Start New Query
An Hour of Light and Sound a Day Might Keep Alzheimer’s at Bay
Angus Chen - Scientific American March 14, 2019 (Publication) 4521
This is an summary article about the work of Shannon Macauley, a neuroscientist at Wake Forest School of Medicine who found that light and sound has a siginicant impact on the Alzheimer's
View Resource
There is no cure for Alzheimer’s disease. Although a few drugs manage temporarily certain cognitive symptoms of the illness, none can stop or meaningfully slow its progression. “We really don’t have much to offer people,” says Shannon Macauley, a neuroscientist at Wake Forest School of Medicine. Virtually all new treatments have failed in clinical trials. But new research is looking beyond drugs to see what relief might come from a simple LED light and a speaker. Bathing patients in flashing light and pulsing sounds both tuned to a frequency of 40 hertz might reverse key signs of Alzheimer’s in the brain, according to a paper published in Cell on Thursday. “I think it’s an absolutely fascinating paper to be honest,” says Macauley, who was not involved in this work. “It’s a very provocative idea. It’s noninvasive and easy and low cost, potentially, so if it were to come to fruition in humans—that’s fabulous.” Still, all this is a big if, Macauley acknowledges. The work was done in mice with genetic alterations that doomed them to develop key symptoms and pathology of Alzheimer’s disease. One batch of mice formed neurofibrillary tangles inside their neurons—dysfunctional knots of a protein called tau that can lead to the cell’s death. Another batch of the mice developed amyloid beta plaques—sticky heaps of protein that dam the flow of communication between neurons. All the mice also had a third hallmark of the disease—irregular brain activity in the gamma range of brain waves that oscillate between 30 and 100 times a second. In 2015 neuroscientist Li-Huei Tsai, director at The Picower Institute for Learning and Memory at Massachusetts Institute of Technology, was working on an experiment to manipulate that brain activity by flashing a white light at these mice. Like light strobes, our brains flicker. Brain waves are generated when large groups of neurons oscillate on and off together. Neurons encode our thoughts and actions and senses in this rhythmic electrical flutter. So when Tsai tuned her light to flash 40 times a second, or 40 hertz, and flickered it at the mice, their brains flickered back—generating gamma waves at a corresponding 40 hertz. Then, something unexpected happened. When Tsai dissected the mice brains afterward, the amount of amyloid plaques and tau tangles in the mice that saw the light had plummeted. “It was the most remarkable thing,” Tsai says. “The light flicker stimulation triggers a tremendous microglia response. These are the brain’s immune cells that clear cell debris and toxic waste including amyloid. They’re impaired in Alzheimer’s disease, but [the light] seems to restore their abilities.” When Tsai dissected the mice brains afterward, the amount of amyloid plaques and tau tangles in the mice that saw the light had plummeted. “It was the most remarkable thing,” Tsai says. “The light flicker stimulation triggers a tremendous microglia response. These are the brain’s immune cells that clear cell debris and toxic waste including amyloid. They’re impaired in Alzheimer’s disease, but [the light] seems to restore their abilities.” This clearing-out process only happened in the visual cortex where the brain processes light information. To get these effects to penetrate deeper into the brain, she added a clicking sound like a dolphin’s chirrup that also had a 40-hertz frequency. When the mice sat in a room with both the flashing light and the droning sound for an hour day, seven days in a row, amyloid plaques and tau tangles began falling in not just the audio and visual cortices but the prefrontal cortex and the hippocampus as well. “This was one of the big jumps in the new paper,” Macauley says. “These are the learning and memory centers of the brain. And there was about a 40 or 50 percent decrease in amyloid and tau levels. It’s an absolutely impressive feat.” That showed when Tsai put the mice through a set of cognitive tests. In one, where the mice were given a familiar and an unfamiliar object to explore, mice that didn’t get the treatment acted as though they’d never seen the familiar object. “That shows some memory problems,” Tsai says. Mice that saw the light and heard the sound spent about two thirds of the time that untreated mice did examining the familiar object. “It was unbelievable,” Tsai says. “This is the first time we’ve seen that this noninvasive stimulation can improve cognitive function. It’s not a drug or an antibody or anything, it’s just light and sound.” One possible explanation for this is brains with Alzheimer’s have irregular, often hyperactive, neurons, says Jorge Palop, a neurologist at the University of California, San Francisco, who did not work on the study. By providing the brains with a steady and regular beat, the repeating light and sound might work as a kind of metronome for brain activity. “This could be like resetting the mice every day and correcting some of this abnormal activity that they have,” he says. “Then downstream of that are all these beneficial effects.” All of this is still at the level of speculation. Researchers simply do not know why these brain waves, specifically ones rising from light and sound stimulation at 40 hertz and no other frequencies, can lead to a reversal of Alzheimer’s disease symptoms. “That’s a mystery,” says Terrence Town, a neuroscientist, at the University of Southern California who was not involved with the work. It’s also not clear if these beneficial effects would appear or if 40 hertz is the “magic” frequency in humans, he says. Tsai is already working on answering those questions. In human studies underway at Cognito Therapeutics, a start-up she founded with her colleague Ed Boyden, she says light and sound seem to increase gamma waves in healthy participants without negative side effects. “Nobody gets sick or even complains about it,” Tsai says. “But to see a [therapeutic] effect in humans, you’ll have to wait a long time. If this approach has an impact, the experiment could easily take five years to have some conclusive answer.”
Original Source: https://www.scientificamerican.com/article/an-hour-of-light-and-sound-a-day-might-keep-alzheimers-at-bay/
Review of transcranial photobiomodulation for major depressive disorder: targeting brain metabolism, inflammation, oxidative stress, and neurogenesis
Paolo Cassano; Samuel R. Petrie; Michael R. Hamblin; Theodore A. Henderson; Dan V. Iosifescu; - Neurophotonics, 3(3), 031404 (2016). doi:10.1117/1.NPh.3.3.031404 March 4, 2016 (Publication) 4471
This study shows some of the most detailed parameters (power, wavelenght, dosage) for working with the brain and seems to be unbiased because of the diverse background of authors..
View Resource
Abstract
We examined the use of near-infrared and red radiation (photobiomodulation, PBM) for treating major depressive disorder (MDD). While still experimental, preliminary data on the use of PBM for brain disorders are promising. PBM is low-cost with potential for wide dissemination; further research on PBM is sorely needed. We found clinical and preclinical studies via PubMed search (2015), using the following keywords: “near-infrared radiation,” “NIR,” “low-level light therapy,” “low-level laser therapy,” or “LLLT” plus “depression.” We chose clinically focused studies and excluded studies involving near-infrared spectroscopy. In addition, we used PubMed to find articles that examine the link between PBM and relevant biological processes including metabolism, inflammation, oxidative stress, and neurogenesis. Studies suggest the processes aforementioned are potentially effective targets for PBM to treat depression. There is also clinical preliminary evidence suggesting the efficacy of PBM in treating MDD, and comorbid anxiety disorders, suicidal ideation, and traumatic brain injury. Based on the data collected to date, PBM appears to be a promising treatment for depression that is safe and well-tolerated. However, large randomized controlled trials are still needed to establish the safety and effectiveness of this new treatment for MDD.
1.
Introduction
Infrared (IR) light is ubiquitously present to most life on the earth. Of the total amount of solar energy reaching the human skin, 54% is IR and 30% is IR type A—near-infrared—(NIR; with a wavelength range of 760 to 1440 nm),1 which penetrates through the human skin and reaches deeply into tissue, depending on wavelength and energy.2
NIR is used to treat a variety of conditions such as muscle pain,3 wounds,4 neuropathic pain,5 and headache.6 NIR is also used for wellness and lifestyle purposes such as for cosmetic improvement in peri-orbital wrinkles.7,8 The clinical use of NIR light applied in NIR-spectroscopy dates from the mid-1980s, when it was used for monitoring of the brain in the neonate and the fetus.9
The use of transcranial phototherapy for treating brain disorders started with its application to acute stroke. Numerous preclinical animal studies1011.–12 suggested that the application of NIR laser (810 nm) to the head at various times (hours) after induction of an acute stroke had beneficial effects on subsequent neurological performance and reduced lesion size. Evidence was obtained for the anti-inflammatory, anti-apoptotic, and proneurogenesis effects in the brain stimulated by this approach.13,14 These promising animal studies led to the conduction of a series of clinical trials called NeuroThera Effectiveness and Safety Trials (NEST). All together there were three large studies conducted in 1410 stroke patients [NEST-1 (n = 120
), NEST-2 (n = 660 n = 630
)] that demonstrated that NIR light delivered transcranially with a class-IV laser is safe, with no significant differences in rates of adverse events with NIR, when compared to sham exposure.1516.–17 Other preclinical studies and clinical trials have suggested that transcranial photobiomodulation (PBM: laser or light emitting diodes—LED) is safe and effective for acute1819.20.21.–22 and chronic2324.–25 traumatic brain injury (TBI) and has beneficial effects on neurodegenerative diseases (Alzheimer’s and Parkinson’s).26,27
For the transcranial treatment of major depressive disorder (MDD), both PBM LEDs and lasers have been experimentally tested, although PBM is not FDA-approved for the treatment of MDD. Certain forms of PBM treatment are also referred to as low-level light therapy (LLLT), since it utilizes light at a low power (0.1 to 0.5 W output at the source) to avoid any heating of tissue. The irradiance of the PBM medical devices (or power density) typically ranges from 1 to 10 times the NIR irradiance from sunlight on the skin (33.6 mW / cm 2
at the zenith). However, most PBM medical devices only deliver light energy at one or two selected wavelengths, as opposed to the whole spectrum of IR that is contained in sunlight. To our knowledge and to this date, transcranial PBM treatment has not caused any retinal injury—one of the most likely postulated adverse events, although care is taken routinely in such studies to protect the eyes with goggles or eye covers.28
In this review, we will first discuss the mechanisms of action by which NIR and red light (PBM) might improve symptoms of depression, and then present the clinical evidence for their use as a treatment for MDD and other comorbid psychiatric syndromes.
2.
Methods
We found clinical and preclinical studies via PubMed search (December 15, 2015), using the following keywords: “near-infrared radiation,” “NIR,” “low-level light therapy,” “low-level laser therapy,” or “LLLT” plus “depression.” We chose studies that had a clinical focus, and we excluded studies involving NIR spectroscopy. We also located studies using the references from the articles found in the PubMed search. As the searched literature encompassed different conditions and disorders frequently comorbid with depression, a specific section of this review was devoted to the effect of PBM on psychiatric comorbidity. In the latter section, the following conditions were included, based on available literature: TBI, anxiety and post-traumatic stress syndromes, insomnia, and suicidal ideation. The literature search for the use of PBM to treat comorbid conditions was neither systematic nor extensive, but rather a secondary focus of this review. The information is presented in an organized fashion to allow the reader to easily grasp the potential applications of PBM for the treatment of depression and of its comorbid conditions. To attain this goal, the authors have allowed a margin of redundancy, by distributing different information derived from any given publication in separate sections of this review. To avoid an artificial inflation of the extant literature on the chosen topic, we referenced the main authors—and when appropriate their affiliation—when referring to the same articles more than once. The reader will find a table summarizing the six key clinical articles reviewed, also to avoid unintended inflation of the literature. The six clinical reports included in this review where extracted from a pool of 58 articles, that were originally identified with the literature search.
In addition, we used PubMed to find articles that examined the link between PBM and each of the various biological processes including metabolism, inflammation, oxidative stress, and neurogenesis.
3.
Targeting Brain Metabolism
Multiple studies have reported regional and global hypometabolism in MDD, which could be related (either causally or consequentially) to the neurobiology of mood disorders.2930.31.–32 Positron emission tomography studies have shown abnormalities in glucose consumption rates and in blood flow in several brain regions of subjects with major depression.33 Moreover, metabolic abnormalities in the anterior cingulate, the amygdala-hippocampus complex, the dorsolateral prefrontal cortex (DLPFC), and inferior parietal cortex seem to improve after antidepressant treatment or after recovery.3435.–36
With phosphorus magnetic resonance spectroscopy (P 31 - MRS
), the baseline pool of nucleotide triphosphate (NTP)—a product of the cellular utilization of glucose and a marker of the cellular energy availability—was low in subjects who subsequently responded to antidepressant treatment.32 Iosifescu et al.32 also demonstrated for the first time with P 31 - MRS P 31 - MRS
metabolite changes were noted in brain-only voxels of responders, but not in nonresponders to antidepressants.
In experimental and animal models, PBM (NIR and red light) noninvasively delivers energy to the cytochrome c oxidase and by stimulating the mitochondrial respiratory chain leads to increased ATP production (see Fig. 1).3738.–39 A study of the effects of NIR on patients with MDD found that a single session of NIR led to a marginally significant increase in regional cerebral blood flow.40 Whether the observed changes in cerebral blood flow resulted from fundamental changes in neuronal metabolism or changes in vascular tone remain to be clarified. Given the correlation of both hypometabolism and abnormal cerebral blood flow with MDD, the beneficial effect of NIR on brain metabolism is one potential mechanism for its antidepressant effect.
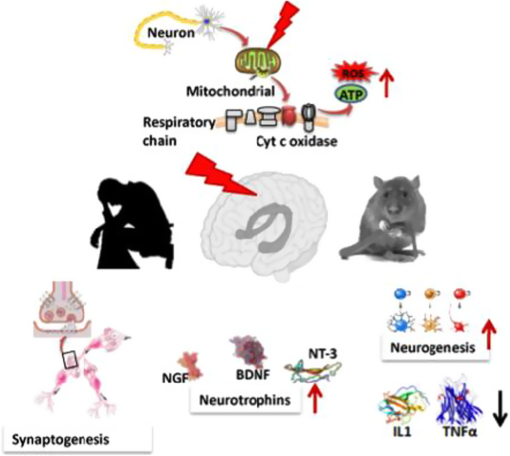
Fig. 1
Cellular targets of NIR radiation mechanisms of transcranial NIR for psychiatric disease. The NIR photons are absorbed by cytochrome c oxidase in the mitochondrial respiratory chain. This mitochondrial stimulation increases production of ATP but also activates signaling pathways by a brief burst of ROS. This signaling activates antioxidant defenses reducing overall oxidative stress. Proinflammatory cytokines and neuroinflammation are reduced. Neurotrophins such as brain-derived neurotrophic factor are upregulated, which in turn activate synaptogenesis (formation of new connections between existing neurons) and neurogenesis (formation of new neurons from neural stem cells).
4.
Targeting Inflammation
Animal and clinical research suggests that the inflammatory arm of the immune system contributes to MDD. Post-mortem gene expression profiling on tissue samples from Brodmann area 10 (BA10—prefrontal cortex) have shown that MDD is characterized by increased inflammation and apoptosis.41 In a case-control study, Simon et al.42 found that antidepressant-naive MDD subjects had significant elevations in the following cytokines and chemokines when compared to healthy controls: MIP - 1 α
, IL - 1 α IL - 1 β IFN γ
. Although IL-10 is an anti-inflammatory cytokine, the results suggested that the elevated levels of this IL-10 were likely induced in response to the overall elevation of proinflammatory cytokine levels. In a review of the research on inflammation in MDD, Raison et al.43 proposed that proinflammatory cytokines might cause brain abnormalities that are characteristic of MDD. Indeed, animal research has shown that IL-1 mediates chronic depression in mice by suppressing hippocampal neurogenesis.44
One proinflammatory cytokine that may be of particular relevance to depression is CSF IL-6 (IL6 measured in cerebrospinal fluid). In a recent report, patients with MDD had significantly higher CSF IL-6 levels compared to healthy controls; CSF IL-6 levels were significantly higher than in the serum, and there was no significant correlation between CSF and serum IL-6 levels.45 These findings are consistent with a prior report showing a positive correlation between CSF IL-6 levels and the severity of depression and suicide attempts, with the strongest correlation found in violent suicide attempters.46 One report in a smaller sample of depressed patients has shown that CSF IL-647 was lower or comparable to healthy controls.
NIR light and red light (600 to 1600 nm) decreased synovial IL-6 gene expression (decreased mRNA levels) in a rat model of rheumatoid arthritis.48 In another study, NIR (810 nm) used as a treatment for pain in patients with rheumatoid arthritis decreased production of the following proinflammatory cytokines: TNF - α
, IL - 1 β
, and IL-8.49 Khuman et al.50 showed that transcranial NIR improved cognitive function and reduced neuroinflammation as measured by Iba1+ activated microglia in brain sections from mice that had suffered a TBI. Finally, NIR (970 nm) has been found to be an effective treatment for inflammatory-type acne.51 In summary, it is reasonable to predict that transcranial NIR treatment would likewise have an anti-inflammatory effect in patients suffering from MDD.
5.
Targeting Oxidative Stress
Research has demonstrated a correlation between MDD and vulnerability to oxidative stress.52 For example, depression-induced rats show a significant decrease in glutathione peroxidase (GSH-Px) activity in the cortex.53 Glutathione (GSH) is the most abundant and one of the important antioxidants in the brain; GSH-Px enzymes protect against oxidative stress via reducing hydroperoxides and scavenging free radicals.54 GSH also appears reduced in the brains of MDD subjects.55 Additionally, a study by Sarandol et al.52 demonstrated that MDD patients have higher levels of malondialdehyde, a toxic molecule and a biomarker of oxidative stress.56 Moreover, depressed patients have more red blood cell (RBC) oxidation compared to healthy controls.52 In the same study, the authors found a significant positive correlation between RBC superoxide dismutase (SOD) activity and depression severity. SOD serves to catalyze the removal of the toxic superoxide radical.57 Thus, elevated SOD activity in depressed patients might indicate higher levels of oxidative stress. Finally, catalase activity and nitric oxide (NO) levels have also been shown to be lower in depressed patients than in healthy controls.58 Catalase is an enzyme that protects cells against damaging reactive oxygen species (ROS) via degradation of hydrogen peroxide to water and oxygen.59 NO has protective effects against cell damage, which are likely due to its pleiotropic functions in regulating antioxidant enzymes and many other aspects of cell metabolism.60,61
Oxidative stress may be an effective target for antidepressant treatments. However, successful treatments for MDD vary in regard to their protective effects against oxidative stress.52,53,62 Animal research suggests that PBM may have beneficial effects on oxidative stress. In a rat model of traumatized muscle, NIR (904 nm) blocked the release of harmful ROS and the activation of the transcription factor, nuclear factor κB (NF-κB), both induced by muscle trauma. Trauma activates NF-κB by destroying a specific protein inhibitor of NF-κB called IκB, and this destruction was inhibited by NIR light. Furthermore, NIR reduced the associated overexpression of the inducible form of nitric oxide synthase (iNOS) and reduced the production of collagen.63 This regulation of iNOS is important because excessive levels of iNOS can lead to formation of large amounts of NO that combine with superoxide radicals to form the damaging species peroxynitrite, and can interfere with the protective benefits of other forms of NO synthase.64 These findings suggest that NIR protects against oxidative stress induced by trauma. Finally, an in vitro study of the effects of red light and NIR (700 to 2000 nm) on human RBCs found that NIR significantly protected RBCs against oxidation.65
Original Source: https://www.spiedigitallibrary.org/journals/Neurophotonics/volume-3/issue-03/031404/Review-of-transcranial-photobiomodulation-for-major-depressive-disorder--targeting/10.1117/1.NPh.3.3.031404.full?SSO=1
Treatments for Traumatic Brain Injury With Emphasis on Transcranial Near-Infrared Laser Phototherapy
Larry D Morries, Paolo Cassano, Theodore A Henderson, - This article was published in Neuropsychiatric Disease and Treatment, 20 August 2015 (Publication) 4406
This exceptional research indicated prefered wavelenghts and dosages for treating patients with traumatic brain injuries. The found some surprising results.
