an aggregation of existing published resources and it is up to the user to determine if the source of the resources has any value. The information provided through this web site should not be used for diagnosing or treating a health problem or disease. If you have or suspect you may have a health problem, you should consult your local health care provider.
Biophtonica Introduction
Biophotonica - (Website) 4523Welcome to the BioPhotonica Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
- Fertility and Reproductive Health
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only PBM related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
Light House Health Introduction
LightHouse - (Website) 4515Welcome to the Lighthouse Health Education Center. There are over 5000 successful studies showing the efficacy of PBM, light therapy and sound therapy. This is a searchable collection of technical publications, books, videos and other resources about the best practices in the industry and about treating a wide variety of problems. All the resources include links to the original source (where available) so we are not making any claims about the use of our technology for treating "non-FDA cleared" applications, we are simply summarizing what the expert are saying about proper application of these technologies.
Enter a keyword above and click on one of the following links to see a set of publications about that subject. HINT: Shorter keywords work better.
Here are some of our favorite queries:
- Whole Body Systems
- Treating Post Covid with PBMT
- Info for beginners (See several graphic representation of the PBM chemical process)
- Best wavelength for light therapy (980nm vs. 810nm vs 660nm...)
- Pulsing Versus continuous wave
- Dosage: Searching for the best practices for dosage
- Great summary of positive double blind studies
Testimonials
- Amazing results
- Video testimonials from patients
- Video testimonials from doctors
- Incredible results in animals
- Increasing athletic performance
Research Info for other Applications
- Stem cell production in laser therapy
- Cosmetic and skin therapy parameters
- Laser acupuncture
- Traumatic Brain Injury (TBI)
- Soft Tissue Injury
Autoimmune Research
Contraindications
This tool uses a broad match query so:
- It does not correct spelling and searches only cold laser related subjects so do not use LLLT, cold or laser in the search bar
- It works better with shorter search terms or even parts of search terms
- It searches all the available fields so you can enter a body part, author, condition or laser brand.
- This system is only for photobiomodulation or cold laser therapy research (including LLLT, laser acupuncture and high power laser therapy) only. It does NOT include photodynamic laser therapy (where the laser is used to react with a pharmaceutical), hot surgery lasers or cosmetic therapies.
The query result(s) can be shared using the following direct link. Anyone who clicks on this link in an email or on a web site will be shown the current results for the query.
https://www.laser-therapy.us/research/index.cfm?researchinput=softtissue
2021 Laser-Therapy.US