View Resource
Abstract:
Traumatic brain injury (TBI) is a growing health concern affecting civilians and military personnel. In this review, treatments for the chronic TBI patient are discussed, including pharmaceuticals, nutraceuticals, cognitive therapy, and hyperbaric oxygen therapy. All available literature suggests a marginal benefit with prolonged treatment courses. An emerging modality of treatment is near-infrared (NIR) light, which has benefit in animal models of stroke, spinal cord injury, optic nerve injury, and TBI, and in human trials for stroke and TBI. The extant literature is confounded by variable degrees of efficacy and a bewildering array of treatment parameters. Some data indicate that diodes emitting low-level NIR energy often have failed to demonstrate therapeutic efficacy, perhaps due to failing to deliver sufficient radiant energy to the necessary depth. As part of this review, we present a retrospective case series using high-power NIR laser phototherapy with a Class IV laser to treat TBI. We demonstrate greater clinical efficacy with higher fluence, in contrast to the bimodal model of efficacy previously proposed. In ten patients with chronic TBI (average time since injury 9.3 years) given ten treatments over the course of 2 months using a high-power NIR laser (13.2 W/0.89 cm2 at 810 nm or 9 W/0.89 cm2 at 810 nm and 980 nm), symptoms of headache, sleep disturbance, cognition, mood dysregulation, anxiety, and irritability improved. Symptoms were monitored by depression scales and a novel patient diary system specifically designed for this study. NIR light in the power range of 10-15 W at 810 nm and 980 nm can safely and effectively treat chronic symptoms of TBI. The clinical benefit and effects of infrared phototherapy on mitochondrial function and secondary molecular events are discussed in the context of adequate radiant energy penetration. Keywords: infrared, traumatic brain injury, TBI, transcranial infrared light therapy, transcranial laser therapy
INTRODUCTION
Traumatic brain injury (TBI) has recently moved into the limelight due to the recognition of its impact on professional athletes and military personnel. Yet, TBI is neither a new problem nor limited to those two populations. The Centers for Disease Control and Prevention estimated that 1.5 million Americans sustained TBI annually in 2000.1 As of 2006, the estimates had risen to 1.7 million brain injuries annually.2,3 Undoubtedly, these point prevalence proportions will increase as military personnel return home,4 and the problem of repeated mild TBI (mTBI) becomes more recognized in sports.5 Current estimates of the prevalence of TBI among veterans range from 9.6%6 to 20%,7 with an estimated total of more than 300,000 cases of TBI among military personnel since 2000.4 The current estimates of the combined number of sportsrelated concussions and brain injuries in the US are 1.6-3.8 million annually.8-10 TBI results in a wide spectrum of neurological, psychiatric, cognitive, and emotional consequences. In part, the variation is related to the severity of the injury (mild, moderate, severe TBI), which is stratified based on Glasgow Coma score, periods of unconsciousness, and degrees of amnesia. Furthermore, the diversity of sequalae can be related to the areas of the brain that are injured, the severity of the injury (highly variable within the classification of “mild” and “moderate”), and the evolution of the injury over time due to neuroinflammatory processes.11,12 Additional mechanisms thought to underlie the damage of TBI include decreased mitochondrial function, calcium and magnesium dysregulation, excitotoxicity, disruption of neural networks, free radicalinduced damage, excessive nitric oxide, ischemia, and damage to the blood-brain barrier. Together, these can contribute to a progression of the damage over time. Patients with TBI can experience headache, visual disturbances, dizziness, cognitive impairment, loss of executive skills, memory impairment, fatigue, impulsivity, impaired judgment, emotional outbursts, anxiety, and depression.3,13-23 The situation can be further clouded by secondary and/ or comorbid posttraumatic stress disorder (PTSD), depression, and anxiety,17-25 which can have symptoms that overlap with those described above and appear to be increasingly likely with repetitive concussive or subconcussive brain injury.5,24,26
TREATMENTS FOR TBI
Pharmacological treatments Pharmacological treatment largely targets the neuropsychiatric sequalae of TBI, rather than providing any means of healing or repairing injury. In general, pharmacological treatment is focused on the modulation of major neurotransmitter systems – dopaminergic, serotonergic, noradrenergic, acetylcholinergic, and glutaminergic.20 Disruption of the major neurotransmitter pathways may result from direct injury or excitotoxicity and other cytotoxic mechanisms. The treatment of depression secondary to TBI is often approached with serotonin reuptake inhibitors. Several studies have examined the benefit of sertraline in post- TBI depression.27-29 Other serotonin reuptake inhibitors also have been examined. Tricyclic antidepressants appear to have some use in the treatment of post-TBI depression, although cautious dose titration is required. Patients with TBI are at greater vulnerability to sedation and cholinergic side effects of confusion and memory impairment. With serotonergic agents other than sertraline, cognitive effects also have been reported.30 Similarly, lithium may be a less desirable agent in this population due to sedation and cognitive impairment. Patients with TBI may respond at lower doses and lower blood levels than expected. Modulation of the dopaminergic system may improve alertness, attention, and cognitive processing speed. The stimulants are most commonly used for this purpose. Methylphenidate facilitates the release of dopamine and slows its reuptake. Dextroamphetamine strongly inhibits reuptake of dopamine, slows down the breakdown of dopamine by monoamine oxidase, and somewhat increases the release of dopamine. These subtle differences are sometimes imperceptible to the patient, but at other times, a patient will do best on one or the other stimulant. Increasing dopamine in the reticular activating system leads to enhanced arousal. Increasing dopamine within the frontal cortex and the striatum leads to enhanced processing speed and attention. Some evidence suggests that the stimulants may enhance neuronal recovery after injury.31-33 There are numerous potential side effects with stimulants, including abnormal heart rhythms, decreased seizure threshold, and death, but these severe side effects are extremely rare. The most common side effects with stimulants are decreased appetite, stomach upset, and headache. These are most severe at the beginning of treatment and improve over time for most patients. Insomnia is another common side effect, which may be more frequent in those with a TBI. Amantadine and bromocriptine may also increase dopamine. Studies of these agents have shown reduced abulia, anergia, and anhedonia in those with TBI.34,35 Amantadine may cause confusion, hallucinations, and hypotension. Small studies have suggested some benefits of bromocriptine in cognitive function.36,37 Arousal-enhancing agents also have found a use in the treatment of the neurocognitive sequalae of TBI. Modafinil is the oldest form of these medications, and armodafinil is an isomer of modafinil with longer activity and less side effects. These medications help to increase alertness and wakefulness. The precise mechanism of action of odafinil is unclear. It appears to increase histamine in parts of the brain involved in controlling the sleep-wake cycle; however, knock-out mice that lack histamine receptors still show increased wakefulness with modafinil.38,39 The picture is also murky for modafinil’s effect on orexins, which are wakefulness molecules in the hypothalamus.40 Modafinil has been shown to weakly bind to the dopamine transporter – like the stimulants,41 and dopamine transporter knock-out mice show no response to modafinil.42 A number of research studies have examined the benefit of these agents in fatigue associated with multiple sclerosis, TBI, cancer, and other conditions. Cognitive and memory impairments after TBI may reflect disruption of cholinergic function. The impact of anticholinergic agents on cognitive function of those with TBI supports this contention. Donepezil is the safest and most widely used of the cholinesterase inhibitors. Several easonably large studies have shown improved memory and cognitive function.43-45 Donepezil has benefits in memory and cognition even several years after injury.45,46 Anticonvulsants are often prescribed initially after a TBI due to heightened risk for seizures. Post-TBI mania or mood lability may respond well to anticonvulsants, such as carbamazepine or sodium valproate. They are also often used to treat aggression after TBI. The anticonvulsant agent, topiramate, has been shown to adversely affect cognitive function in the TBI patients.47 While insomnia is a significant issue for patients with TBI, affecting between 15% and 84% (mean of 40%),3,13,19,21,23,48,49 little has been published on the treatment of this aspect of TBI. Benzodiazepines may be effective but carry a risk of disinhibition. Kemp et al48 found that commonly used sleep aid, melatonin, was not effective. Antidepressants, including serotonin reuptake inhibitors and tricyclic antidepressants, are not effective in resolving insomnia in this population.49 No single agent has emerged as a good solution for this symptom. Cognitive rehabilitation Cognitive rehabilitation now takes many forms and is often individualized to the particular needs of the patients. Protocols have been devised to remediate cognitive difficulties often encountered in those with TBI, such as impaired concentration, executive dysfunction, inattention, visual disturbances, memory dysfunction, and impaired language function. They range from simple strategies (using a planner to aid memory and organization) to specific protocols targeting particular cognitive functions (eg, short-term memory) that can be monitored with sequential neuropsychological testing. These interventions have been extensively reviewed elsewhere.50,51 Comprehensive programs which include psychotherapy and social skills components have been shown to have greater efficacy.50,52,53 Overall, reports of benefits have been mixed.54,55 Behavioral therapies Behavioral remediation strategies to eliminate problematic behaviors following TBI have met with mixed success, most often in terms of the poor generalization of specific skills to the outside world. Behavioral deficits that create difficulties for those with TBI and their families include poor hygiene, decline in tidying/cleaning habits, social withdrawal, reduced social comprehension, impaired memory, and poor organization. Behavioral excesses that create difficulties for those with TBI and their families include aggression, sleep disruption, and perseverations. These have been reviewed elsewhere.56 Nutritional supplements Nutritional supplements, herbs, and nootropics have been utilized for many years and are increasingly popular among the patient populations. There remains little clinical research on many of these agents, perhaps reflecting a lack of funding more than a lack of efficacy. Acetyl-l-carnitine is an ester of l-carnitine and is thought to protect brain cells after injury when glucose metabolic pathways are compromised. During this period, acetyll- carnitine supports alternative ketogenic pathways for metabolism.57 It is also believed to enhance cholinergic function. While there are several clinical studies on patients with Alzheimer’s disease and preclinical data on animal models of TBI, the clinical literature on TBI remains sparse. Ginkgo biloba is a natural product of the tree by the same name. It has been shown to improve membrane fluidity and increase resistance to free-radical damage. It provides some subtle benefits to cognitive function in clinical studies of stroke, dementia, aging, and hypoxia damage.58 It has not been systematically studied in TBI but is used extensively in clinic, often in combination with meclofenoxate which is an avid scavenger of free radicals.59 S-Adenosylmethionine (SAMe) is a nutritional supplement which improves cell membrane fluidity and promotes the production of glutathione, an antioxidant. The benefit of SAMe has been assessed in a single clinical study of TBI.60 Patients receiving SAMe had a 77% improvement in clinical scores of post-concussive symptoms. Citicholine provides a source of choline which can cross the bloodbrain barrier. It has been used extensively in Europe and Japan as a treatment for TBI, stroke, and dementia. However, two large US studies failed to demonstrate significant benefit.61,62 Piracetam and the related oxiracetam and phenylpiracetam have shown some promise as nootropic agents. In one double-blind, placebo-controlled study, piracetam improved several symptoms of postconcussive syndrome, including headache and vertigo.63 More recent clinical studies have shown marginal benefit.64 Huperzine-A, an extract of Japanese club moss, is a natural acetylcholinesterase inhibitor. It may serve as a natural alternative to donepezil, rivastigmine, or galantamine. Galantamine warrants special mention as it appears to also modulate nicotinic eceptors and appears to have more persistent benefit in the treatment of Alzheimer’s disease. It appears to modulate neuroimmune responses, in addition to its effects on acetylcholinesterase.65 Cerebrolysin is a polypeptide that purportedly mimics the actions of neurotrophic factors.66,67 Studies have shown that it can reduce beta amyloid and phosphorylated tau protein accumulation. It may promote neurogenesis, synapse formation, and functional recovery.66 In animal models of acute TBI, cerebrolysin-treated rats had more surviving neurons in the area of impact and showed greater functional recovery.67 In a clinical trial of acute TBI, patients were recruited within 24 hours of injury and treated for 3 months with daily intravenous infusion of cerebrolysin. At 3 months, those receiving cerebrolysin performed significantly better on the Cognitive Abilities Screening Instrument.68 It remains unclear if cerebrolysin provides long-term nootropic benefit. The elevation of free radicals in TBI suggests that antioxidants should be beneficial. Clinical trials of pharmacological antioxidants over the past 30 years have not yielded a useful agent in acute TBI.69 Agents, such as tirilazad70 and polyethylene glycol- onjugated superoxide dismutase, have failed to show benefit in acute TBI. Omega-3 fatty acids may enhance brain repair and recovery, based on animal and clinical studies.71 Similarly, vitamin D may offer neuroprotective and restorative benefits72 in the acute TBI setting. In chronic TBI, vitamin D and omega-3 fatty acids may work synergistically, as they both may reduce neuroinflammation, apoptosis, and oxidative stress.73 Other nutritional supplements have been recommended, but prolonged therapy is necessary to possibly see benefits in TBI. A 6-month trial of ginkgo, vinpocetine, acetyl-lcarnitine, huperzine, alpha-lipoic acid, n-acetyl-cysteine, multivitamins, and over 5 g of omega-3 fatty acids daily yielded improved performance in cognitive testing and increased perfusion (function) in single-photon emission computed tomography (SPECT) scan.74 Long-term use of dietary flavanols may improve cognition in mTBI.75 Transcranial magnetic stimulation Transcranial magnetic stimulation (TMS) has shown some promise in animal models of TBI.76 However, a Cochrane review of the clinical application of TMS for depression noted no difference between repetitive TMS (rTMS) and sham rTMS using the Beck Depression Inventory (BDI) or the Hamilton Depression Rating Scale, except during the initial 2-week period.77 The application of TMS in the post-TBI patients is limited by the risk of seizure induction.78 Hyperbaric oxygen Hyperbaric oxygen treatment has been explored as a treatment for TBI.79-91 Hyperbaric oxygen therapy is neither a benign treatment, given the concerns of oxygen toxicity,79 nor a clear treatment in that the placebo condition of moderate hyperbaric room air also effectively improves cognitive function.80,81 The most carefully performed study compared a group in a cross-over design with an interval of both null treatment and hyperbaric oxygen at 100% oxygen and 1.5 atm.82 The study described improvement in many of the symptoms associated with persistent TBI including headache, tinnitus, vision disturbance, memory dysfunction, and impaired cognitive function. Cognitive testing also showed improvement in attention, information processing speed, and a battery of cognitive tests. In an uncontrolled case series of 16 subjects, Harch et al83 demonstrated that an abbreviated series of hyperbaric treatments using 100% oxygen at 1.5 atm could mitigate subjective symptoms of TBI (eg, headache, sleep disruption, irritability), improve cognitive testing scores, and improve cortical function based on SPECT imaging.83 A study of a higher dose (2.4 atm) did not reveal any significant benefit of hyperbaric oxygen therapy compared to a sham-control group treated with 1.3 atm,84 and this result has been extended and confirmed by a related group.85 However, this may reflect an inverse dose- esponse curve, rather than an absence of benefit, in that the low-dose sham group demonstrated significant changes in cognitive testing and symptom frequency.86 Hyperbaric oxygen remains a controversial area in both acute TBI86-89 and chronic TBI.82,83,85,86,90,91 Physical exercise High-energy activities and exercise programs completed through a health club facility or comprehensive rehabilitation program should focus on the same parameters of an age-adjusted and diagnosis-specific program for aerobic conditioning – flexibility, stabilization, and strength. Though it appears safe and is an accepted intervention for TBI, there is a need for further welldesigned studies.92 Exercise was a part of a 6-month study of lifestyle changes described above which yielded improved function based on cognitive testing and perfusion SPECT scans.74
A NEW TREATMENT FOR TBI
Unfortunately, little has been found to reverse the damage of TBI or repetitive concussion which is the root cause of residual cognitive and psychological impairment following TBI.20,93 One potential avenue of treatment for TBI is infrared light, which has shown promising data in a number of applications. Near-infrared (NIR) light has been investigated for its ability to modulate intracellular mechanisms related to healing. The application of NIR light by low-power laser or by light-emitting diode (LED) is also known as laser phototherapy94 or near-infrared photobiomodulation.92 NIR irradiation can facilitate wound healing,95,96 promote muscle repair,95 and stimulate angiogenesis.95,96 NIR phototherapy has been studied and applied clinically in a wide array of ailments, including skin ulcers,97 osteoarthritis,98 peripheral nerve injury,95,96 low back pain,99 myocardial infarction,100 and stem cell induction.101 The finding that NIR light passes relatively efficiently through bone has spurred interest in its application to treating disorders of the brain. Over the past decade, transcranial near-infrared light therapy (NILT)102 has been studied in animal models to understand its ability to repair damaged or dysfunctional brain tissue resulting from stroke and TBI. The first published study of NILT for TBI in humans described two cases of chronic mTBI with significant disability.103 Each patient was treated with an LED device delivering low-level low-level light therapy (LLLT) in the red and NIR range for 6-10 minutes per area daily for several months. Both patients had marked neuropsychological improvement after a minimum of 7-9 months of LLLT treatment. The precise mechanisms underlying photobiomodulation and its therapeutic benefits are not fully understood. The purported effects of NIR are illustrated in Figure 1. Light in the wavelength range of 600-1,200 nm has significant photobiomodulation capability.104 Current data most strongly support that absorption of NIR photons by cytochrome c oxidase in the mitochondrial respiratory chain is the key initiating event in photobiomodulation.95,96,104,105 This induces an increase in cytochrome c oxidase activity which in turn increases adenosine triphosphate (ATP) production. Such an increase in ATP in wounded or underperfused cells may be sufficient to activate cells in areas of injury or metabolic derangement.106 Data from numerous tissue culture and animal studies point to the importance of several secondary molecular and cellular events. For example, NIR photonic energy can modulate reactive oxygen species,95,96,102 activate mitochondrial DNA replication,95,96 increase early-response genes,95 increase growth factor expression, induce cell proliferation, and alter nitric oxide levels.95,96,102 These mechanisms are more fully described in the companion paper.105 When examined in the specific model of neural tissue injury, NIR phototherapy can lead to demonstrable neural repair and recovery. For example, LLLT of a power density of 0.9-36 J/cm2 applied at 24 hours poststroke in a rodent model yielded a 32% reduction in neurological deficits, as well as histochemical evidence of neuron proliferation and migration.106-108 LLLT had similar benefits in a rodent model of TBI.96,109-111 Interestingly, these cellular changes evolved over a period of days after light exposure and persisted for considerably longer than the interval of actual NIR exposure. These findings are consistent with a progressive regeneration cascade set in motion by the NIR light exposure. NILT in stroke NILT, predominately in the form of LLLT, has been investigated in laboratory models of stroke. LLLT applied in a single dose to an ischemic stroke model appeared to induce expression of the growth factor transforming growth factor – beta 1 and suppress the production of peroxynitrite.112 In a rat model of middle cerebral artery occlusion, LLLT at a dose of 0.5-7.5 mW/ cm2 using continuous wavelength light at 808 nm was administered at 24 hours after the acute stroke.108,113 This single application was estimated to deliver 1.8 J/cm2 in total to the cortex surface and resulted in demonstrable neurological improvement. Functional changes were not manifested until approximately 2 weeks after the single treatment. While there was no significant change in the size of the stroke lesion, histochemical evidence of neurogenesis and migrating neurons108 indicate that a cascade of secondary processes was initiated by NILT. A rabbit model of stroke utilizing injection of a blood clot embolus also demonstrated benefit from LLLT.102,114,115 Herein, 808 nm light was applied with an LED delivering 7.5 mW/cm2 and an estimated 0.9-2.6 J/cm2 to the cortical surface. Cortical ATP levels were increased, indicative of increased mitochondrial activity.114 Significant behavioral recovery was also noted; however, neither ATP increased nor neurological function changed at doses less than 0.3-0.7 J/cm2.114,115 At higher doses of 0.9-15 J/cm2, neurological improvement was seen.114,115 The clinical trials of NILT in acute stroke, the Neuro- Thera Effectiveness and Safety Trials 1, 2, and 3 (NEST- 1,-2, -3), were conducted between 2006 and 2009. The Phase II clinical trial (NEST-1) involved 120 patients in a double-blind, placebo- ontrolled study of the effects of NILT within 24 hours of ischemic stroke.116,117 Approximately 60% of the patients experienced clinical benefit, and the safety profile was very good. Thus, NEST-2, a Phase III clinical trial, was undertaken in 2007. A total of 660 patients were enrolled.118 Somewhat surprisingly, the study did not demonstrate statistical clinical improvement using a different outcome measure.119 Post hoc analysis revealed that a portion of the patients who were moderately affected and/or had strokes limited to the cerebral cortex did realize clinically and statistically significant improvement.102 The NEST-3 trial was halted midpoint when it failed to demonstrate statistical benefit on futility analysis.120 A key factor in the interpretation of the results of NEST-3 is that, different from NEST-1, all types of stroke were included as opposed to just cortical strokes. Continuous laser light has a limited depth of penetration (#1 cm into brain tissue) which likely prevents an effect on deeper brain matter. Therefore, the lack of significant benefits from NIR phototherapy in NEST-3 could be related to the fact that ischemic penumbra was not reached by the light (Luis DeTaboada, personal communication, January 2015). While pulsed NIR was not used in the NEST-3 study, it is estimated that pulsed NIR could penetrate up to 3 cm in depth from the cortical surface, therefore possibly extending the therapeutic target to deeper strokes (Luis DeTaboada, personal communication, January 2015). Figure 1 Hypothesized mechanism of action of NiR light therapy. Notes: NiR light (600-980 nm) penetrates tissue to variable depths depending on wavelength, the tissue involved, coherence, and time. A fraction of the photonic energy reaches the mitochondria and is absorbed by cytochrome c oxidase. This activates increased ATP production, increases production of ROS and RNS, and possibly increases NO. Downstream events include increased early-response genes (c-fos and c-jun) and activation of NF-?B, which in turn induces increased transcription of gene products leading to synaptogenesis, neurogenesis, and increased production of inflammatory mediators and growth factors. Abbreviations: NiR, near-infrared; ATP, adenosine triphosphate; ROS, reactive oxygen species; RNS, reactive nitrogen species; NO, nitric oxide; NF-?B, nuclear factor kappa B. NILT in TBi Oron et al109 conducted the first animal studies of NILT for TBI. They found that a single application of NIR light at 808 nm from a 200 mW emitter at 4 hours post-injury resulted in a significant reduction in lesion size by 5 days.109 To date, several groups have studied NILT in animal models, and this material has previously been reviewed.95,121-123 Single applications of 800-810 nm NIR light within 4 hours of injury have been shown to improve neurological function significantly.110,124-126 The same dose of NIR light at 6 hours was less effective125 and at 8 hours had no appreciable benefit.125 NIR photonic energy at other wavelengths was less effective. Wu et al110 examined red light (670 nm) at 4 hours and found a similar improvement in neurological function; however, 730 nm and 980 nm had no neurological benefit. Similar data for lesion volume have been reported. A single dose of 800-810 nm NIR light (fluence of 36 J/cm2) yielded an approximate 50% reduction in the volume of the lesion at 3-4 weeks110,111,124-126 and a possible reduction in the initial spread of neurological injury, based on the marked reduction in lesion volume found at 5 days post-injury.109 Repeated NIR phototherapy treatments appear to have some benefit, but the frequency and number of treatments are critical factors. While a single NIR light application had benefit, daily applications for 3 days yielded much greater neurological benefit126,127 with smaller lesion size,126 fewer degenerating neurons,126 more proliferating cells,126 and greater levels of brain-derived neurotrophic factor (BDNF)127 compared to a single treatment in a mouse model. In contrast, daily treatment for 7 days128 or 14 days126 showed no difference from controls. NIR energy densities in the range of 0.9-36 J/cm2 resulted in significant biochemical and behavioral changes.109-111,124-127 Pulsing of NIR light appears to yield a greater neurological response but only within certain parameters. Pulsing at 10 Hz yielded greater neurological improvement and a significant reduction in lesion size compared to either continuous-wave or pulsed NIR at 100 Hz.111 In the mouse model of moderate TBI, NILT (800-810 nm) improved learning and memory (Morris water maze performance),128 as well as behaviors associated with depression and anxiety (immobility during tail suspension).111,124 The finding that NILT brought about a smaller lesion in the rodent model of TBI compared to untreated mice suggests that decreased apoptosis, reduced spreading lesion penumbra, and/or neurogenesis are induced by NILT. Indeed, NILT can decrease BAX expression, a pro-apoptosis gene,129 increase expression of BCL-2, an anti-apoptosis gene,129 increase nerve growth factor,95 increase BDNF,127 decrease inflammatory markers,130 and decrease numbers of degenerating neurons.126 Together, these mechanisms may reduce the enlargement of the initial lesion during the first day following the lesion.109 Moreover, increased BDNF and nerve growth factor may contribute to synaptogenesis as shown by increased levels of synapsin-1,127 and neurogenesis, as shown by increased numbers of proliferating cells.127 In a double-blind study in healthy volunteers, NILT was beneficial – compared to sham – in memory and attention.131 In this study, the authors shed only one application of NIR light to the right forehead, targeting the right frontal pole of the cerebral cortex (Brodmann’s area 9 and 10). The device was a Class IV laser CG-5000 (Cell Gen Therapeutics, Dallas, TX, USA), and the parameters were as follows: wavelength 1,064 nm, irradiance 250 mW/cm2, fluence 60 J/cm2, and time 4 minutes per site (two sites).131 The subjects who received the NIR treatment had better attention after 2 weeks, measured by the psychomotor vigilance test. They also had better delayed visual memory at the Delayed Match-to-Sample test. This is the only published controlled trial assessing the impact of NILT on cognition; however, other reports have shown the therapeutic effects of NILT in small numbers of TBI patients. In a two-case report in TBI patients,103 NILT (870 nm) improved sustained attention, memory, and executive functions. Both patients were treated with an instrument with three separate LED cluster heads. The parameters used for the treatment were the following: NIR wavelength 870 nm and 633 nm (red light), irradiance 2.2-25.8 mW/cm2, fluence 13.3 J/cm2, and time 10 minutes per site.103 The same group reported on a cohort of eleven subjects with persistent cognitive dysfunction and treated with a similar NILT protocol for chronic mTBI.132 The eleven subjects received NILT with a device with three LED cluster heads (Model 1100; MedX Health, Toronto, ON, Canada). The parameters used for the treatment were the following: NIR wavelength 870 nm and 633 nm (red light), irradiance 22.2 mW/cm2, fluence 13 J/cm2, and approximate time 10 minutes per site. The NIR light was applied three times per week for 6 weeks (18 sessions), on eleven sites for 10 minutes per site (the total duration of each session was 20 minutes).132 The sites on the skull were chosen on the midline, and bilaterally on frontal, parietal, and temporal areas. At the follow-up neuropsychological testing, NILT had a powerful effect on attention, inhibition, and inhibition switching in the Stroop task, and similarly improved verbal learning and memory, as well as enhanced long-delay free recall on the California Verbal Learning Test. Eight subjects, from the same cohort, were identified as having mild, moderate, or severe depression based on the BDI-II total score (range: 15-34).132 The three cases, who entered the study with only mild depression, remained the same after NILT treatment. Results for the five cases with moderate-severe depression were as follows: two moderate cases improved to mild/minimal depression 8 weeks after the end of NILT series, and one severe case improved to moderate depression. Two moderate or severe depression cases remained the same after 8 weeks of follow-up from the last NILT session.132 Dose response and photonic penetration A prevailing theory in photobiomodulation postulates that a bimodal response curve exists for the biological effects of NIR light.95 The so-called Arndt-Schulz curve (a fundamental principle in homeopathic medicine) is frequently used to describe this biphasic dose response. Some data indicate that low levels of light have a much better effect on stimulating and repairing tissues than higher levels of light. Laboratory studies of cells in culture have demonstrated a bimodal dose response to light exposure in lymphocytes133 and fibroblasts.134,135 For example, Chen et al135 found that a range of 0.03-0.3 J/cm2 was beneficial in activating transcription factors in culture, while 3-30 J/cm2 inhibited the activation of these factors. In contrast, an order-of-magnitude greater dose (2 J/cm2) was best at activating fibroblasts in a superficial wound model.136 Furthermore, an order-ofmagnitude greater dose (30 J/cm2) proved to be best in a rodent joint inflammation model.137 Thus, a dosedependent effect for many biological responses to NIR light has been demonstrated,95,137-139 but the critical parameter is dose at the level of the target tissue, rather than at the surface.137,140 The amount of energy that reaches a volume of tissue at depth is determined by the attenuation of the photonic energy as it passes through the overlying tissue. For example, only 2.45% of the energy from a 980 nm laser emitter penetrates to the level of the peroneal nerve.140 Nevertheless, the biphasic dose response does not appear to be universally true. In primary microglial cell culture, a dose-dependent response to NIR was demonstrated with no detrimental effects at doses as high as 30 J/cm2.141 So a critical question in the use of NILT is that of radiant energy penetration. In particular, some authors have challenged the efficacy of low-power LEDs used in LLLT.142-144 In laboratory studies, LLLT radiant energy is almost entirely absorbed in the first 1 mm of skin.145,146 In two unrelated studies, LLLT diode devices proved to be ineffective in the treatment of diabetic neuropathy,142,144 in contrast with prior reports.147 Similarly, laboratory studies of NILT using LLLT transcranially have not consistently yielded positive results. For example, in a rat model of TBI, Giacci et al148 found no benefit from daily 30-minute irradiation with either 670 nm or 830 nm 0.5 W LED emitters for a period of 7 days. Doses at the skin surface were 28.4 J/cm2 and 22.6 J/cm2, respectively.148 Similarly, treatment of a rat model of contusive spinal cord injury with LLLT (830 nm at 22.6 J/cm2 or 670 nm at 28.4 J/cm2) for 30 minutes per day for 5 days resulted in no significant functional improvement and no reduction in lesion size, despite delivering 2.6 J/cm2 to the spinal cord.148 Lapchak102 reported that the physical parameters of NILT in the clinical trials for the treatment of stroke utilized in the NEST-1 and NEST-2 trials116-120 may have delivered insufficient energy to cortical tissues to be effective. Therein, NIR light of 808 nm wavelength with infrared energy densities of 0.9 J/cm2 was applied to the human scalp for a total of 40 minutes with applications at multiple sites during that time.116,118 Recall that animal models of both stroke and TBI suggest that NIR energy densities in the range of 0.9-36 J/cm2 resulted in significant biochemical and behavioral changes.96,106-115,125-127 The concern raised from the NEST studies102 is that current clinical trials testing the effectiveness of lowenergy NIR diodes to treat TBI may yield negative or inaccurate efficacy data, not because of a failure of infrared light to invoke a change but due to a dose error. Doses that are effective when directly applied to cells in a Petri dish149,150 or to 3-5 mm thick rodent brains96,109-111,125,126,128 may be insufficient to penetrate 2-4 cm into the human brain. In a companion paper, our own studies of photonic energy penetration are detailed.105 To summarize, the laboratory tissue studies showed that 0.5 W LED emitters did not penetrate the 2 mm thickness of human skin. No detectable energy from 0.5 W LED NIR light emitters could be detected penetrating a similar thickness (1-2 mm) of sheep skin or 3 cm thick section containing sheep skin, skull, and brain. In contrast, 11% of the photonic energy from a 10 W 810/980 nm coherent NIR laser penetrated 2 mm of human skin. Similarly, 17% of the photonic energy from a 15 W 810 nm coherent NIR laser penetrated the same distance.105 Energy from these more powerful NIR emitters could be detected penetrating 3 cm of sheep skin, skull, and brain with 0.4% of the 10 W 810/980 nm NIR laser’s energy reaching the depth of 3 cm and 2.9% of the 15 W 810 nm NIR laser’s energy traversing the same distance.105 Anders also has demonstrated penetration of 808 nm light to 40 mm in the brain using a 5 W laser emitter (JJ Anders, personal communication, January 2015). Prompted by the mixed results in the literature and the observations by Lapchak,102 Franzen-Korzendorfer et al,144 Wan et al151 and Lavery et al142 we have been utilizing relatively high-power (10- 5 W) lasers at the wavelengths of 810 nm and 980 nm in the clinic to treat patients with TBI. Clinically, the patients have shown excellent responses with resolution of many of their long-standing symptoms of TBI or post-concussive syndrome. Below is a retrospective series of such patients to illustrate the extent and character of response to this modality. Methods Patients in the case series were sequentially treated patients at a clinic which is engaged in ongoing NILT for a number of clinical conditions. The risks, benefits, and current state of research on the use of NILT were explained to each patient. Each patient consented to treatment. Institutional Review Board approval was obtained in a post hoc review, noting that the risk-benefit ratio was acceptable. Between March 16, 2011 and February 20, 2013, sequential new referrals for chronic mild-to-moderate TBI were evaluated for treatment and selected for NILT using Class IV lasers, either the LT1000 (LiteCure, Newark, DE, USA), a 10 W adjustable NIR laser emitter with wavelengths of 810/980 nm capable of delivering continuous or pulsed NIR light, or the Diowave 810 (Diowave, Riviera Beach, FL, USA), an adjustable NIR emitter up to 15 W with a wavelength of 810 nm capable of delivering continuous or pulsed NIR energy. Demographics and laser treatment settings are detailed in Table 1. The fluence delivered to the skin of patients ranged from 55 J/cm2 to 81 J/cm2. No other treatment modalities (medications, exercise regimen, supplements) were added, discontinued, or changed while receiving NILT. Symptoms were monitored clinically. A baseline Quick Inventory of Depressive Symptomatology Self-Report (QIDS-SR)152 was completed for all patients, and the BDI153 was administered to seven of the ten patients before and after the course of treatment. In addition, each patient was instructed on how to create and maintain a patient and spousal diary of symptoms and subjective progress. Each of six patients received a single series of ten treatments with the LT1000 Class IV laser. Three additional patients each received a single series of 20 treatments with the LT1000 Class IV laser. One patient was treated with the Diowave 810 nm Class IV laser device in a series of 20 treatments. The patients and treating clinician wore protective eyewear. There were no incidents of burns or thermal discomfort (Figure 2). The impact of high-watt NILT While the patient group represented a diverse mix (Table 1 presents demographics), some notable commonalities of symptoms emerged. Over 90% of the patients had complaints of anxiety, depression, irritability, and insomnia. Other symptoms included headache (60%), suicidal ideation (50%), cognitive difficulties (50%), attention problems (50%), short-term memory problems (40%), loss of libido (30%), substance abuse (20%), fatigue (20%), and panic attacks (20%). Six of the patients were unemployed prior to treatment. Three of the patients were experiencing severe marital difficulties. All carried or had a confirmed diagnosis of TBI, but other comorbid diagnoses included PTSD, major depressive disorder, generalized anxiety disorder, bipolar disorder, and attention deficit/hyperactivity disorder. The patients’ baseline scores on the BDI were 25.3±12.1 (moderate depression range), and baseline scores on the QIDS-SR were 12.9±4.6 (moderate depression range). During NILT treatments, skin temperature increased no more than 3°C with rapid cooling after removal of the NIR light. A continuous sweeping motion was utilized to minimize skin heating and cover a larger area. After a course of ten treatments of NILT (20 treatments in four patients), each patient experienced significant clinical improvement with resolution of many of their symptoms (Table 2). In addition, the BDI scores dropped to 12±6.5 (nondepressed range). This represented a significant decrease (P,0.01, Student’s t-test, one-tailed, Microsoft Excel). The QIDS-SR scores after treatment were 2.2±2.3 (nondepressed range), and the difference from baseline was highly significant (P,0.00001, Student’s t-test, one-tailed). Patients noted improvement in cognitive function, mood, anxiety, and sleep. None of the patients continued to have suicidal thoughts (50% at baseline). Other symptoms, such as anxiety and irritability, were markedly improved. Most notable were the nonquantifiable changes in patients’ lives. Patients reported improved cognitive ability and a desire to return to meaningful work. Five of the six unemployed patients have returned to work. The two patients who were Iraq/ Afghanistan veterans have found new careers in highly skilled trades. The patients with marital difficulties have reconciled and were purchasing homes or otherwise solidifying their marriages. The clinical change can be attributed to NILT because no changes in medications, supplements, or exercise regimen were permitted during the course of NILT treatment. All patients in the case series experienced significant clinical improvement which supports the conjecture that high-power NIR laser delivers sufficient energy to the human brain for photobiomodulation to occur. Insomnia and suicidal ideation, common symptoms in those with TBI or post-concussive syndromes,3,17-20,24,25 resolved in 100% of cases. Headache, another common symptom for patients following a TBI,6,14,15,23 was reduced or resolved in the six patients so afflicted. Symptoms such as anxiety,14,15,21,24 depression,21,24,25,27-29 and irritability resolved or were dramatically reduced in all patients. Cognitive function appeared to improve based on return to work or improved work performance, although cognitive tests were not performed. The quality of life dramatically improved in all cases, based on the observations of the patients, their family members, and the treating clinician. At follow-up intervals of 6-7 months post-treatment, patients have reported continued improvements in symptoms. The precise areas of brain injury were not elucidated in Figure 2 Treatment parameters per individual, based on area of the skull treated. Notes: Dimensions varied per head/skull size and hair line. Treatment was warm and comfortable for each patient. There were no incidences of discomfort. Areas treated were (A) temporal- ilateral, (B) frontal, and in patients 1-3, 5, and 6 (B) frontal only. Table 1: Infrared light treatment parameters for each of the ten patients in the case series Patient Area treated Sex Mechanism of TBI Interval since TBI Wavelength of Dosage per area Duration before treatment NIR-PT dual wave Scanning technique per area pulsed 10 Hz 1 B, bilateral frontal Male Concussive blast 2 years 810 and 980 nm 2,700 J 10 minutes Fluence – 20.45 J/cm2 2 areas Area – 132 cm2 10 visits 2 B, bilateral frontal Female MVA 18 years 810 and 980 nm 2,400 J 9 minutes Fluence – 18 J/cm2 2 areas Area – 133 cm2 10 visits 3 B, bilateral frontal Female MVA 5 years 810 and 980 nm 2,400 J 8 minutes Abuse Fluence – 18.3 J/ cm2 2 areas Area – 131 cm2 10 visits 4 A–B, bilateral frontal, left temporal Female MVA x2 8 years and 13 years 810 and 980 nm 2,400 J 8 minutes Fluence – 18.3 J/cm2 3 areas Area – 131 cm2 10 visits 5 B, bilateral frontal Male Vietnam Veteran 20+ years 810 and 980 nm 3,000 J 10 minutes Concussion Fluence – 28.3 J/cm2 2 areas Child abuse Area – 106 cm2 10 visits 6 B, bilateral frontal Male Concussion 5+ years 810 and 980 nm 2,400 J 12 minutes Fluence – 14.8 J/cm2 2 areas Area – 162 cm2 10 visits 7 B–A, bilateral frontal, left temporal Male Afghanistan, Iraqi Disability 810 and 980 nm 3,000 J 10 minutes Disability due to TBI 2 years Fluence – 22.7 J/cm2 3 areas Area – 132 cm2 20 visits B–A, bilateral frontal, bilateral temporal Female Hypoxic encephalopathy Childbirth-related 810 and 980 nm 2,700 J 9 minutes injury, 8 years Fluence – 27.8 J/cm2 3 areas Area – 97 cm2 20 visits 9 B–A, bilateral frontal, bilateral temporal Male MVA-TBI Numerous episodes 810 and 980 nm 3,000 J 10 minutes Concussions Fluence – 22.72 J/cm2 3 areas Area – 132 cm2 20 visits 10 B–A, bilateral frontal, left temporal Female Bicycle vs car >30 days 810 nm single 2,700 J 9 minutes Concussion, amnesia, LOC wavelength – Fluence – 17.1 J/cm2 3 areas different device Area – 158 cm2 20 visits Note: All safety precautions were followed, including metal protective eyewear (laser eye protection). Abbreviation: LOC, loss of consciousness; MvA, motor vehicle accident; TBi, traumatic brain injury. the majority of these cases, so a correlation of symptoms changes and cortical function changes cannot be made; however, perfusion SPECT imaging in other patients has shown significant increases in perfusion in injured areas of the brain and overall improved cortical function following similar courses of high-watt NILT.154 One concern that has been expressed about high-watt NIR lasers is the risk of tissue heating.155 We explored this issue in our companion paper on NIR penetration.105 Temperature change was 1°C-3°C at the skin surface using continuous-wave NIR lasers in the range of 10-15 W. Using pulsed settings, the high-powered lasers showed no significant temperature change in tissue samples. The temperature change on human skin was 1°C or less in the in vivo penetration studies while maintaining continuous movement of the laser probe head.105 Clinically, patients in this case series reported only slight warming of the skin, but no discomfort, using the continuous motion technique. Laboratory studies have largely focused on treatment of acute brain injury. The processes involved in the benefits of NIR light in chronic TBI as seen in this clinical case series may be quite distinct. Nevertheless, Schiffer et al156 found that a single application of LLLT at 810 nm and 250 mW to the forehead over 8 minutes reduced depression and anxiety symptoms in ten patients for approximately 2 weeks. Similarly, the small case series by Naeser et al103 demonstrated some benefit using NIR light, albeit at very low power levels over a prolonged course of several months with only transient benefit. Together with our clinical data, these findings suggest that at least some of the photobiomodulatory effects of NIR energy likely do occur in chronic neurological conditions. Prior presentations on NILT for the treatment of TBI or stroke in humans have focused on getting photonic energy through the skull to the cortex surface which traverses a distance of about 6-10 mm; however, this model is flawed in that the distance to the areas of damage may be far greater. In other words, the cortex immediately subjacent to a portion of the skull may be 10 mm from the surface, but the NIR light energy may need to penetrate 3-7 cm to reach areas of damage. Much of the cortical surface is actually lining the walls and floors of sulci, rather than immediately subjacent to the skull. Analysis of NIR spectroscopy reveals that light propagation through varying media with irregular boundaries is subject to high levels of scatter.157 In addition, review of the neuroimaging literature on TBI has revealed that the most common areas injured in TBI are the orbitofrontal cortex (at the ventral surface of the frontal lobe) and the anterior and medial temporal lobes.158 It is not anatomically possible to position an NIR light emitter immediately exterior to the skull overlying these areas. Indeed, the orbitofrontal cortex positioned immediately above the eyes can only be reached from the forehead by angling the light emitter. Similarly, the temporal lobes are separated from the surface by epidermis, dermis, subcutaneous fat, subcutaneous blood vessels, accessory head of the temporalis muscle, connective tissue, temporalis muscle, skull, and dura mater.159 Each of these structures has different absorption and refraction properties, and each interface between different materials also creates a barrier to transmission of photonic energy.157 Blood flowing in the subcutaneous vessels is believed to create a unique barrier to transmission.160 In summary, effectively targeting the areas most commonly injured in TBI with sufficient photonic energy to initiate reparative processes represents a significant challenge in NILT. This appears to have been overcome with the high-power laser protocol presented here and in a related paper.154 As yet, the mechanism of action of NILT in treating TBI is not entirely clear. Moreover, the neurological benefits are not immediately apparent. Rather, a delay of 1-4 weeks was noted, consistent with a progressive regeneration cascade set in motion by the NILT.96,103,105 ,107,109,121,122,124,127,135 Similarly, most of the patients in the present case series did not notice benefits immediately or within the first few treatments. Instead, they reported benefits emerging over an interval of weeks, and in some cases, continuing after completion of the course of NILT. In addition, the clinical improvement reported by the patients in the above case series is more profound than that reported by patients treated with LLLT or low-powered lasers.103 In fact, we observed that among seven subjects with documented moderate depression, per BDI scores, four had an antidepressant response (≥50% decrease of depression severity). In contrast, Naeser et al132 reported that out of eight subjects with TBI and comorbid depression, only three had a significant improvement in their depressive symptoms (37.5%). Our results may be due to the greater penetration of more powerful, coherent, and pulsed NIR light from a laser source. A unique outcome measure was developed for this protocol (Morries and Henderson, unpublished data, 2015). A patient diary and separate spousal diary provided a weekly update of patient’s response in his or her home environment. This novel approach to capturing the patient treatment experience provided the patient and family with tangible and pertinent documentation of the clinical response. While time consuming, the experiences recorded in these diaries proved to be valuable clinical tools to the treating clinicians.
CONCLUSION
To date, there has been little progress in developing effective treatments for chronic mild-to-moderate TBI or repetitive concussions. This area of need has become even more pressing with the return of veterans from military conflicts in Iraq and Afghanistan4,6,7,16,17,19,161 and the recognition of the magnitude of sport-related TBI.5,8-10 In addition, the dramatic growth in the geriatric population with attendant proprioceptive dysfunction has resulted in a rising incidence of fall-related TBI.162 NILT has shown promise as a tool for the treatment of TBI. A critical issue is to assure that adequate photonic energy reaches the injured areas of the brain. The use of high-wattage lasers, as we have demonstrated, results in marked clinical improvement in patients with chronic TBI. Moreover, symptoms consistent with PTSD, anxiety, and/or depression also improved considerably or resolved in this group of patients. Further work in the use of highwattage NILT in the treatment of TBI, depression, and other neurological disorders is encouraged.
ACKNOWLEDGMENTS
The authors would like to acknowledge the technical assistance of Mr Charles Vorwaller (Aspen Lasers) and Lite Cure Corporation. The authors also acknowledge the contribution of Ms. Taylor Tuteur in the artistic creation of Figure 1.
DISCLOSURE
Dr. Larry D Morries is the CEO of Neuro-Laser Foundation, a nonprofit foundation. He has a private practice in Lakewood, CO. Theodore A Henderson is the president of The Synaptic Space, a medical consulting firm. He is Table 2 NiLT case series with demographics, symptoms, and treatment response
PRETREATMENT POSTTREATMENT
Patient # Sex Occupation Mechanism of TBI Diagnoses Sleep Symptoms Suicidal BDI Sleep Symptoms Suicidal BDI 1 M Veteran, Blast – 5 years; TBI, PTSD, MDD Primary and H, S, I, D, X, L, A, M, + – Resolved None, back No – unemployed Iraqi middle C, SL with spouse, insomnia working 2 F Nurse, MVA – 8 years TBI, PTSD Middle and H, F, I, X, C, A, STM, L, + 18 Resolved A and HA – No 15 unemployed terminal HA, SL but mild, insomnia return to work 3 F Unemployed Assault and TBI, PTSD, MDD, Primary and D, X, P, M, L, HA, S, + 23 Resolved HA – mild, No – MVA, 5 years GAD, ADHD middle insomnia, SA, C, N, STM back with Prior nightmares spouse, no SA, working 4 F Unemployed MVA – 3 years, TBI, PTSD, MDD Primary and D, X, HA, I, M, SA, S, N + 23 Resolved None, marriage No 17 assault middle insomnia, improved, numerous violent nightmares no SA, working 5 M Veteran, Blast – 20+ years TBI, MDD, GAD Primary and D, X, I, S, SL + 18 Resolved None No 1 unemployed 1960s; Vietnam middle insomnia 6 M executive Trauma – TBI, GAD, MDD Primary D, X, I, P, HA, A, S – – Resolved HA, X, and P – No – chronic insomnia but improved 7 M Veteran, Multiple blasts TBI, MDD, GAD Primary and S, D, I, X, C, A, S, STM, – 22 Resolved HA and C – No 16 disability (>12); Afghan middle HA mild, new and Iraqi wars insomnia career 8 F Student Childbirth TBI, learning Primary D, I, X, C, A, SL, F, STM – 16 Resolved, STM improved, No 7 disorder insomnia no bads reading .20% dream more animated 9 F Sales MVA and TBI, LOC Primary and HA, SL, N, D, I, X, H, A – 29 Resolved Mild HA, No 9 sports TBI middle insomnia, job nightmares promotion 10 F Physicist Recent car– TBI, LOC, amnesia Primary and D, I, X, neck, knee pain – 51 Resolved No loss No 19 bicycle middle of skills, accident insomnia maintain intellectual job Notes: Demographics for each of the ten patients in this case study is presented. Also presented is their history of mechanism of injury, diagnosis, and related symptoms. Changes in anxiety levels, sleep patterns, depression, and suicidal ideation were important symptoms and outcomes to track. Patients were instructed for no medication changes, with their primary treatment provider’s approval. Cognitive difficulties, attention problems, and short-term memory difficulties were by patient interpretation of their symptomatic improvement and patient diary changes. Symptom occurrence % was as follows: Anxiety – 100%, Depression – 90%, Irritability – 90%, Primary And Middle Insomnia – 90%, Headache – 60%, Sadness – 60%, Suicidal Ideation – 50%, Cognitive Difficulties – 50%, Attention Problems – 50%, Short-Term Memory Problems – 40%, Marital Difficulties – 30%, Loss Of Libido – 30%, Substance Abuse – 20%, Fatigue – 20%, Panic Attacks – 20%. Abbreviations: NILT: Near-Infrared Light Therapy, TBI: Traumatic Brain Injury, PTSD: Post-traumatic Stress Disorder, MDD: Major Depressive Disorder, GAD: General Anxiety Disorder, ADHD: Attention Deficit/Hyperactivity Disorder, H: Hyperarousal, S: Sadness, I: Irritability, D: Depression, X: Anxiety, L: Loss Of Libido, A: Attention Problems, M: Marital Difficulties, C: Cognitive Problems, SL: Sleep Issues, F: Fatigue, STM: Short- erm Memory Problems, HA: Headache, P: Panic Attacks, SA: Substance Abuse, N: Nightmares, BDI: Beck Depression Inventory, LOC: Loss of Consciousness, MVA: Motor Vehicle Accident. the president of Dr. Theodore Henderson, Inc., a clinical service firm. He is the co-owner of Neuro-Luminance, a clinical service organization. He is the president of the International Society of Applied Neuroimaging. He is the CFO of the Neuro-Laser Foundation, a nonprofit foundation. Dr. Paolo Cassano received funding from the Brain and Behavior Research Foundation; Photothera Inc and from the Dupont Warren Fellowship (Harvard Medical School) to conduct research on NIR light for the treatment of major depressive disorder.
ABOUT THE AUTHORS:
Larry D. Morries, DC brings a distinguished 30-year career studying and treating the brain and body through his private practice based in Lakewood, Colorado. As Neuro-Laser Foundation’s co-founder, his chiropractic expertise is complemented with extensive study of near infrared-light therapy applications, clinical radiology, clinical neurology and sports injury and rehabilitation. In practice since 1973, Dr. Morries has contributed extensively to both chiropractic and medical professions throughout his career. He is a recognized expert often called upon for review services, treatment utilizations, and documentation presentations. In recent years, he has guided the Colorado State of Colorado Workers Compensation Board with a review of treatment guidelines for Chronic Pain, and Complex Regional Pain Syndrome, Shoulder Pain, Low Back Pain, Traumatic Brain Injury, and was asked to present in 2016 on Thoracic Outlet Syndrome.
Other professional involvement include:
• Colorado Chiropractic Association, Board member, President in 1982, Chairman in 1984
• Colorado Chiropractic Society, Vice President and Secretary in 1995-2004
• Colorado Chiropractic Journal Club, Chairman,since 2008
Dr. Morries has continued his study of the human body and brain with postgraduate work in Neurodiagnostic testing at the American Academy of Neurology, and Harvard Medical School-Massachusetts General Hospital. He is also educated on Spinal Mechanics at Chicago Rehabilitation Institute. He earned his Doctorate in Chiropractic from Logan Chiropractic College, with recognition as Student Clinical Director, Teaching Assistant in Radiology. Dr. Morries is most proud of his research papers and awards, in America Academy of Pain Medicine, Sciatic and Suprascapular Nerve Blocks with Dr. Steve Gulevich, MD. He was asked to share two Poster presentations at the North American Laser Foundation in 2011on Low Back Pain, plus Polyneuropathy treatment with Laser (NIR) therapy. His Podium Presentation and publication on Hip dysplasia, in American Board of Chiropractic Sports Physicians®. Additionally, he has given presentations abroad at State of Chiropractic Research, Foundation of Chiropractic Education and Research, in Bournemouth England and Vancouver, BC, Canada. Dr. Theodore Henderson has extensive training and experience to the practice of Psychiatry. He trained in Psychiatry at the prestigious Barnes/Jewish Hospitals at Washington University in St. Louis. Dr. Henderson completed a fellowship in Child & Adolescent Psychiatry at the University of Colorado. He also has training in Radiology, Nuclear Medicine, and the genetics of psychiatry. He established his private practice in Centennial Colorado in July of 2000. Dr. Henderson brings a unique blend of expertise in psychopharmacology, neurobiology, and an understanding of human nature to the practice of psychiatry. Dr. Henderson attended medical school at Saint Louis University School of Medicine. While in medical school, he began studying heart pathology under Dr. Vernon Fischer. He earned an American Heart Association Medical Student Research Fellowship. With this fellowship, he spent one year at the University of Washington studying the pathology of atherosclerosis. In 1991, Dr. Henderson founded the Child Abuse Prevention Task Force at Saint Louis University. This program taught children, parents, and teachers about child sexual abuse and how to prevent it. Each year, this program reached over 8,000 children throughout the metro St. Louis area, primarily in the poor inner-city schools. The program was awarded numerous awards, including a Saint Louis University Community Service Award, Commendations from the school districts, and an award from the American Medical Student Association. Dr. Henderson was nominated for a Student Life Leadership Award and earned a Departmental Award from the Department of Community and Family Medicine. He also received a Weis Humanitarian Award recognizing outstanding humanitarian care as a medical student. Dr. Henderson wrote a training manual on this program that was implemented at other medical schools and he cowrote a book chapter in the book, A Parent’s & Teacher’s Handbook on Identifying and Preventing Child Abuse (1998). During graduate school and medical school, Dr. Henderson published numerous research studies. He published 9 articles and 27 abstracts about his research in brain development. He also published a book chapter on brain development in collaboration with his research professor, Dr. Mark Jacquin. His research focused on the role of neural growth factors and impulse activity on the development of brain organization. He collaborated with leading researchers, including Drs. Thomas Woolsey, Eugene Johnson, and Thomas Rhoades. While a medical student, Dr. Henderson wrote two research grants (as part of program project grants). Both were funded. He continued conducting research at Saint Louis University and Washington University throughout his residencies. Dr. Henderson trained for one year in Radiology, focusing on neuroimaging and pediatrics. With this strong base, he then undertook a residency in Psychiatry at Washington University’s program at Barnes/Jewish Hospitals in St. Louis. His residency included extended training in general pediatrics at St. Louis Children’s Hospital. In 1997, He was awarded the National Institute of Mental Health Outstanding Resident Award for his ongoing work in child abuse prevention and his neurobiological research while a resident. Dr. Henderson completed a residency in Adult (or General) Psychiatry and then undertook a fellowship in Child Psychiatry at the University of Colorado. This included additional specialization in Autism and Autism Spectrum Disorders. He compl
Photobiomodulation for Traumatic Brain Injury and Stroke
Michael R Hamblin - J Neurosci Res. Author manuscript; available in PMC 2018 Oct 1. (Publication) 4533
This study compare wavelength and pulsing frequencies to find the highest efficacy. It shows how much better 810nm (fig 3)and 10Hz (fig 4) are superior for TBI.
View Resource
There is a notable lack of therapeutic alternatives for what is fast becoming a global epidemic of traumatic brain injury (TBI). Photobiomodulation (PBM) employs red or near-infrared (NIR) light (600-1100nm) to stimulate healing, protect tissue from dying, increase mitochondrial function, improve blood flow and tissue oxygenation. PBM can also act to reduce swelling, increase antioxidants, decrease inflammation, protect against apoptosis, and modulate microglial activation state. All these mechanisms of action strongly suggest that PBM delivered to the head should be beneficial in cases of both acute and chronic TBI. Most reports have used NIR light either from lasers or from light-emitting diodes (LEDs). Many studies in small animal models of acute TBI have found positive effects on neurological function, learning and memory, and reduced inflammation and cell death, in the brain. There is evidence that PBM can help the brain to repair itself by stimulating neurogenesis, upregulating BDNF synthesis, and encouraging synaptogenesis. In healthy human volunteers (including students and healthy elderly women) PBM has been shown to increase regional cerebral blood flow, tissue oxygenation and improve memory, mood and cognitive function. Clinical studies have been conducted in patients suffering from the chronic effects of TBI. There have been reports of improvements in executive function, working memory, and improved sleep. Functional magnetic resonance imaging has shown modulation of activation in intrinsic brain networks likely to be damaged in TBI (default mode network and salience network).
Keywords: photobiomodulation, low-level laser therapy, traumatic brain injury, stroke, chromophores, animal studies, clinical trials, human studies
1. Introduction
Photobiomodulation (PBM) formerly known as low-level laser (light) therapy (LLLT) is approaching its 50th anniversary, after being discovered by Endre Mester working in Hungary in 1967 (Hamblin et al. 2016). Originally thought to be a property of red lasers (600-700 nm), PBM has broadened to include near-infrared (NIR) wavelengths 760-1200 nm, and even blue and green wavelengths. Moreover the advent of inexpensive and safe light emitting diodes (LEDs) has supplanted the use of expensive lasers in many indications. The better tissue penetration properties of NIR light, together with its good efficacy, has made it the most popular wavelength range overall. The best-known medical applications of PBM have been for indications such as stimulation of wound healing (Hopkins et al. 2004; Kovacs et al. 1974), reduction of pain and inflammation in orthopedic and musculoskeletal conditions (Aimbire et al. 2006; Gam et al. 1993), and mitigation of cancer therapy side-effects (Zecha et al. 2016a; Zecha et al. 2016b). However in recent years there has been growing interest in the use of PBM in various brain disorders (Hamblin 2016b; Hennessy and Hamblin 2016; Naeser and Hamblin 2011; Naeser and Hamblin 2015). The almost complete lack of any adverse side-effects of PBM, coupled with growing disillusion with pharmaceutical drugs that affect brain function, have combined together to suggest an alternative physical therapy approach to improving brain function.
Traumatic brain injury (TBI) is caused by some type of trauma to the head, often resulting from road traffic accidents, assaults, falls, sports injuries, or blast injuries suffered in military conflict. TBI is classified as mild (loss of consciousness 0-30 minutes; altered mental state <24 hours; post-trauma amnesia <1 day); moderate (loss of consciousness 30 minutes to 24 hours; altered mental state >24 hours; post-trauma amnesia >1-7 days), or severe (loss of consciousness >24 hours; altered mental state >24 hours; post-trauma amnesia >7 days) (Blennow et al. 2016). There are three cases of TBI sustained each minute in the US (Faul et al. 2010). Repeated mild episodes of TBI (also known as concussions) even without loss of consciousness, may have devastating cumulative effects (Kamins and Giza 2016). Chronic traumatic encephalopathy is a recently recognized condition resulting from repeated head trauma, found in boxers, football players, and military personnel (McKee et al. 2016; Safinia et al. 2016). There is presently no accepted treatment for TBI, although some investigational approaches are being tested in both the acute (neuroprotection) and chronic (neurorehabilitation) settings (Loane and Faden 2010). One of these novel approaches is PBM or LLLT (Hamblin 2016a; Hamblin 2016b; Huang et al. 2012; Thunshelle and Hamblin 2016).
2. Mechanisms of action
Uncertainties about the mechanism of action of PBM at the molecular and cellular levels, have undoubtedly held back its acceptance in the wider biomedical community. However in recent years substantial progress has been made in this regard (de Freitas and Hamblin 2016). In the following section the state-of-the-art knowledge about the mechanisms of PBM is summarized. Figure 1 shows a graphical representation of the cellular and molecular mechanisms of PBM.
Molecular mechanisms of tPBM
Light passes through the scalp and skull, where depending on the wavelength it is absorbed by two different chromophores. Red and NIR (up to 940nm) is primarily absorbed by cytochrome c oxidase in the mitochondrial respiratory chain of the cortical neurons. Longer wavelength NIR light (980nm, 1064nm) is primarily absorbed by heat and light-sensitive transient receptor potential ion channels. In both cases cell signaling and messenger molecules are upregulated as a result of stimulated mitochondrial activity, including reactive oxygen species (ROS), and adenosine triphosphate (ATP). hv is light, TRPV is transient receptor potential vanilloid (ion channels).
2.1 Chromophores
The first law of photobiology states that a photon must be absorbed by some molecule within the tissue to have any biological effect. The identity of these chromophores has been the subject of much scientific investigation and speculation. Largely due to the efforts of Tiina Karu in Russia, the enzyme cytochrome c oxidase (CCO) has been identified as a major chromophore of red/NIR light (Karu 1999; Karu and Kolyakov 2005; Karu et al. 2004a; Karu et al. 2004b). CCO is unit IV in the mitochondrial respiratory chain and has absorption peaks reaching well into the NIR spectral region (up to 900 nm) as well as in the red and blue regions. The most discussed hypothesis to explain exactly how photon absorption can stimulate the activity of CCO involves the photodissociation of inhibitory nitric oxide (NO) that can bind to the copper and heme centers in the enzyme and prevent oxygen from gaining access to the active sites (Lane 2006). In experimental models (such as isolated mitochondria) oxygen consumption and ATP production are increased, and the mitochondrial membrane potential is raised (Passarella et al. 1984).
A less well-appreciated mechanism involves light and heat-gated ion channels. These cation ion channels are thought to be members of the transient receptor potential (TRP) superfamily consisting of over 28 distinct members organized into six subfamilies, based on their primary amino acid structures (Caterina and Pang 2016). TRPV (vanilloid sub-family) members including TRPV1 (capsaicin receptor) have been shown to be activated by various wavelengths of light including green, red and NIR.
2.2 Cellular mechanisms
After the primary photon absorption event occurs, whether that the photons are absorbed by CCO, or by TRP ion channels a series of secondary events occurs. One of these events is the generation of reactive oxygen species (ROS), which are thought to be produced inside the mitochondria due to an increase in electron transport, and a rise in the mitochondrial membrane potential above the baseline levels (Suski et al. 2012). It should be noted that mitochondrial ROS can be produced when MMP is raised above normal, and also when ROS is reduced below normal. It is thought that the ROS produced when MMP is lowered (mitochondrial dysfunction) are more damaging than ROS produced when MMP is raised (mitochondrial stimulation). Nitric oxide is produced after PBM (Hamblin 2008), possibly by photodissociation from CCO where it inhibits oxygen consumption and electron transport (Lane 2006). Cyclic adenosine monophosphate (cAMP) (Gao and Xing 2009) and intracellular calcium are increased (Alexandratou et al. 2002). Many of these secondary mediators in the signaling pathways triggered by PBM, can induce activation of transcription factors, that go on to upregulate or downregulate expression levels of a large number of genes. One of the best-known transcription factors is NF-kB that can regulate expression of over one hundred genes including proteins with antioxidant, anti-apoptotic, pro-proliferation, and pro-migration functions. PBM (810 nm 3J/cm2) was shown to activate NF-kB in mouse embryonic fibroblasts via ROS production (Chen et al. 2011a). Since NF-kB is known to be a pro-inflammatory transcription factor, it might be thought that PBM would be pro-inflammatory. However it was shown that NF-KB was decreased in already activated (treated with Toll-like receptor ligands) inflammatory dendritic cells by PBM (810 nm 3J/cm2) (Chen et al. 2011b).
2.3 Tissue mechanisms
The changes in expression levels of proteins involved in antioxidant and redox-regulation, anti-apoptotic and pro-survival, cellular proliferation, etc mean that distinct changes in tissue homeostasis, healing and regeneration can be expected after PBM. For instance, structural proteins such as collagen are newly synthesized in order to repair tissue damage (Tatmatsu-Rocha et al. 2016). Cells at risk of dying in tissue that has been subjected to ischemic or other insults are protected (Sussai et al. 2010). Stem cells are activated to leave their niche, proliferate and differentiate (Oron and Oron 2016; Zhang et al. 2016). Pain and inflammation are reduced (Chow et al. 2009). Blood flow is increased (Samoilova et al. 2008) (possibly as a result of the release of NO (Mitchell and Mack 2013)), which also stimulates lymphatic drainage thereby reducing edema (Dirican et al. 2011).
2.4 Brain specific mechanisms
In addition to the foregoing, there are some PBM tissue mechanisms that are specific to the brain. One of the most important is an increase in cerebral blood flow often reported after transcranial photobiomodualtion (tPBM) (Salgado et al. 2015), leading to increased tissue oxygenation, and more oxidized CCO as measured by NIR spectroscopy (Rojas and Gonzalez-Lima 2013). tPBM has been shown to reduce activated microglia in the brains of TBI mice as measured by IBA1 (ionized calcium-binding adapter molecule-1) expression thus demonstrating reduced neuroinflammation (Khuman et al. 2012). tPBM has been shown to increase neurogenesis (formation of new brain cells derived from neuroprogenitor cells) (Xuan et al. 2014), and synaptogenesis (formation of new connections between existing brain cells) (Xuan et al. 2015) both in TBI mice. Figure 2 shows a graphical representation of a variety of these brain-specific tissue mechanisms.
Brain-specific mechanisms of tPBM
The gene transcription process described in Figure 1 can lead to decreases in neuronal apoptosis and excitotoxicity and lessening of inflammation and reduction of edema due to increased lymphatic flow, which together with protective factors such as antioxidants, will all help to reduce progressive brain damage. Increases in angiogenesis, expression of neurotrophins leading to activation of neural progenitor cells and more cell migration, and increased synaptogenesis may all contribute to the brain repairing itself from damage sustained in the trauma. AUC is area under the curve.
3. Transcranial photobiomodulation
Transcranial PBM is a growing approach to many different brain disorders that may be classified as sudden onset (stroke, TBI, global ischemia), neurodegenerative (Alzheimer's, Parkinson's, dementia), or psychiatric (depression, anxiety, posttraumatic stress disorder)(Hamblin 2016b; Hennessy and Hamblin 2016; Thunshelle and Hamblin 2016). In the following section some issues concerning where the light should be delivered, and the effects of PBM on uninjured mice and humans are addressed.
3.1 Light penetration
Several laboratories working in the field of tissue optics, have investigated the penetration of light of different wavelengths though the scalp and the skull, and to what depths into the parenchyma of the brain this light can penetrate. Answering the question “can light shone on the head sufficiently penetrate to reach the brain?” is difficult. The main reason is that at present it is unclear exactly what threshold of power density is necessary (expressed in mW/cm2) at some depth inside the brain to have a biological effect. There clearly must be a minimum value below which, the light can be delivered for an infinite time without having any effect, but whether this threshold is in the region of μW/cm2 or mW/cm2 is unknown at present.
Haeussinger et al. estimated that the mean penetration depth (5% remaining intensity) of NIR light through the scalp and skull was 23.6 + 0:7 mm (Haeussinger et al. 2011). Other studies have found comparable results with some variations depending on the precise location on the head and the precise wavelength studied (Okada and Delpy 2003; Strangman et al. 2014).
Jagdeo et al. (Jagdeo et al. 2012) used human cadaver heads (skull with intact soft tissue) to measure penetration of 830 nm light, and found that penetration depended on the anatomical region of the skull (0.9% at the temporal region, 2.1% at the frontal region, and 11.7% at the occipital region). Tedord et al. (Tedford et al. 2015) also used human cadaver heads to compare penetration of 660 nm, 808 nm, and 940 nm light. They found that 808 nm light penetrated best, and could reach a depth in the brain of 40–50 mm. Lapchak et al. compared the transmission of 810 nm light through the skulls (no soft tissue) of four different species, and found the mouse skull transmitted 40%, while for rat it was 21%, for rabbit it was 11.3 and for the human skull it was only 4.2% (Lapchak et al. 2015). Pitzschke and colleagues compared penetration of 670 nm and 810 nm light into the brain when delivered by a transcranial or a transphenoidal approach, and found that the best combination was 810 nm delivered transphenoidally (Pitzschke et al. 2015). Yaroslavsky et al. examined light penetration of different wavelengths through different parts of the brain tissue (white brain matter, gray brain matter, cerebellum, and brainstem tissues, pons, thalamus). Best penetration was found with wavelengths between 1000 and 1100 nm (Yaroslavsky et al. 2002). Henderson and Morries found that between 0.45% and 2.90% of 810 nm or 980 nm light penetrated through 3 cm of scalp, skull and brain tissue in ex vivo lamb heads (Henderson and Morries 2015a).
3.2 Local vs systemic effects of light
It is possible that the beneficial effects of PBM on the brain cannot be entirely explained by penetration of light through the scalp and skull into the brain itself, at a sufficient intensity to have an effect on the brain cells. The surface power density that can be safely applied to the head, is limited by heating of the skin. Perceptible heating of the skin starts to be felt when the power density is over about 500 mW/cm2, and can become severe at 1 W/cm2.
There has been one study that explicitly addressed whether direct transcranial PBM or indirect PBM is best for the brain. In a study of PBM for Parkinson's disease in a mouse model, Mitrofanis and colleagues compared the direct delivery of light to the mouse head, and they also covered up the head with aluminum foil so that the light was delivered to the remainder of the mouse body. They found that there was a highly beneficial effect on brain histology with light delivered to the head, but nevertheless there was also a statistically significant although less pronounced benefit (referred to as an “abscopal effect”) when the head was shielded from light. Moreover Oron and co-workers (Farfara et al. 2015) have shown that delivering NIR light to the mouse tibia (using either surface illumination or a fiber optic) resulted in improvements in memory and spatial learning in a transgenic mouse model of Alzheimer's disease. They proposed the mechanism involved PBM stimulating c-kit-positive mesenchymal stem cells (MSCs) that were normally resident in autologous bone marrow. These MSCs were proposed to be able to infiltrate the brain, and clear β-amyloid plaques (Oron and Oron 2016). It should be noted in general that the calvarial bone marrow of the skull contains substantial numbers of stem cells (Iwashita et al. 2003).
3.3 PBM for brain in uninjured animals
Several laboratories have reported that shining light onto the head of uninjured healthy mice or rats can improve various cognitive and emotional parameters. The first study reported that exposure of the middle aged (12 months) CD1 female mice to 1072 nm LED arrays (Michalikova et al. 2008) produced improved performance in a 3D maze compared to sham treated age-matched controls. Gonzalez-Lima and coworkers (Gonzalez-Lima and Barrett 2014) showed that transcranial PBM (9 mW/cm2 with a 660 nm LED array) delivered to rats induced dose-dependent increases in oxygen consumption (5% after 1 J/cm2 and 16% after 5 J/cm2) [113]. They also found that tPBM reduced fear renewal and prevented the reemergence of the extinguished conditioned fear-responses (Rojas et al. 2012).
3.4 PBM for enhancement of brain function in uninjured human volunteers
Gonzalez-Lima et al delivered transcranial PBM (1064 nm laser, 60 J/cm2 at 250 mW/cm2) to the forehead in uninjured human volunteers in a placebo-controlled, randomized study. The goal was to improve performance of cognitive tasks related to the prefrontal cortex, including a psychomotor vigilance task (PVT), a delayed match-to-sample (DMS) memory task, and improved mood as measured by the positive and negative affect schedule (PANAS-X) (Barrett and Gonzalez-Lima 2013). Subsequent studies in uninjured humans showed that tPBM with 1064 nm laser could improve performance in the Wisconsin Card Sorting Task (considered the gold standard test for executive function) (Blanco et al. 2015). They also showed that tPBM to the right forehead (but not the left forehead) could improve attention bias modification (ABM) in humans with depression (Disner et al. 2016).
Salgado et al. applied transcranial LED to enhance cerebral blood flow in healthy elderly women, as measured by transcranial Doppler ultrasound (TCD) of the right and left middle cerebral artery and basilar artery. Twenty-five non-institutionalized elderly women (mean age 72 years), with cognitive status > 24, were assessed using TCD before and after transcranial LED therapy. tPBM (627 nm, 70 mW/cm2, 10 J/cm2) was performed at four points of the frontal and parietal region for 30 s each twice a week for 4 weeks. There was a significant increase in the systolic and diastolic velocity of the left middle cerebral artery (25 and 30%, respectively) and the basilar artery (up to 17 and 25%), as well as a decrease in the pulsatility index and resistance index values of the three cerebral arteries analyzed (Salgado et al. 2015).
3.5 PBM for acute stroke
Transcranial PBM delivered to the head, has been investigated as a possible treatment for acute stroke (Lapchak 2010). Animal models such as rats and rabbits, were first used as laboratory models, and these animals had experimental strokes induced by a variety of methods and were then treated with light (usually 810 nm laser) within 24 h of stroke onset (Lampl 2007). In these studies intervention by tLLLT within 24 h had meaningful beneficial effects.
Treatment of acute stroke in human patients was then addressed in a series of three clinical trials called “Neurothera Effectiveness and Safety Trials” (NEST-1 (Lampl et al. 2007), NEST-2 (Huisa et al. 2013), and NEST-3 (Zivin et al. 2014)). The protocol used an 810 nm laser applied to the shaved head (20 separate points in the 10/20 EEG system) within 24 h of patients suffering an ischemic stroke. The first study, NEST-1, enrolled 120 patients between the ages of 40 to 85 years of age and found a significantly improved outcome (p < 0.05 real vs sham, NIH Stroke Severity Scale) 5 days after a single laser treatment had been administered (Lampl et al. 2007). This significantly improved status was still present 90 days post-stroke in 70% of the PBM patients (but only 51% of the sham patients). The second clinical trial, NEST-2, enrolled 660 patients, aged 40 to 90, who were randomly assigned to one of two groups (331 to PBM, 327 to sham) (Zivin et al. 2009). Significant improvements (p < 0.04) were found in the moderate and moderate-severe (but not for the severe) stroke patients. The last clinical trial, NEST-3, was planned for 1000 patients enrolled, but the study was prematurely terminated by the DSMB for futility (an expected lack of statistical significance) (Lapchak and Boitano 2016). Many commentators have asked how tPBM could work so well in the first trial, yet fail in the third trial. Insufficient light penetration, too long an interval between stroke onset and PBM, inappropriate stroke severity measurement scale, use of only one single tPBM treatment, and failure to illuminate different specific areas of the brain for individual patients, have all been suggested as contributory reasons (Hamblin 2016b). It is undoubtedly the case that the failure of NEST-3 has cast a cloud over the whole application of PBMT for TBI as well as for stroke. Many commentators have asked “Why are you testing PBMT for TBI, if it has been shown not to work for stroke?” The failure of the investigators not to take into account the anatomical location of the stroke (and also whether it was deep or superficial) was also likely to have played a role in the failure of NEST-3. It is logical that light should be applied to the same side of the head where the lesion was located, not both sides of the head (Naeser et al. 2012). In my opinion the use of a single application of PBMT also bore some of the responsibility. Although a single application of PBM to the head works very well for experimental animals (mice, rats, rabbits) who have suffered a stroke or a TBI, the same may not apply to humans.
4. Animal studies of PBM in acute TBI models
4.1 Studies from the Oron laboratory
Oron's group was the first (Oron et al. 2007) to demonstrate that a single exposure of the head of a mouse a few hours after creation of a TBI lesion using a NIR laser (808 nm) could improve neurological performance and reduce the size of the brain lesion. A weight-drop device was used to induce a closed-head TBI in the mice. An 808 nm diode laser with two energy densities calculated at the surface of the brain (1.2-2.4 J/cm2 delivered by 2 minutes of irradiation with 200mW laser power to the scalp) was delivered to the head 4 hours after TBI was induced. Neurobehavioral function was assessed by the neurological severity score (NSS). There was no significant difference between the control and laser-treated group in NSS between the power densities (10 vs 20 mW/cm2), and no significant difference at early time points (24 and 48 hours) post TBI. However, there was a significant improvement (27% lower NSS score) in the PBM group at times between 5 days and 4 weeks. The laser treated group also showed a smaller loss of cortical tissue than the sham group (Oron et al.). In another study (Oron et al. 2012) they varied the pulse parameters (CW, 100Hz, or 600Hz) and tested whether the tPBM was equally effective when delivered at 4, 6, or 8 hours post-TBI. They first established that a calculated dose to the cortical surface of 1.2 J/cm2 of 808nm laser at 200mW applied to the head, was more effective when delivered at 6 hours post TBI than at 8 hours. They then selected an even shorter time post-TBI (4 hours) and compared CW with 100Hz and 600Hz. At 56 days, more mice in the 100Hz group (compared to the CW and 600 Hz groups) had fully recovered. The 600Hz group had lower NSS scores than the CW and 100Hz groups up to 20 days. Magnetic resonance imaging (MRI) analysis demonstrated significantly smaller lesion volumes in PBM-treated mice compared to controls.
4.2 Studies from the Hamblin laboratory
Wu et al. (Wu et al. 2012) first explored the effect of varying the laser wavelengths of PBM had on closed-head TBI in mice. Mice were randomly assigned to a PBM treatment group with a particular wavelength, or to a sham treatment group as a control. Closed-head injury (CHI) was induced via a weight- drop apparatus. To analyze the severity of the TBI, the neurological severity score (NSS) was measured and recorded. The injured mice were then treated with varying wavelengths of laser light (665, 730, 810 or 980 nm) at an energy density of 36 J/cm2 directed onto the scalp at 4 hours post-TBI. The 665 nm and 810 nm laser groups showed significant improvement in NSS when compared to the control group between days 5 to 28. By contrast, the 730 nm and 980 nm laser groups did not show any significant improvement in NSS (Wu et al. 2012) (Figure 3). The tissue chromophore cytochrome c oxidase (CCO) is proposed to be responsible for the underlying photon absorption process that underlies many PBM effects. CCO has absorption bands around 665 nm and 810 nm while it has a low absorption region at the wavelength of 730 nm (Karu et al.). It should be noted that this particular study (Wu et al. 2012) found that the 980 nm did not produce the same positive effects as the 665 nm and 810 nm wavelengths did; nevertheless previous studies did find that the 980 nm wavelength was an active one for PBM (Anders et al. 2014). Wu et al. suggested that these dissimilar results may be due to differences in the energy density, irradiance etc. between the other studies and the Wu study (Wu et al. 2012). In particular a much lower dose of 980 nm might have been effective had it been tested (Wang et al. 2016). Ando et al. (Ando et al. 2011) next used the 810 nm wavelength produced by a Ga-Al-As diode laser delivered at parameters used in the Wu study, and varied the pulse modes of the laser. These modes consisted of either pulsed wave at 10 Hz or at 100 Hz (50% duty cycle) or continuous wave laser. They used a different mouse model of TBI induced with a controlled cortical impact device directly inflicting a lesion on the cortex via an open craniotomy. A single treatment with a power density of 50 mW/m2 and an energy density of 36 J/cm2 (duration of 12 minutes) was given via tLLLT to the closed head in mice at 4 hours post CCI. At 48 hours to 28 days post TBI, all laser treated groups had significant decreases in the measured neurological severity score (NSS) when compared to the controls. Although all laser treated groups had similar NSS improvement rates up to day 7, the PW 10 Hz group began to show even greater improvement beyond this point as seen in Figure 4. At day 28, the forced swim test for depression and anxiety was used and showed a significant decrease in the immobility time for the PW 10 Hz group. In the tail suspension test, which measures depression and anxiety, there was also a significant decrease in the immobility time at day 28, and also at day 1, in the PW 10 Hz group.
Effect of different laser wavelengths of tPBM in closed-head TBI in mice
(A) Sham-treated control versus 665 nm laser. (B) Sham-treated control versus 730 nm laser. (C) Sham-treated control versus 810 nm laser. (D) Sham-treated control versus 980 nm laser. Points are means of 8–12 mice and bars are SD. *P < 0.05; **P < 0.01; ***P < 0.001 (one-way ANOVA). Reprinted with permission from (Wu et al. 2012)
Effects of pulsing in tPBM for CCI-TBI in mice
(A) Time course of neurological severity score (NSS) of mice with TBI receiving either control (no laser-treatment), or 810 nm laser (36 J/cm2 delivered at 50 mW/cm2 with a spot size of 0.78 cm2 in either CW, PW 10 Hz or PW 100 Hz modes. Results are expressed as mean +/- S.E.M ***P < 0.001 vs. the other conditions. (B) Mean areas under the NSS-time curves in the two-dimensional coordinate system over the 28-day study for the 4 groups of mice. Results are means +/- SD (n = 10). Reprinted from (Ando et al. 2011) (open access).
Studies using immunofluorescence staining of sections cut from mouse brains showed that tPBM increased neuroprogenitor cells (incorporating BrdU) in the dentate gyrus (DG) of the hippocampus and the subventricular zone (SVZ) at 7 days after the treatment (Xuan et al. 2014). The neurotrophin known as brain derived neurotrophic factor (BDNF) was also increased in the DG and SVZ at 7 days, while the protein marker (synapsin-1) for synaptogenesis and neuroplasticity was increased in the cortex at 28 days but not in the DG, SVZ or in any location at 7 days (Xuan et al. 2015). Learning and memory as measured by the Morris water maze was also improved by tPBM (Xuan et al. 2014).
4.3 Studies from the Wu laboratory
Zhang et al. (Zhang et al. 2014) first showed that secondary brain injury occurred to a worse degree in mice that had been genetically engineered to lack “Immediate Early Response” gene X-1 (IEX-1). When these mice were exposed to a gentle head impact (thought to closely resemble mild TBI in humans) they had a worse NSS than uninjured mice with the same TBI. Exposure of IEX-1 knockout mice to PBM (150 mW/cm2, 4 min, and 36 J/cm2) delivered at 4 hours post injury, restored the NSS to almost baseline levels, suppressed proinflammatory cytokine expression of interleukin (IL)-Iβ and IL-6, but upregulated TNF-α. The original lack of IEX-1 decreased ATP production, but exposing the injured brain to LLLT elevated ATP production back to near normal levels.
Dong et al. (Dong et al. 2015) asked whether the beneficial effects of PBM on TBI in mice could be enhanced by combining PBM with administration of metabolic substrates such as pyruvate and/or lactate. The goal was to even further improve mitochondrial function in the brain. This combinatorial treatment was able to reverse memory and learning deficits in TBI injured mice back to normal levels as well as leaving the hippocampal region completely protected from tissue loss; a stark contrast to control TBI mice that exhibited severe tissue loss from secondary brain injury.
4.4 Studies from the Whalen laboratory
Khuman et al (Khuman et al. 2012) delivered PBM (800nm) either directly to the injured brain tissue (through the craniotomy) or transcranially in mice beginning 60-80 min after CCI TBI. At a dose of 60J/cm2 (500mW/cm2) the mice showed increased performance in the Morris water maze (latency to the hidden platform, p<0.05, and probe trial, p<0.01) compared to non-treated controls. When PBM was delivered via open craniotomy there was reduced microgliosis at 48h (IbA-1+ cells, p<0.05). Little or no effect of tPBM on post-injury cognitive function was observed using lower or higher doses, a 4-h administration time point or 60J/cm2 at 7-days post-TBI.
4.5 Studies from the Whelan laboratory
Quirk et al (Quirk et al. 2012) studied Sprague-Dawley rats who had received a severe CCI TBI and were divided into three groups: real TBI, sham surgery, and anesthetization only. Each group received either real or sham PBM consisting of 670nm LED treatments of 15J/cm2, 50mW/cm2, 5min, given two times per day for 3 days (chemical analysis) or 10 days (behavioral analysis using a TruScan nose-poke device). Significant differences in task entries, repeat entries, and task errors were seen in the TBI rats treated with PBM vs untreated TBI mice, and in sham surgery mice treated with PBM vs untreated sham surgery mice. A statistically significant decrease was found in the pro-apoptotic marker Bax, and increases in the anti-apoptotic marker Bcl-2 and reduced glutathione (GSH) levels in tPBM TBI mice.
4.6 Studies from the Marques laboratory
Moreira et al used a different model of TBI (Moreira et al. 2009). Wistar rats received a craniotomy and a copper probe cooled in liquid nitrogen was applied to the surface of the brain to create a standardized cryogenic injury. They treated the rats with either a 780nm or 660nm laser at one of two different doses (3J/cm2 or 5J/cm2) twice (once immediately after the injury and again 3 hours later). Rats were sacrificed 6h and 24h after the injury. The 780nm laser was better at reducing levels of pro-inflammatory cytokines (TNFα, IL1β, IL6) particularly at early timepoints (Moreira et al. 2009). In a follow-up study using 3 J/cm2 (Moreira et al. 2011) these workers reported on the healing of the injuries in these rats at timepoints 6h, 1, 7 and 14 days after the last irradiation. Cryogenic injury created focal lesions in the cortex characterized by necrosis, edema, hemorrhage and inflammatory infiltrate. The most striking findings were: PBM-treated lesions showed less tissue loss than control lesions at 6h. During the first 24h the amount of viable neurons was significantly higher in the PBM groups. PBM reduced the amount of GFAP (glial fibrillary acidic protein, a marker of astrogliosis) and the numbers of leukocytes and lymphocytes, thus demonstrating its anti-inflammatory effect.
5. Patients with chronic TBI
The majority of studies of PBM for TBI in laboratory animals have been conducted in the acute setting, while the majority of human studies of PBM for TBI have been conducted in patients who have suffered head injuries at various times in the past (sometimes quite a long time ago).
5.1 Naeser case reports
In 2011 Naeser, Saltmarche et al., published the first report describing two chronic, TBI cases treated with tPBM (Naeser et al. 2011). A 500 mW CW LED source (mixture of 660 nm red and 870 nm NIR LEDs) with a power density of 22.2 mW/cm2 (area of 22.48 cm2), was applied all over the head, for 10 minutes at each placement location (13.3 J/cm2). In the first case study the patient reported that she could concentrate on tasks for a longer period of time (the time able to work at a computer increased from 20 minutes to 3 hours). She had a better ability to remember what she read, decreased sensitivity when receiving haircuts in the spots where PBM was applied, and improved mathematical skills after undergoing PBM. The second patient had statistically significant improvements compared to prior neuropsychological tests after 9 months of treatment. The patient had a 2 standard deviation (SD) increase on tests of inhibition and inhibition accuracy (9th percentile to 63rd percentile on the Stroop test for executive function and a 1 SD increase on the Wechsler Memory scale test for the logical memory test (83rd percentile to 99th percentile) (Naeser et al. 2011).
5.2 Naeser case series
Naeser et al then went on to report a case series containing a further eleven patients (Naeser et al. 2014). This was an open protocol study that examined whether scalp application of red and NIR LED could improve cognition in patients with chronic, mild TBI (mTBI). This study enrolled 11 participants ranging in age from 26 to 62 years (6 males, 5 females) who suffered from persistent cognitive dysfunction after mTBI. The injuries in the participants had been caused by motor vehicle accidents, sports related events and for one participant, an improvised explosive device (IED) blast. tPBM consisted of 18 sessions (Monday, Wednesday, and Friday for 6 weeks) and was started anywhere from 10 months to 8 years post-TBI. A total of 11 LED cluster heads (5.25 cm in diameter, 500 mW, 22.2 mW/cm2, 13 J/cm2) were applied for 10 minutes per set (5 or 6 LED placements per set, Set A and then Set B, in each session). Neuropsychological testing was performed pre-LED application and 1 week, 1 month and 2 months after the final treatment. They found that there was a significant positive linear trend for the Stroop Test for executive function, in trial 3 inhibition (p = 0.004); Stroop, trial 4 inhibition switching (p = 0.003); California Verbal Learning Test (CVLT)-II, total trials 1-5 (p = 0.003); CVLT-II, long delay free recall (p = 0.006). Improved sleep and fewer post-traumatic stress disorder (PTSD) symptoms, if present beforehand, were observed after treatment. Participants and family members also reported better social function and a better ability to perform interpersonal and occupational activities. Although these results were significant, the authors suggested that further placebo-controlled studies would be needed to ensure the reliability of this approach (Naeser et al. 2014).
Naeser has proposed (Naeser et al. 2016; Naeser et al. 2014) that specific scalp placements of the LED cluster heads may affect specific cortical nodes in the intrinsic networks of the brain, such as the default mode network (DMN), the salience network (SN), and the central executive network (CEN). These intrinsic networks are often dysregulated after TBI (Sharp et al. 2014). Naeser proposed that the specific areas of the head to receive light, to target cortical nodes in these networks were as follows:
-
For the DMN, placement of the LED cluster head on the midline of face, centered on the upper forehead and the front hairline, targeted the left and right mesial prefrontal cortex; and on a midline, scalp location half-way between the occipital protuberance and the vertex of the head, targeted the precuneus; and on left and right LED placements superior to the tip of each ear and posterior to each ear, targeted the inferior parietal cortex/angular gyrus areas.
-
For the SN, placement of LED cluster heads on the left and right temple areas, to target the anterior insula (but due to depth of insula, unknown if the photons reached the target); midline of the vertex of the head, to target the left and right presupplementary motor areas; and the LED cluster head placed on the midline of face, centered on the upper forehead and the front hairline, also targeted the left and right dorsal anterior cingulate cortex.
-
For the CEN, left and right scalp LED placements immediately posterior to the front hairline (on a line directly superior from the pupils of the eyes), targeted the dorso-lateral prefrontal cortex areas; and the left and right LED placements superior to the tip of each ear and posterior to each ear, also targeted the posterolateral inferior parietal cortex/angular gyrus areas (also treated as part of the DMN).
Further studies from Naeser and colleagues (Naeser et al. 2016) tested an intranasal LED (iLED) device. Two small iLEDs (one red and the other NIR) were clipped into each nostril and used at the same time for 25 min. The parameters were as follows: red, 633nm, 8mW CW, 1 cm2, energy density 12 J/cm2 (25 min); NIR 810nm, 14.2mW, pulsed 10Hz, 1cm2, 21.3J/cm2. The first mTBI participant (24-year old female) who had sustained four sports-related concussions (two during snowboarding and two during field hockey), received iLED PBM three times per week for 6 weeks. Significant improvements were observed in tasks measuring executive function and verbal memory as well as attention and verbal fluency. At 1 week after the 18th iLED treatment, the average total time asleep had increased by 61 min per night and her sleep efficiency (total sleep time divided by total time in bed) had increased by 11%. At 12 weeks after the last iLED treatment, she was able to discontinue all sleep medications that she had previously been using. The second, mTBI participant who received the intranasal only, LED treatment series is a 49 Yr. M (non-Veteran) who sustained mTBI in a MVA, 30 years prior to receiving the intranasal LED treatment series. He showed significant improvement on the Controlled Oral Word Association-FAS Test post- the iLED treatment series, improving by +1.3 SD and +1.5 SD at 1 and 2 months post- the 18th iLED treatment. His sleep data indicated he was already a good sleeper, at entry.
5.3 Bogdanova and Naeser studies
Bogdanova reported (Bogdanova et al. 2014) a case report of two patients (1 female) with moderate TBI (medical records and clinical evaluation) and persistent cognitive dysfunction (as measured by neuropsychological tests of executive function and memory). Patients received 18 sessions of transcranial LED therapy (3×/week for 6 weeks) using the mixed red/NIR cluster described above (Naeser et al. 2011).
Standardized neuropsychological tests for executive function, memory, depression, PTSD and sleep measures (PSQI, actigraphy) were administered to participants pre-(T1), mid-(T2), and one week (T3) post-PBM treatment. Both PBM treated cases (P1 and P2) showed marked improvement in sleep (actigraphy total sleep) 1 week post-LED treatment (T3), as compared to pre-treatment (T1). P1 also improved in executive function, verbal memory, and sleep efficiency; while P2 significantly improved on measures of PTSD (PCL-M) and depression. No adverse events were reported.
5.4 Studies from Henderson and Morries
Henderson and Morries (Henderson and Morries 2015b) used a high-power NIR laser (10-15 W at 810 and 980 nm) and applied it to the head to treat a patient with moderate TBI. The patient received 20 NIR applications over a 2-month period. They carried out anatomical magnetic resonance imaging (MRI) and perfusion single-photon emission computed tomography (SPECT). The patient showed decreased depression, anxiety, headache, and insomnia, whereas cognition and quality of life improved, accompanied by changes in the SPECT imaging.
They next reported (Morries et al. 2015) a series of ten patients with chronic TBI (average time since injury 9.3 years) where each patient received ten treatments over the course of 2 months using a high-power NIR laser (13.2 W/0.89 cm2 equivalent to 14.6 W/cm2 at 810nm; or 9 W/0.89 cm2 equivalent to 10.11 W/cm2 at 980nm). A continuous sweeping motion over the forehead was utilized to minimize skin heating and cover a larger area. Skin temperature increased no more than 3°C. Overall symptoms of headache, sleep disturbance, cognition, mood dysregulation, anxiety, and irritability improved. Symptoms were monitored by depression scales and a novel patient diary system specifically designed for this study. These authors have proposed that high power lasers are preferable for tPBM treatments because the photons can better reach the brain (Henderson and Morries 2015a).
5.5 Case study from Nawashiro
Nawashiro et al (Nawashiro et al. 2012) treated a single patient who had suffered a severe TBI. The patient survived but was left in a persistent vegetative state for 8 months after the accident. He showed no spontaneous movement of limbs and a CT scan of the head 8 months after the accident showed a focal low-density area in the right frontal lobe. The device had 23 individual 850nm LEDs (13mW each; total power 299mW, total area 57cm2). A treatment time of 30 min per session delivered 20.5 J/cm2 over the left and right forehead areas repeated twice daily (6h apart), for 73 days. Five days after beginning the PBM (after 10 treatments), the patient began to spontaneously move his left arm and hand, which had not occurred during the previous 8 months. Single-photon emission computed tomography with N-isopropyl-[123I]p-iodoamphetamine (IMP-SPECT) was performed twice. The IMP-SPECT scans showed a focal increase (20% higher) in cerebral blood flow in the uninjured left anterior frontal lobe 30 min after the last (146th) PBM treatment, compared to before PBM began.
6. Conclusion and future prospects
As was mentioned above, one of the most important questions to be answered when contemplating clinical treatment of TBI patients with tPBM, is what is the best time to administer the treatment? All the available reports of studies using PBM in laboratory animal models of TBI and stroke, and also in patients treated for stroke, have been in the acute phase where the overall goal of the intervention can be best described as neuroprotection. Not only that but there are several reports (Lapchak et al. 2007; Oron et al. 2012) that PBM for both TBI and stroke is most effective when it is delivered as soon as possible after the actual event (head impact or ischemic stroke). The protocols for the series of NEST clinical trials specified that patients should be treated with PBM within 24 hours of the stroke occurring. By contrast, all the clinical trials of PBM for patients with TBI, that have so far been carried out, have been with chronic TBI, after varying periods of time having elapsed after the original head injury, sometimes as long as 8 years. Although it would be generally supposed that tPBM would be effective when delivered to acute TBI patients, this has not yet been actually tested. If tPBM were to be used for acute TBI patients, then presumably the PBM should be delivered perhaps beginning at 4 to 6 hours post-TBI, for a limited number of times after the injury; perhaps once a day for 7 days?
The dosimetry and optimum delivery apparatus of tPBM is still uncertain. Although there is some consensus that wavelengths in the region of 800-900nm will penetrate the scalp and skull, other workers have used longer NIR wavelengths, 980nm, 1064nm, or 1072nm. Pulsing or CW is another unresolved question. The exact locations on the head that should receive the light are still unknown. Naeser has proposed (Naeser et al. 2016) some interesting considerations regarding the scalp placements of the tLEDS, and their effect on various intrinsic cortical networks of the brain. Targeted LED placements could promote better neuromodulation (activation/deactivation) in specific cortical nodes. It is possible that communication between nodes within one single network, and/or across networks could be improved. Moreover preliminary data indicate that intranasal, red plus near-infrared LEDs can also benefit TBI patients, although the degree to which light incident on the nasal mucosa, and possibly delivered transsphenoidally (Pitzschke et al. 2015) can penetrate directly into the brain, remains to be determined.
An advantage of intranasal and/or transcranial LED PBM therapy is that it can be performed in the home, for long-term use (Naeser et al. 2011). Also, 5 chronic, mild to moderately-severe dementia cases recently showed significant improvement on the Mini-Mental State Examination (p<0.003), and on the Alzheimer's Disease Assessment Scale-Cognitive subscale (p<0.023) after 12 weeks of daily, at-home, intranasal, near-infrared LED PBM treatments (810nm, pulsed at 10 Hz), and once-a-week in-office, tLED treatments applied to the cortical nodes of the Default Mode Network (Saltmarche et al. 2017). Anecdotally, there was also improved sleep, fewer angry outbursts, and less wandering. When all LED treatments were withdrawn after 12 weeks of active LED PBM treatment, there was precipitous decline in cognition and behavior. Thus, at-home, long-term use of iLED plus tLED PBM offers a potential therapy to mitigate the sequelae of Alzheimer's disease and possibly other neurodegenerative disorders, as well as TBI and stroke.
One highly distressing aspect of TBI symptomatology that has not so far been addressed by PBM, is that of post-traumatic epilepsy (PTE). TBI is the most significant cause of symptomatic epilepsy in people from 15 to 24 years of age. The frontal and temporal lobes are the most frequently affected regions, but imaging (MRI) often fails to show the precise cause. During PTE seizures there is an abnormal electrical discharge in the brain, with staring and unresponsiveness, stiffening or shaking of the body, legs, arms or head; strange sounds, tastes, visual images, feelings or smells; inability to speak or understand, etc (Cotter et al. 2017). Epilepsy has traditionally been considered to be a contra-indication for PBMT (Navratil and Kymplova 2002). However the knowledge that has recently been gained concerning the beneficial effects of PBMT on the damaged brain, suggests that this view may need to be critically revisited.
Moreover there is also potential of tPBM to treat a wide range of brain disorders only loosely associated with TBI, including Parkinson's disease (Purushothuman et al. 2013), depression, anxiety, post-traumatic stress disorder, autism spectrum disorder and so on (Hamblin 2016b).
The ongoing and accelerating clinical research efforts in testing PBM for TBI, are expected to lead to the answering of many of these questions in the coming years.
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5803455/
Shining light on the head: Photobiomodulation for brain disorders
Michael R. Hamblin - 10.1016/j.bbacli.2016.09.002 (Publication) 4466
This is 27 pages of independent analysis of how photobiomodulation effects the brain. Covers wavelengths, dosage, pulsing, depths and underlying reactions. Amazing.
View Resource
Photobiomodulation (PBM) describes the use of red or near-infrared light to stimulate, heal, regenerate, and protect tissue that has either been injured, is degenerating, or else is at risk of dying. One of the organ systems of the human body that is most necessary to life, and whose optimum functioning is most worried about by humankind in general, is the brain. The brain suffers from many different disorders that can be classified into three broad groupings: traumatic events (stroke, traumatic brain injury, and global ischemia), degenerative diseases (dementia, Alzheimer's and Parkinson's), and psychiatric disorders (depression, anxiety, post traumatic stress disorder). There is some evidence that all these seemingly diverse conditions can be beneficially affected by applying light to the head. There is even the possibility that PBM could be used for cognitive enhancement in normal healthy people. In this transcranial PBM (tPBM) application, near-infrared (NIR) light is often applied to the forehead because of the better penetration (no hair, longer wavelength). Some workers have used lasers, but recently the introduction of inexpensive light emitting diode (LED) arrays has allowed the development of light emitting helmets or “brain caps”. This review will cover the mechanisms of action of photobiomodulation to the brain, and summarize some of the key pre-clinical studies and clinical trials that have been undertaken for diverse brain disorders.
Keywords: Photobiomodulation, Low level laser (light) therapy, Ischemic stroke, Traumatic brain injury, Alzheimer's disease, Parkinson's disease, Major depression, Cognitive enhancement
Graphical abstract

1. Introduction
Photobiomodulation (PBM) as it is known today (the beneficial health benefits of light therapy had been known for some time before), was accidently discovered in 1967, when Endre Mester from Hungary attempted to repeat an experiment recently published by McGuff in Boston, USA [1]. McGuff had used a beam from the recently discovered ruby laser [2], to destroy a cancerous tumor that had been experimentally implanted into a laboratory rat. However (unbeknownst to Mester) the ruby laser that had been built for him, was only a tiny fraction of the power of the laser that had previously been used by McGuff. However, instead of curing the experimental tumors with his low-powered laser, Mester succeeded in stimulating hair regrowth and wound healing in the rats, in the sites where the tumors had been implanted [3], [4]. This discovery led to a series of papers describing what Mester called “laser biostimulation”, and soon became known as “low level laser therapy” (LLLT) [5], [6], [7].
LLLT was initially primarily studied for stimulation of wound healing, and reduction of pain and inflammation in various orthopedic conditions such as tendonitis, neck pain, and carpal tunnel syndrome [8]. The advent of light emitting diodes (LED) led to LLLT being renamed as “low level light therapy”, as it became more accepted that the use of coherent lasers was not absolutely necessary, and a second renaming occurred recently [9] when the term PBM was adopted due to uncertainties in the exact meaning of “low level”.
2. Mechanisms of action of photobiomodulation
2.1. Mitochondria and cytochrome c oxidase
The most well studied mechanism of action of PBM centers around cytochrome c oxidase (CCO), which is unit four of the mitochondrial respiratory chain, responsible for the final reduction of oxygen to water using the electrons generated from glucose metabolism [10]. The theory is that CCO enzyme activity may be inhibited by nitric oxide (NO) (especially in hypoxic or damaged cells). This inhibitory NO can be dissociated by photons of light that are absorbed by CCO (which contains two heme and two copper centers with different absorption spectra) [11]. These absorption peaks are mainly in the red (600–700 nm) and near-infrared (760–940 nm) spectral regions. When NO is dissociated, the mitochondrial membrane potential is increased, more oxygen is consumed, more glucose is metabolized and more ATP is produced by the mitochondria.
2.2. Reactive oxygen species, nitric oxide, blood flow
It has been shown that there is a brief increase in reactive oxygen species (ROS) produced in the mitochondria when they absorb the photons delivered during PBM. The idea is that this burst of ROS may trigger some mitochondrial signaling pathways leading to cytoprotective, anti-oxidant and anti-apoptotic effects in the cells [12]. The NO that is released by photodissociation acts as a vasodilator as well as a dilator of lymphatic flow. Moreover NO is also a potent signaling molecule and can activate a number of beneficial cellular pathways [13]. Fig. 2 illustrates these mechanisms.
Tissue specific processes that occur after PBM and benefit a range of brain disorders. BDNF, brain-derived neurotrophic factor; LLLT, low level light therapy; NGF, nerve growth factor; NT-3, neurotrophin 3; PBM, photobiomodulation; SOD, superoxide dismutase. ...
2.3. Light sensitive ion channels and calcium
It is quite clear that there must be some other type of photoacceptor, in addition to CCO, as is clearly demonstrated by the fact that wavelengths substantially longer than the red/NIR wavelengths discussed above, can also produce beneficial effects is some biological scenarios. Wavelengths such as 980 nm [14], [15], 1064 nm laser [16], and 1072 nm LED [17], and even broad band IR light [18] have all been reported to carry out PBM type effects. Although the photoacceptor for these wavelengths has by no means been conclusively identified, the leading hypothesis is that it is primarily water (perhaps nanostructured water) located in heat or light sensitive ion channels. Clear changes in intracellular calcium can be observed, that could be explained by light-mediated opening of calcium ion channels, such as members of the transient receptor potential (TRP) super-family [19]. TRP describes a large family of ion channels typified by TRPV1, recently identified as the biological receptor for capsaicin (the active ingredient in hot chili peppers) [20]. The biological roles of TRP channels are multifarious, but many TRP channels are involved in heat sensing and thermoregulation [21].
2.4. Signaling mediators and activation of transcription factors
Most authors suggest that the beneficial effects of tPBM on the brain can be explained by increases in cerebral blood flow, greater oxygen availability and oxygen consumption, improved ATP production and mitochondrial activity [22], [23], [24]. However there are many reports that a brief exposure to light (especially in the case of experimental animals that have suffered some kind of acute injury or traumatic insult) can have effects lasting days, weeks or even months [25]. This long-lasting effect of light can only be explained by activation of signaling pathways and transcription factors that cause changes in protein expression that last for some considerable time. The effects of PBM on stimulating mitochondrial activity and blood flow is of itself, unlikely to explain long-lasting effects. A recent review listed no less than fourteen different transcription factors and signaling mediators, that have been reported to be activated after light exposure [10].
Fig. 1 illustrates two of the most important molecular photoreceptors or chromophores (cytochrome c oxidase and heat-gated ion channels) inside neuronal cells that absorb photons that penetrate into the brain. The signaling pathways and activation of transcription factors lead to the eventual effects of PBM in the brain.
Molecular and intracellular mechanisms of transcranial low level laser (light) or photobiomodulation. AP1, activator protein 1; ATP, adenosine triphosphate; Ca2 +, calcium ions; cAMP, cyclic adenosine monophosphate; NF-kB, nuclear factor kappa ...
Fig. 2 illustrates some more tissue specific mechanisms that lead on from the initial photon absorption effects explained in Fig. 1. A wide variety of processes can occur that can benefit a correspondingly wide range of brain disorders. These processes can be divided into short-term stimulation (ATP, blood flow, lymphatic flow, cerebral oxygenation, less edema). Another group of processes center around neuroprotection (upregulation of anti-apoptotic proteins, less excitotoxity, more antioxidants, less inflammation). Finally a group of processes that can be grouped under “help the brain to repair itself” (neurotrophins, neurogenesis and synaptogenesis).
2.5. Biphasic dose response and effect of coherence
The biphasic dose response (otherwise known as hormesis, and reviewed extensively by Calabrese et al. [26]) is a fundamental biological law describing how different biological systems can be activated or stimulated by low doses of any physical insult or chemical substance, no matter how toxic or damaging this insult may be in large doses. The most well studied example of hormesis is that of ionizing radiation, where protective mechanisms are induced by very low exposures, that can not only protect against subsequent large doses of ionizing radiation, but can even have beneficial effects against diseases such as cancer using whole body irradiation [27].
There are many reports of PBM following a biphasic dose response (sometimes called obeying the Arndt-Schulz curve [28], [29]. A low dose of light is beneficial, but raising the dose produces progressively less benefit until eventually a damaging effect can be produced at very high light [30]. It is often said in this context that “more does not mean more”.
Another question that arises in the field of PBM is whether the coherent monochromatic lasers that were used in the original discovery of the effect, and whose use continued for many years, are superior to the rather recent introduction of LEDs, that are non-coherent and have a wider band-spread (generally 30 nm full-width half-maximum). Although there are one or two authors who continue to believe that coherent lasers are superior [31], most commentators feel that other parameters such as wavelength, power density, energy density and total energy are the most important determinants of efficacy [8].
3. Tissue optics, direct versus systemic effects, light sources
3.1. Light penetration into the brain
Due to the growing interest in PBM of the brain, several tissue optics laboratories have investigated the penetration of light of different wavelengths through the scalp and the skull, and to what depths into the brain this light can penetrate. This is an intriguing question to consider, because at present it is unclear exactly what threshold of power density in mW/cm2 is required in the b5rain to have a biological effect. There clearly must be a minimum value below which the light can be delivered for an infinite time without doing anything, but whether this is in the region of μW/cm2 or mW/cm2 is unknown at present.
Functional near-infrared spectroscopy (fNIRS) using 700–900 nm light has been established as a brain imaging technique that can be compared to functional magnetic resonance imaging (fMRI) [32]. Haeussinger et al. estimated that the mean penetration depth (5% remaining intensity) of NIR light through the scalp and skull was 23:6 + 0:7 mm [33]. Other studies have found comparable results with variations depending on the precise location on the head and wavelength [34], [35].
Jagdeo et al. [36] used human cadaver heads (skull with intact soft tissue) to measure penetration of 830 nm light, and found that penetration depended on the anatomical region of the skull (0.9% at the temporal region, 2.1% at the frontal region, and 11.7% at the occipital region). Red light (633 nm) hardly penetrated at all. Tedord et al. [37] also used human cadaver heads to compare penetration of 660 nm, 808 nm, and 940 nm light. They found that 808 nm light was best and could reach a depth in the brain of 40–50 mm. Lapchak et al. compared the transmission of 810 nm light through the skulls of four different species, and found mouse transmitted 40%, while for rat it was 21%, rabbit it was 11.3 and for human skulls it was only 4.2% [38]. Pitzschke and colleagues compared penetration of 670 nm and 810 nm light into the brain when delivered by a transcranial or a transphenoidal approach, and found that the best combination was 810 nm delivered transphenoidally [39]. In a subsequent study these authors compared the effects of storage and processing (frozen or formalin-fixed) on the tissue optical properties of rabbit heads [40]. Yaroslavsky et al. examined light penetration of different wavelengths through different parts of the brain tissue (white brain matter, gray brain matter, cerebellum, and brainstem tissues, pons, thalamus). Best penetration was found with wavelengths between 1000 and 1100 nm [41].
Henderson and Morries found that between 0.45% and 2.90% of 810 nm or 980 nm light penetrated through 3 cm of scalp, skull and brain tissue in ex vivo lamb heads [42].
3.2. Systemic effects
It is in fact very likely that the beneficial effects of PBM on the brain cannot be entirely explained by penetration of photons through the scalp and skull into the brain itself. There have been some studies that have explicitly addressed this exact issue. In a study of PBM for Parkinson's disease in a mouse model [43]. Mitrofanis and colleagues compared delivering light to the mouse head, and also covered up the head with aluminum foil so that they delivered light to the remainder of the mouse body. They found that there was a highly beneficial effect on neurocognitive behavior with irradiation to the head, but nevertheless there was also a statistically significant (although less pronounced benefit, referred to by these authors as an ‘abscopal effect”) when the head was shielded from light [44]. Moreover Oron and co-workers [45] have shown that delivering NIR light to the mouse tibia (using either surface illumination or a fiber optic) resulted in improvement in a transgenic mouse model of Alzheimer's disease (AD). Light was delivered weekly for 2 months, starting at 4 months of age (progressive stage of AD). They showed improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. They proposed that the mechanism of this effect was to stimulate c-kit-positive mesenchymal stem cells (MSCs) in autologous bone marrow (BM) to enhance the capacity of MSCs to infiltrate the brain, and clear β-amyloid plaques [46]. It should be noted that the calvarial bone marrow of the skull contains substantial numbers of stem cells [47].
3.3. Laser acupuncture
Laser acupuncture is often used as an alternative or as an addition to traditional Chinese acupuncture using needles [48]. Many of the applications of laser acupuncture have been for conditions that affect the brain [49] such as Alzheimer's disease [50] and autism [51] that have all been investigated in animal models. Moreover laser acupuncture has been tested clinically [52].
3.4. Light sources
A wide array of different light sources (lasers and LEDs) have been employed for tPBM. One of the most controversial questions which remains to be conclusively settled, is whether a coherent monochromatic laser is superior to non-coherent LEDs typically having a 30 nm band-pass (full width half maximum). Although wavelengths in the NIR region (800–1100 nm) have been the most often used, red wavelengths have sometimes been used either alone, or in combination with NIR. Power levels have also varied markedly from Class IV lasers with total power outputs in the region of 10 W [53], to lasers with more modest power levels (circa 1 W). LEDs can also have widely varying total power levels depending on the size of the array and the number and power of the individual diodes. Power densities can also vary quite substantially from the Photothera laser [54] and other class IV lasers , which required active cooling (~ 700 mW/cm2) to LEDs in the region of 10–30 mW/cm2.
3.5. Usefulness of animal models when testing tPBM for brain disorders
One question that is always asked in biomedical research, is how closely do the laboratory models of disease (which are usually mice or rats) mimic the human disease for which new treatments are being sought? This is no less critical a question when the areas being studied include brain disorders and neurology. There now exist a plethora of transgenic mouse models of neurological disease [55], [56]. However in the present case, where the proposed treatment is almost completely free of any safety concerns, or any reported adverse side effects, it can be validly questioned as to why the use of laboratory animal models should be encouraged. Animal models undoubtedly have disadvantages such as failure to replicate all the biological pathways found in human disease, difficulty in accurately measuring varied forms of cognitive performance, small size of mice and rats compared to humans, short lifespan affecting the development of age related diseases, and lack of lifestyle factors that adversely affect human diseases. Nevertheless, small animal models are less expensive, and require much less time and effort to obtain results than human clinical trials, so it is likely they will continue to be used to test tPBM for the foreseeable future.
4. PBM for stroke
4.1. Animal models
Perhaps the most well-investigated application of PBM to the brain, lies in its possible use as a treatment for acute stroke [57]. Animal models such as rats and rabbits, were first used as laboratory models, and these animals had experimental strokes induced by a variety of methods and were then treated with light (usually 810 nm laser) within 24 h of stroke onset [58]. In these studies intervention by tLLLT within 24 h had meaningful beneficial effects. For the rat models, stroke was induced by middle cerebral artery occlusion (MCAO) via an insertion of a filament into the carotid artery or via craniotomy [59], [60]. Stroke induction in the “rabbit small clot embolic model” (RSCEM) was by injection of a preparation of small blood clots (made from blood taken from a second donor rabbit) into a catheter placed in the right internal carotid artery [61]. These studies and the treatments and results are listed in Table 1.
Reports of transcranial LLLT used for stroke in animal models.
CW, continuous wave; LLLT, low level light therapy; MCAO, middle cerebral artery occlusion; NOS, nitric oxide synthase; RSCEM, rabbit small clot embolic model; TGFβ1, transforming growth factor β1.
4.2. Clinical trials for acute stroke
Treatment of acute stroke was addressed in a series of three clinical trials called “Neurothera Effectiveness and Safety Trials” (NEST-1 [65], NEST-2 [66], and NEST-3 [67]) using an 810 nm laser applied to the shaved head within 24 h of patients suffering an ischemic stroke. The first study, NEST-1, enrolled 120 patients between the ages of 40 to 85 years of age with a diagnosis of ischemic stroke involving a neurological deficit that could be measured. The purpose of this first clinical trial was to demonstrate the safety and effectiveness of laser therapy for stroke within 24 h [65]. tPBM significantly improved outcome in human stroke patients, when applied at ~ 18 h post-stroke, over the entire surface of the head (20 points in the 10/20 EEG system) regardless of stroke [65]. Only one laser treatment was administered, and 5 days later, there was significantly greater improvement in the Real- but not in the Sham-treated group (p < 0.05, NIH Stroke Severity Scale). This significantly greater improvement was still present at 90 days post-stroke, where 70% of the patients treated with Real-LLLT had a successful outcome, while only 51% of Sham-controls did. The second clinical trial, NEST-2, enrolled 660 patients, aged 40 to 90, who were randomly assigned to one of two groups (331 to LLLT, 327 to sham) [68]. Beneficial results (p < 0.04) were found for the moderate and moderate-severe (but not for the severe) stroke patients, who received the Real laser protocol [68]. These results suggested that the overall severity of the individual stroke should be taken into consideration in future studies, and very severe patients are unlikely to recover with any kind of treatment. The last clinical trial, NEST-3, was planned for 1000 patients enrolled. Patients in this study were not to receive tissue plasminogen activator, but the study was prematurely terminated by the DSMB for futility (an expected lack of statistical significance) [67]. NEST-1 was considered successful, even though as a phase 1 trial, it was not designed to show efficacy. NEST-2 was partially successful when the patients were stratified, to exclude very severe strokes or strokes deep within the brain [66]. There has been considerable discussion in the scientific literature on precisely why the NEST-3 trial failed [69]. Many commentators have wondered how could tPBM work so well in the first trial, in a sub-group in the second trial, and fail in the third trial. Lapchak's opinion is that the much thicker skull of humans compared to that of the other animals discussed above (mouse, rat and rabbit), meant that therapeutically effective amounts of light were unlikely to reach the brain [69]. Moreover the time between the occurrence of a stroke and initiation of the PBMT may be an important factor. There are reports in the literature that neuroprotection must be administered as soon as possible after a stroke [70], [71]. Furthermore, stroke trials in particular should adhere to the RIGOR (rigorous research) guidelines and STAIR (stroke therapy academic industry roundtable) criteria [72]. Other contributory causes to the failure of NEST-3 may have been included the decision to use only one single tPBM treatment, instead of a series of treatments. Moreover, the optimum brain areas to be treated in acute stroke remain to be determined. It is possible that certain areas of the brain that have sustained ischemic damage should be preferentially illuminated and not others.
4.3. Chronic stroke
Somewhat surprisingly, there have not as yet been many trials of PBM for rehabilitation of stroke patients with only the occasional report to date. Naeser reported in an abstract the use of tPBM to treat chronic aphasia in post-stroke patients [73]. Boonswang et al. [74] reported a single patient case in which PBM was used in conjunction with physical therapy to rehabilitate chronic stroke damage. However the findings that PBM can stimulate synaptogenesis in mice with TBI, does suggest that tPBM may have particular benefits in rehabilitation of stroke patients. Norman Doidge, in Toronto, Canada has described the use of PBM as a component of a neuroplasticity approach to rehabilitate chronic stroke patients [75].
5. PBM for traumatic brain injury (TBI)
5.1. Mouse and rat models
There have been a number of studies looking at the effects of PBM in animal models of TBI. Oron's group was the first [76] to demonstrate that a single exposure of the mouse head to a NIR laser (808 nm) a few hours after creation of a TBI lesion could improve neurological performance and reduce the size of the brain lesion. A weight-drop device was used to induce a closed-head injury in the mice. An 808 nm diode laser with two energy densities (1.2–2.4 J/cm2 over 2 min of irradiation with 10 and 20 mW/cm2) was delivered to the head 4 h after TBI was induced. Neurobehavioral function was assessed by the neurological severity score (NSS). There were no significant difference in NSS between the power densities (10 vs 20 mW/cm2) or significant differentiation between the control and laser treated group at early time points (24 and 48 h) post TBI. However, there was a significant improvement (27% lower NSS score) in the PBM group at times of 5 days to 4 weeks. The laser treated group also showed a smaller loss of cortical tissue than the sham group [76].
Hamblin's laboratory then went on (in a series of papers [76]) to show that 810 nm laser (and 660 nm laser) could benefit experimental TBI both in a closed head weight drop model [77], and also in controlled cortical impact model in mice [25]. Wu et al. [77] explored the effect that varying the laser wavelengths of LLLT had on closed-head TBI in mice. Mice were randomly assigned to LLLT treated group or to sham group as a control. Closed-head injury (CHI) was induced via a weight drop apparatus. To analyze the severity of the TBI, the neurological severity score (NSS) was measured and recorded. The injured mice were then treated with varying wavelengths of laser (665, 730, 810 or 980 nm) at an energy level of 36 J/cm2 at 4 h directed onto the scalp. The 665 nm and 810 nm groups showed significant improvement in NSS when compared to the control group at day 5 to day 28. Results are shown in Fig. 3. Conversely, the 730 and 980 nm groups did not show a significant improvement in NSS and these wavelengths did not produce similar beneficial effects as in the 665 nm and 810 nm LLLT groups [77]. The tissue chromophore cytochrome c oxidase (CCO) is proposed to be responsible for the underlying mechanism that produces the many PBM effects that are the byproduct of LLLT. COO has absorption bands around 665 nm and 810 nm while it has low absorption bands at the wavelength of 730 nm [78]. It should be noted that this particular study found that the 980 nm did not produce the same positive effects as the 665 nm and 810 nm wavelengths did; nevertheless previous studies did find that the 980 nm wavelength was an active one for LLLT. Wu et al. proposed that these dissimilar results may be due to the variance in the energy level, irradiance, etc. between the other studies and this particular study [77].
tPBM for TBI in a mouse model. Mice received a closed head injury and 4 hours later a single exposure of the head to one of four different lasers (36 J/cm2 delivered at 150 mW/cm2 over 4 min with spot size 1-cm diameter) ...
Ando et al. [25] used the 810 nm wavelength laser parameters from the previous study and varied the pulse modes of the laser in a mouse model of TBI. These modes consisted of either pulsed wave at 10 Hz or at 100 Hz (50% duty cycle) or continuous wave laser. For the mice, TBI was induced with a controlled cortical impact device via open craniotomy. A single treatment with an 810 nm Ga-Al-As diode laser with a power density of 50 mW/m2 and an energy density of 36 J/cm2 was given via tLLLT to the closed head in mice for a duration of 12 min at 4 h post CCI. At 48 h to 28 days post TBI, all laser treated groups had significant decreases in the measured neurological severity score (NSS) when compared to the control (Fig. 4A). Although all laser treated groups had similar NSS improvement rates up to day 7, the PW 10 Hz group began to show greater improvement beyond this point as seen in Fig. 4. At day 28, the forced swim test for depression and anxiety was used and showed a significant decrease in the immobility time for the PW 10 Hz group. In the tail suspension test which measures depression and anxiety, there was also a significant decrease in the immobility time at day 28, and this time also at day 1, in the PW 10 Hz group.
tPBM for controlled cortical impact TBI in a mouse model. (A) Mice received a single exposure (810 nm laser, 36 J/cm2 delivered at 50 mW/cm2 over 12 min) [121]. (B) Mice received 3 daily exposures starting 4 h post-TBI ...
Studies using immunofluorescence of mouse brains showed that tPBM increased neuroprogenitor cells in the dentate gyrus (DG) and subventricular zone at 7 days after the treatment [79]. The neurotrophin called brain derived neurotrophic factor (BDNF) was also increased in the DG and SVZ at 7 days , while the marker (synapsin-1) for synaptogenesis and neuroplasticity was increased in the cortex at 28 days but not in the DG, SVZ or at 7 days [80] (Fig. 4B). Learning and memory as measured by the Morris water maze was also improved by tPBM [81]. Whalen's laboratory [82] and Whelan's laboratory [83] also successfully demonstrated therapeutic benefits of tPBM for TBI in mice and rats respectively.
Zhang et al. [84] showed that secondary brain injury occurred to a worse degree in mice that had been genetically engineered to lack “Immediate Early Response” gene X-1 (IEX-1) when exposed to a gentle head impact (this injury is thought to closely resemble mild TBI in humans). Exposing IEX-1 knockout mice to LLLT 4 h post injury, suppressed proinflammatory cytokine expression of interleukin (IL)-Iβ and IL-6, but upregulated TNF-α. The lack of IEX-1 decreased ATP production, but exposing the injured brain to LLLT elevated ATP production back to near normal levels.
Dong et al. [85] even further improved the beneficial effects of PBM on TBI in mice, by combining the treatment with metabolic substrates such as pyruvate and/or lactate. The goal was to even further improve mitochondrial function. This combinatorial treatment was able to reverse memory and learning deficits in TBI mice back to normal levels, as well as leaving the hippocampal region completely protected from tissue loss; a stark contrast to that found in control TBI mice that exhibited severe tissue loss from secondary brain injury.
5.2. TBI in humans
Margaret Naeser and collaborators have tested PBM in human subjects who had suffered TBI in the past [86]. Many sufferers from severe or even moderate TBI, have very long lasting and even life-changing sequelae (headaches, cognitive impairment, and difficulty sleeping) that prevent them working or living any kind or normal life. These individuals may have been high achievers before the accident that caused damage to their brain [87]. Initially Naeser published a report [88] describing two cases she treated with PBM applied to the forehead twice a week. A 500 mW continuous wave LED source (mixture of 660 nm red and 830 nm NIR LEDs) with a power density of 22.2 mW/cm2 (area of 22.48 cm2), was applied to the forehead for a typical duration of 10 min (13.3 J/cm2). In the first case study the patient reported that she could concentrate on tasks for a longer period of time (the time able to work at a computer increased from 30 min to 3 h). She had a better ability to remember what she read, decreased sensitivity when receiving haircuts in the spots where LLLT was applied, and improved mathematical skills after undergoing LLLT. The second patient had statistically significant improvements compared to prior neuropsychological tests after 9 months of treatment. The patient had a 2 standard deviation (SD) increase on tests of inhibition and inhibition accuracy (9th percentile to 63rd percentile on the Stroop test for executive function and a 1 SD increase on the Wechsler Memory scale test for the logical memory test (83rd percentile to 99th percentile) [89].
Naeser et al. then went on to report a case series of a further eleven patients [90]. This was an open protocol study that examined whether scalp application of red and near infrared (NIR) light could improve cognition in patients with chronic, mild traumatic brain injury (mTBI). This study had 11 participants ranging in age from 26 to 62 (6 males, 5 females) who suffered from persistent cognitive dysfunction after mTBI. The participants' injuries were caused by motor vehicle accidents, sports related events and for one participant, an improvised explosive device (IED) blast. tLLLT consisted of 18 sessions (Monday, Wednesday, and Friday for 6 weeks) and commenced anywhere from 10 months to 8 years post-TBI. A total of 11 LED clusters (5.25 cm in diameter, 500 mW, 22.2 mW/cm2, 13 J/cm2) were applied for about 10 min per session (5 or 6 LED placements per set, Set A and then Set B, in each session). Neuropsychological testing was performed pre-LED application and 1 week, 1 month and 2 months after the final treatment. Naeser and colleagues found that there was a significant positive linear trend observed for the Stroop Test for executive function, in trial 2 inhibition (p = 0.004); Stroop, trial 4 inhibition switching (p = 0.003); California Verbal Learning Test (CVLT)-II, total trials 1–5 (p = 0.003); CVLT-II, long delay free recall (p = 0.006). Improved sleep and fewer post-traumatic stress disorder (PTSD) symptoms, if present beforehand, were observed after treatment. Participants and family members also reported better social function and a better ability to perform interpersonal and occupational activities. Although these results were significant, further placebo-controlled studies will be needed to ensure the reliability of this these data [90].
Henderson and Morries [91] used a high-power NIR laser (10–15 W at 810 and 980 nm) applied to the head to treat a patient with moderate TBI. The patient received 20 NIR applications over a 2-month period. They carried out anatomical magnetic resonance imaging (MRI) and perfusion single-photon emission computed tomography (SPECT). The patient showed decreased depression, anxiety, headache, and insomnia, whereas cognition and quality of life improved, accompanied by changes in the SPECT imaging.
6. PBM for Alzheimer's disease (AD)
6.1. Animal models
There was a convincing study [92] carried out in an AβPP transgenic mouse of AD. tPBM (810 nm laser) was administered at different doses 3 times/week for 6 months starting at 3 months of age. The numbers of Aβ plaques were significantly reduced in the brain with administration of tPBM in a dose-dependent fashion. tPBM mitigated the behavioral effects seen with advanced amyloid deposition and reduced the expression of inflammatory markers in the transgenic mice. In addition, TLT showed an increase in ATP levels, mitochondrial function, and c-fos expression suggesting that there was an overall improvement in neurological function.
6.2. Humans
There has been a group of investigators in Northern England who have used a helmet built with 1072 nm LEDs to treat AD, but somewhat surprisingly no peer-reviewed publications have described this approach [93]. However a small pilot study (19 patients) that took the form of a randomized placebo-controlled trial investigated the effect of the Vielight Neuro system (see Fig. 5A) (a combination of tPBM and intranasal PBM) on patients with dementia and mild cognitive impairment [94]. This was a controlled single blind pilot study in humans to investigate the effects of PBM on memory and cognition. 19 participants with impaired memory/cognition were randomized into active and sham treatments over 12 weeks with a 4-week no-treatment follow-up period. They were assessed with MMSE and ADAS-cog scales. The protocol involved in-clinic use of a combined transcranial-intranasal PBM device; and at-home use of an intranasal-only PBM device and participants/ caregivers noted daily experiences in a journal. Active participants with moderate to severe impairment (MMSE scores 5–24) showed significant improvements (5-points MMSE score) after 12 weeks. There was also a significant improvement in ADAS-cog scores (see Fig. 5B). They also reported better sleep, fewer angry outbursts and decreased anxiety and wandering. Declines were noted during the 4-week no-treatment follow-up period. Participants with mild impairment to normal (MMSE scores of 25 to 30) in both the active and sham sub-groups showed improvements. No related adverse events were reported.
tPBM for Alzheimer's disease. (A) Nineteen patients were randomized to receive real or sham tPBM (810 nm LED, 24.6 J/cm2 at 41 mW/cm2). (B) Significant decline in ADAS-cog (improved cognitive performance) in real but not sham (unpublished ...
An interesting paper from Russia [95] described the use of intravascular PBM to treat 89 patients with AD who received PBM (46 patients) or standard treatment with memantine and rivastigmine (43 patients). The PBM consisted of threading a fiber-optic through a cathéter in the fémoral artery and advancing it to the distal site of the anterior and middle cerebral arteries and delivering 20 mW of red laser for 20–40 min. The PBM group had improvement in cerebral microcirculation leading to permanent (from 1 to 7 years) reduction in dementia and cognitive recovery.
7. Parkinson's disease
The majority of studies on PBM for Parkinson's disease have been in animal models and have come from the laboratory of John Mitrofanis in Australia [96]. Two basic models of Parkinson's disease were used. The first employed administration of the small molecule (MPTP or 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) to mice [97]. MPTP was discovered as an impurity in an illegal recreational drug to cause Parkinson's like symptoms (loss of substantia nigra cells) in young people who had taken this drug [98]. Mice were treated with tPBM (670-nm LED, 40 mW/cm2, 3.6 J/cm2) 15 min after each MPTP injection repeated 4 times over 30 h. There were significantly more (35%–45%) dopaminergic cells in the brains of the tPBM treated mice [97]. A subsequent study showed similar results in a chronic mouse model of MPTP-induced Parkinson's disease [99]. They repeated their studies in another mouse model of Parkinson's disease, the tau transgenic mouse strain (K3) that has a progressive degeneration of dopaminergic cells in the substantia nigra pars compacta (SNc) [100]. They went on to test a surgically implanted intracranial fiber designed to deliver either 670 nm LED (0.16 mW) or 670 nm laser (67 mW) into the lateral ventricle of the brain in MPTP-treated mice [101]. Both low power LED and high power laser were effective in preserving SNc cells, but the laser was considered to be unsuitable for long-term use (6 days) due to excessive heat production. As mentioned above, these authors also reported a protective effect of abscopal light exposure (head shielded) in this mouse model [43]. Recently this group has tested their implanted fiber approach in a model of Parkinson's disease in adult Macaque monkeys treated with MPTP [102]. Clinical evaluation of Parkinson's symptoms (posture, general activity, bradykinesia, and facial expression) in the monkeys were improved at low doses of light (24 J or 35 J) compared to high doses (125 J) [103].
The only clinical report of PBM for Parkinson's disease in humans was an abstract presented in 2010 [104]
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5066074/
Photobiomodulation of the Brain
Michael R. Hamblin and Yng-Ying Huang - 2019 (Publication) 4512
This is Hamblin and Huang's best summary of PBM for treating the brain.
View Resource
Photobiomodulation (PBM) also known as low-level laser (or light) therapy has been known for over 50 years (since
1967), but it is only relatively recently that it has begun to make the transition into the mainstream. PBM describes the
use of red or near-infrared light at levels that do not produce undue heating of the tissue to produce beneficial effects
on the human body. The introduction of light-emitting diodes (LEDs) has made this approach more accessible than the
previously used laser sources, as LEDs are safer, cheaper, and can easily be used at home. Another factor that has led
to PBM becoming more widely accepted is the growing understanding of the mechanisms of action at a molecular and
cellular level. The lack of a clear mechanism of action was a deterrent to many biomedical scientists who maintained a
healthy level of skepticism.
Among the wide range of tissues, organs, diseases, and conditions that can be beneficially affected by PBM, the subject
of this book is the brain. The brain is probably the single human organ that engenders the most concern, interest,
and expenditure in the 21st century. Brain disorders that cause widespread morbidity, mortality, and loss of quality of
life can be divided into four broad categories. Traumatic brain disorders include stroke, traumatic brain injury (TBI),
global ischemia, and perinatal difficulties. Neurodegenerative diseases include Alzheimer’s disease, Parkinson’s disease,
and a range of dementias. Psychiatric disorders include major depression, anxiety, addiction, and insomnia, among
many others. Finally there are neurodevelopmental disorders (autism and ADHD) and the possibility of cognitive
enhancement in healthy individuals. Many of these brain disorders are specifically addressed in the present volume.
The book is divided into three parts. The first part covers some basic considerations, dosimetry, and devices, and
discusses the mechanisms of action at a cellular level and on the brain as a whole organ. The second part includes contributions
from researchers who have carried out studies on a variety of animal models in their investigations of brain
disorders, stroke, TBI, and Alzheimer’s and Parkinson’s diseases, to name a few. The third part concentrates on human
studies, including controlled clinical trials, pilot trials, case series, and clinical experience. Disorders treated include
TBI, stroke, Alzheimer’s and Parkinson’s diseases, depression, and others.
The book is expected to play a role in stimulating the further increase and acceptance of PBM for brain disorders,
which has really started to take off in recent years. It will also act as a resource for researchers and physicians wishing
to get a broad overview of the field and who are contemplating entering it themselves. The number of individuals considering
obtaining a home-use PBM device is also steadily increasing and this book will act as
Original Source: https://books.google.com/books/about/Photobiomodulation_in_the_Brain.html?id=P0qiDwAAQBAJ&source=kp_book_description
“Quantum Leap” in Photobiomodulation Therapy Ushers in a New Generation of Light-Based Treatments for Cancer and Other Complex Diseases: Perspective and Mini-Review
Luis Santana-Blank, MD, Elizabeth Rodríguez-Santana, MD, Karin E. Santana-Rodríguez, BS, and Heberto Reyes, MD - Photomedicine and Laser Surgery (Publication) 4490
A broad article, but follow the references located at the original document to learn more about more applicable topics.
View Resource
Abstract
Objective: Set within the context of the 2015 International Year of Light and Light-Based Technologies,and of a growing and aging world population with ever-rising healthcare needs, this perspective and mini-review focuses on photobiomodulation (PBM) therapy as an emerging, cost-effective, treatment option for cancer (i.e., solid tumors) and other complex diseases, particularly, of the eye (e.g., age-related macular degeneration, diabetic retinopathy, glaucoma, retinitis pigmentosa) and the central nervous system (e.g., Alzheimer's and Parkinson's disease). Background data: Over the last decades, primary and secondary mechanisms of PBM have been revealed. These include oxygen-dependent and oxygen-independent structural and functional action pathways. Signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a given set of parameters. Methods: This study was a perspective and nonsystematic literature mini-review. Results: Studies support what we describe as a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photo-cceptors to restore physiologic function. Conclusions: Based on existing evidence, it is argued that PBM therapy can raise the standard of care and improve the quality of life of patients for a fraction of the cost of many current approaches. PBM therapy can, therefore,benefit large, vulnerable population groups, including the elderly and the poor, whilehaving a major impact on medical practice and public finances.
Introduction
The United Nations declared 2015 to be the International Year of Light and Light-Based Technologies (IYL 2015) in recognition of the vital role of light-based systems in our daily lives, and their growing importance to meeting the world's challenges in areas as diverse as energy, education, telecommunication, agriculture, and health.1 Although our perception of light is often limited to the visible band of the electromagnetic (EM) spectrum,2 both lower and shorter wavelengths are increasingly used in new medical technologies3 including soft, injectable, and bioresorbable electronics.4 Described as an imperative cross-cutting discipline of in the twenty-first century, light science has already revolutionized the physical sciences and industry. The control of light at the nanoscale has unveiled a plethora of phenomena, leading to powerful new applications and setting high expectations for years to come.5 In particular, light's ability to control materials and transport coded signals forms the bases for many new photonic devices and systems, wherein photons act as tailor-made EM energy packets that can perform various functions.
Here, we describe a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photoacceptors to control biologic function in medicine through photobiomodulation (PBM) therapy. We propose that progress will lead to the imminent inception of PBM therapy as a mainstream treatment for multiple complex diseases, including solid tumors, as well as neurodegenerative diseases (NDs) of the eye and central nervous system (CNS)6–10 (Fig. 1). PBM therapy can raise the standard of care and improve the quality of life of patients at a fraction of the cost of many current approaches. Thus, a “quantum leap” in PBM therapy will benefit large and vulnerable population groups, including the elderly and the poor, while having a major impact on medical practice and public finances.11 This is particularly important because the high price of drug therapies, which can reach hundreds of thousands of dollars per year,12 as well as a growing and aging world population, are putting a severe strain on family and public finances around the world.13
Flow chart illustrating fields of light-based technologies, highlighting photobiomodulation (PBM) therapy applied to complex diseases as a quantum leap in medical therapeutics.
Origin, Trajectory and Myriad Relationships in PBM's “Quantum Leap” in Medicine
Concurrent with progress in PBM therapy, a long history of discoveries has put medicine at the brink of a revolution in the use of light–water interactions for the treatment of complex diseases.7,8,10,14 Long ago, Albert Szent-Gyorgyi postulated that water was at the core of energy transfer in biological systems (i.e., quantum biology), and that that explained how energy from biomolecules could be translated into free energy for cells.15–17 Ling further elaborated on the physical state of water in living cells,18 and proposed on theoretical grounds that ordered layers of water could extend infinitely under ideal conditions.19,20Later, Huber proposed a structural basis of light energy and electron transfer in biology.21 More recently, Zewail and others showed that, with rapid laser techniques, it is possible to “see” how atoms in a molecule move during a chemical reaction.22 Light science has now reached microscales at the limit of recordable physical observation (e.g., resonant intermolecular transfer of vibrational energy in water at −100 fs)23,24showing, for example, the memory of persistent correlations in water structures within 50 fs, which is important in stabilizing biological systems.25 These and other tremendous achievements have changed our view of water, from a merely passive medium to an integral active player in the physiology of life, and have opened the gates to both direct measurement and control of physiological processes via light–water interaction.
State of the Art in PBM
In 2016, PBM therapy will be added to the MeSH database as an entry term for records spanning five decades of research.26 As argued by Anders et al., this is a key step, as it distinguishes PBM therapy from light-based devices used for heating of tissues, such as near infrared (NIR) lamps or other applications that rely on thermal effects for all or part of their mechanisms of action.26 In contrast, PBM therapy employs low-level monochromatic or quasimonochromatic light, currently from visible blue (∼400 nm) to far-infrared (FIR ∼3200 nm), to induce nonthermal (≤0.01°C) photochemical and photophysical effects. Nonlinear processes through which PBM therapy can stimulate or inhibit; that is, modulate, physiological activity depend upon signal-to-noise rate and target cell/tissue parameters.27–29 Thus, signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a narrow set of parameters.13
Over the last decades, primary and secondary mechanisms of PBM at the tissue, cellular, and molecular levels have been revealed. These include two major structural and functional action pathways. The first, or classic, action pathway relates to oxygen-dependent mechanisms operated by oxidation-reduction enzymes of the respiratory chain, particularly cytochrome c oxidase (CcO), which is partly responsible for light energy absorption and transfer to cells and tissues.30 This pathway is associated to cofactors, pigments, metals, and proteins that act as key redox centers within the body's bioenergetic rack mechanism described by Huber.21 Nitric oxide (NO), as a first-level player, also has an activation and modulation role in the oxygen-dependent pathway.31–33
The second, or oxygen-independent, action pathway centers on the vital role of water not only as the prevalent medium of life but as an active molecule, capable of absorbing radiant energy (e.g., IR light) and transporting/transducing it along extended biological surfaces, from bulk water to confined water in nanoscopic tissue and cell spaces. Light–water dynamics precede/coexist with the classic oxygen-dependent action pathway and complement and facilitate energy transfer for increased adenosine triphosphate (ATP) production.29,34,35 As a point of comparison, correlated internal electron- and proton-transfer reactions have been tracked in real time into the oxidized enzyme (CcO), revealing an overall real time of 3.46 ms.36 This relay is slower by several orders of magnitude than total energy transport through water dynamics from bulk liquid water to confined spaces.34
Oxygen-independent light–water interactions may further power and modulate molecular signaling pathways and gene transcription factors via multiple nonmetabolic pathways.10,35 For examle, the energy of the drive force wave of an infrared pulsed laser device (IPLD) used in our group's previous studies (NIR 0.27 eV) is within the range of the strength of hydrogen bonds,29,37 and the IPLD carrier wave oscillates at a frequency (3x 10e6 Hz) that enters in vibrational resonance with the rate of electron transfer through the DNA double helix.29,37 Theoretical evidence suggests that these wave properties promote the activation of open state dynamics,38,39 allowing the activation of complex chaotic dynamics as well as the regulation of DNA replication and transcription, because the existence of open states in one place of the chain can influence the dynamics of other distant open states.29,34,35 Resulting effects match reported reductions in the frequency of chromosome aberrations induced by that low-energy laser irradiation,40 as well as theoretical,38,39 experimental,27,28 and clinical studies.41–48 These and other oxygen-independent PBM effects are channeled through metabolic control levels to regulate the energy-dependent path from the genotype to the phenotype.49,50
Light–Water Interactions and the Quantum Leap in PBM
We propose that the key to understanding and controlling the biophysics and biochemistry of higher-order organisms stems from their dual aqueous and energy-dependent nature. Water represents ∼70% by mass of an adult human body, or nearly 99% of total molecules by number, given water's low molecular weight. In addition, high-order organisms, including humans, can be represented as complex electrochemical (semiconducting) systems that comprise a vast array of energy-sensitive materials and machinery, such as ion pumps (e.g., chemically driven electron pumping through molecular wires, such as the D pathway in CcO),34 molecular motors (e.g., ATP synthase and Brownian biomotors), transistors-capacitors (e.g., cell membrane), liquid crystals (e.g., membrane structure), and rechargeable electrolytic biological batteries (e.g., hydrophilic interface in cells/tissues). Life system's double nature, whose two main structural and functional pillars are energy and water joined to biomolecules, has, therefore, tremendous consequences for life and health.
Water's permittivity, calculated considering the system as a plane capacitor, is generally high. Therefore, radiant energy can penetrate and be absorbed by tissues to provide powerful tools in medicine.51 One example is the exclusion zone (EZ) described by Pollack.52 High-energy EZ water forms along hydrophilic surfaces (e.g., tissue interfaces) in response to radiant energy.53 Remarkably, EZ water can separate and store electrical charges, and can release up to 70% of such charges when it is perturbed, such as by injury-induced redox potentials.54 We have argued that supplied energy can power and modulate cellular work and signaling pathways, even when the metabolic energy pathway has been compromised, steering cells toward or away from programmed cell death.34 EZ water may, thus, act as an electrolytic bio-battery,35 which can efficiently and selectively transfer energy to sites expressing redox injury potentials, as found in cancer and other complex diseases, triggering reparative and regenerative mechanisms that can lead to restoring homeostasis/homeokinesis and, ultimately, health.29,34,35
Experimentally, IR energy absorption by water has been recently modeled in a porcine model, confirming that absorption depends upon fluence and wavelength. Further, the higher the concentration of water in tissues, the higher IR energy absorption will be.55 This is consistent with controlled clinical studies in solid tumors and complex ophthalmic and neurologic diseases,9,46,56 as well as molecular, biochemical, biophysical, and metabolic mechanistic support for a quantum leap in medical therapeutics based on the simple, but powerful, idea that properly tailored light can power and modulate physiologically reparative mechanisms.30,57–62
Cancer and Tumor Microenvironments
The bases of our understanding of cancer are constantly being questioned and revised, leading to new treatment goals. In a paradigm-changing editorial, Prendergast recently argued that “disorders in microenvironment and peripheral systems that control cancer might increasingly be viewed as primary rather than secondary factors in the root nature of cancer as a clinical disease.” This constitutes “a crucial and radical distinction from prevailing thought, since it implies that cancer may be a symptom of an underlying clinical disorder, rather than the root problem itself that needs to be addressed.” 6,63
Prendergast further suggested that “effective treatment of cancer may not necessarily entail understanding or addressing this complexity, but mastering the use of tissue or systemic systems that have the inherent ability to do so.” Hence, a common thread linking emerging perspectives in oncology and PBM therapy may well be the restitution of tissue homeostasis-homeokinesis via light-energy supplementation, a microenvironment effect that comprises and extends the Warburg effect previously discussed by our group.57,64–67
Photobiomodulation and Cancer
As far back as 1964–1966, McGuff et al. showed 64,65 that “laser energy has a selective effect on certain malignant tumors, resulting in their progressive regression and ultimate dissolution.” Following years of controversy,66,67 editorials by Karu68 and Lanzafame11,69 now stress evidence supporting the potential anticancer effects of PBM.11,68,69 New data confirm that PBM under certain parameters is safe for use in cancer patients.60 This is in accord with clinical results from our group using the abovementioned proof of concept IPLD.4,44
A phase I trial in patients with advanced neoplasias demonstrated that the IPLD studied was safe for clinical use and improved performance status and quality of life.41 Antitumor activity was observed in 88.23% of patients with 10 years of follow-up.41
In that series, T2-weighted MRI data showed increased water content of tumor heterogeneities42,44 preceding tumor-volume reduction and a therapeutic anticancer effect.42,44 Structural, kinetic, and thermodynamic implications of these changes in water dynamics have been analyzed at the tissue, cell, and interstitial levels.27 In conjunction, selective activation of programmed cellular death [i.e., apoptosis, necrosis, and anoikis (cell death by loss of cell adhesion)] and cytomorphologic modification (e.g., reduced size, increased roundness, increased vacuoles) were documented in neoplastic cells, but not in peripheral tissues.8,42 Modulation of cluster of differentiation (CD)4 CD45RA+, CD25 activated, tumor necrosis factor alpha (TNF-α), and soluble interleukin (IL)-2 receptor (sIL-2R) was further documented.43These hallmark results, supported by independent data,70–72 demonstrate that PBM therapy can modulate antitumor effects,6,8 in sharp contrast with long-held views.45,73,74 This evidence is also consistent with growing experimental and clinical reports from multiple other authors.60,75–82
PBM and Ophthalmic and Neurodegenerative Disorders
Recent evidence underscores common mechanisms between cancer and NDs of the eye and CNS. Research suggests that oxidative proteome damage may be the most likely cause of aging and age-related maladies such as cancer and other complex diseases, including NDs.83 Findings also show “common mechanisms of onset,” with a focus on genes such as DJ-1 and Myc-Modulator 1 (MM-1) and signaling pathways that contribute to the onset and pathogenesis of cancer and NDs such as retinitis pigmentosa (RP), Parkinson disease (PD), and cerebellar atrophy.”84 Finally, both disease groups are profoundly energetic in nature, featuring prominent deterioration of metabolic energy pathways.10
External light energy supplementation has been shown to generate neuroprotective, vasoprotective, baroprotective, immunomodulatory, and regenerative effects (Fig. 2). 47 We have documented that such effects may be activated and modulated locally and/or remotely via oxygen-dependent and oxygen-independent pathways that can encompass extended biologic surfaces and may even reach avascular eye tissues (i.e., cornea, lens, aqueous humor, and vitreous) noninvasively. Although a full elucidation of involved mechanisms escapes the scope of this perspective and mini-review, a very brief discussion of results from multiple authors is given subsequently.
Electromagnetic (light) energy supplementation based on water–light interactions. Upper left side shows classic oxygen (O2) dependent pathways by which light energy generates adenosine triphosphate (ATP)/ guanosine-5′-triphosphate (GTP) and other high-energy molecules. Upper right side shows O2 independent pathways by which photoinduced, nonlinear, oscillations in water provide energy for cellular work, signaling, and gene transcription. Top center shows interfacial exclusion zone (EZ) water, which acts as a selective rechargeable electrolytic bio-battery. Together, these pathways activate and modulate physiologically reparative mechanisms which, at appropriate irradiation parameters, can generate neuroprotective, vasoprotective, baroprotective, immunomodulator, and regenerative effects locally and remotely, promoting homeostasis/homeokinesis through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by the second law of thermodynamics. Arrows point to the sequence and direction of events. (Updated from reference 47. Authors retained copyright.)
PBM has shown promise in the treatment of diabetic retinopathy (DR),85,86 age-related macular degeneration (AMD),46 glaucoma,47 RP,87 Stargardt disease,88 Leber's hereditary optic neuropathy,89 Alzheimer's disease (AD), and PD, 90,91 among other conditions.89 Strikingly, although each of these NDs has different etiologies and pathogeneses, “they frequently induce a set of cell signals that lead to well-established and similar morphological and functional changes, including programmed cell death. Furthermore, oxidative stress, activation of apoptotic pathways and inflammatory response, are common features in all these diseases.”92
Remarkably, PBM can modulate apoptosis as well as necrosis.42,45,47 PBM can also be both pro-oxidant in the short term, but antioxidant in the long term,93 thus modulating reactive oxygen species (ROS) generation. We also found clinical evidence of immune regulatory effects over inflammation during treatment of solid tumors with the IPLD, a NIR diode laser pulsed at a frequency of 3 MHz.43 These results are in agreement with the regulating role of the vagal reflex on the inflammatory reflex reported by Tracey, using an electronic device that stimulated nerves to treat inflammation.71,72
In addition, PBM has been shown to protect against retinal dysfunction and photoreceptor cell death in rodent models of retinal injury and retinal degeneration.94 PBM has been further reported to attenuate oxidative stress and inflammation in primary astrocytes induced by amyloid β peptide (Aβ),95 and to reduce Aβ-induced apoptosis,96 which is thought to play a major role in AD. Nevertheless, it has been argued that red to NIR light cannot be transmitted through the scalp to the brain more than a few centimeters,97 which makes it nearly impossible to noninvasively treat AD with PBM 98 using conventional (direct) delivery systems/methods. Similarly, although an absence of adverse effects from 670 and 830 nm PBM applied to the retina in Sprague Dawley albino rats has been reported,94 extreme care must be taken to avoid photodamage of the eye99 from direct PBM procedures.
Conversely, we published an interventional case report of a patient with bilateral geographic atrophic AMD (gaAMD) and associated neurologic disease treated noninvasively, indirectly, and at a distance (i.e., remotely) from ocular structures and the CNS with the above-referenced IPLD/photo-infrared pulsed bio-modulation (PIPBM).46 Results showed neurologic improvement, transitory color vision, enhanced visual acuity, full-field electroretinogram (ERG) modifications toward a normal rhythm, drusen mobilization, decreased lens opacity, and lower intraocular pressure (IOP), in accord with a retrospective noncomparative data analysis from the phase I trial of patients with advanced cancer treated with the IPLD,41 which showed statistically significant evidence of a therapeutic hypotensor effect over IOP,47 and they are consistent with the positive neurological evolution of two trial patients.
Moreover, although trial participants did not develop media opacity, one pre-existing incipient cataract in the right eye of a patient (transitional meningioma) became denser and slightly smaller 3 months post-treatment, and remained unchanged 1 year post-treatment. The left eye lens of the same patient was unaffected. Although the finding could be part of the natural history of the cataract, we stressed that possible deterministic effects related to the initial metabolic or biochemical state of lens opacities should be studied.41
In accordance with the what was described, a robust body of evidence suggests that protein misfolding, insolubility, and aggregation are at the root of both cataracts and other diseases including AD, PD, and Huntington's disease,100 and that external EM energy (light) supplementation can have reparative effect on protein misfolding, activating and modulating metabolic control levels of protein folding/unfolding.10,34In addition, PBM effects on targets such as heat shock proteins (α crystalline), enzymes of the antioxidative system, Na+-K+-ATPase, Ca +2-ATPase, aquaporins (AQPs), and ion pumps have been referred to as part of mechanisms that could have influenced the response observed in the lens on the cases studied.46 We further proposed that, among other effects, PBM can stimulate and/or substitute ATP production via water dynamics, which is vital for the activation and inactivation kinetics in phototransduction.46 PBM can also affect the synthesis of molecules in a liquid crystalline (LC) state (e.g., self-assembly of lipids, water, and other biomolecules such as proteins and sterols, which are sensitive to temperature and/or electric fields) If confirmed, the latter may have multidisciplinary applications in medicine and biology in areas such as photovision, in which LCs are essential functional components.28
A first rapid communication referring to the retina and optic nerve additionally showed first evidence of EZ water as a selective rechargeable bio-battery applicable to PBM, suggesting a new understanding of the eye's energetic environment, which may have deep implications in ocular physiology as well as in the pathophysiology, diagnosis, and treatment of blinding diseases using light-based therapies.48 Therefore, as a promising alternative to drug therapies,101 or in combination with other treatments, PBM therapy may be developed into a viable therapeutic approach with multidisciplinary applications in ophthalmology and neuroscience,46 inducing and modulating physiologically reparative and regenerative effects that can favor homeostasis/homeokinesis27–29 through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by thermodynamics.
Treatment Costs and Availability
At the 2015 American Society of Clinical Oncology (ASCO) annual meeting, Dr. Leonard Saltz, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center, discussed the high cost of cancer drugs. He argued that “the unsustainably high prices of cancer drugs is a big problem, and it's our problem,” citing as examples the cost of nivolumab ($28.78/mg) and ipilimumab ($157.46/mg), which is “approximately 4000 times the cost of gold.”102 Previously, >100 oncologists had protested the high price of cancer drugs, also calling them economically “unsustainable.” They noted that, of 12 cancer drugs approved in 2012, 11 were priced > $100,000 per year,103 with multiple drugs often being required for extended periods. Such high prices and their impact on families, governments, and society at large are leading some to propose that cost should be considered a “financial toxicity” to be assessed with other toxicities when treatments are considered by doctors and patients.104 In contrast, although it has been estimated the cost of developing new drug therapies can run up to USD $1.3–$1.7 billion,105,106 the development cost of new photonics devices can be substantially lower, which can lower therapy costs and increase treatment availability. For the same reasons, PBM can also offer a noninvasive and cost-effective therapeutic option for patients with NDs of the retina, brain, and beyond.56, 85
Conclusions
The celebration of the IYL 2015 by the United Nations1 is a fitting time to announce what we describe here as a “quantum leap” in PBM therapy. It is also a good opportunity to ensure that policy makers and the medical community become aware of and embrace the immense potential of light-based medical technologies, especially PBM therapy, as an emerging treatment option for cancer and other complex diseases.107 Although not all tissues respond to PBM therapy,69 in vitro and in vivo xenografts and evidence from clinical studies does suggest that it is time to begin considering PBM therapy as a potential drug equivalent.11,108 In addition, PBM therapy may have minimal or no adverse effects, improve quality of life and functional status and raise the current standard of care for many cancer patients when used alone or in combination with other therapies.9 PBM therapy further represents a novel hope for the treatment of numerous eye and neurologic diseases. And as stated, PBM may be developed at a lower cost than many current treatments,8,10 which can help meet the healthcare needs of an increasing and aging world population. As such, this perspective and mini-review focuses on the large potential tangible contributions of light-based therapies for large demographic segments of the population, such as aging “baby boomers” who are expected to face a higher incidence of diseases such as cancer, AMD, DR, glaucoma, RP, AD, and PD, as well as other neurologic diseases in the next 15 years. In light of the growing costs of drugs and their impact on developed and developing countries, we propose that PBM therapy may offer a novel, safe, and effective therapy choice that would be more accessible to large vulnerable groups, such as the poor and the elderly.
Concurrently with the United Nations' declaration of 2015 as the year of light and light-based technologies, PBM therapy stands at the brink of delivering a new generation of treatments for complex diseases. New PBM therapies will preserve quality of life and raise standard of care in an efficient and cost-efficient manner. This will particularly benefit the most vulnerable demographic sectors, such as the elderly and the poor, and reduce the strain of growing healthcare costs in both industrialized and developing countries. We propose that such developments and their imminent impact represent a paradigm shift or “quantum leap” in PBM therapy and medicine at large.
Acknowledgments
We thank Jesús Alberto Santana-Rodríguez for reviewing and editing this article, and Luis Rafael Santana-Rodríguez for design and technical support. This study was supported by Fundalas, Foundation for Interdisciplinary Research and Development.
Author Disclosure Statement
No competing financial interests exist.
References
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4782038/
Laser Phototherapy Clinical Practice and Scientific Background
Lars Hode and Jan Tunér - 2014 (Book) 4328
This book is one of the most comprehensive resources for European style laser therapy.
View Resource
This book covers an astonishing amount of information in its near thousand pages, everthing from basic laser physics to dental, and veteranary useage. Here are some of its contents:

- Basic Laser Physics
- physics
- energy
- radiation
- wavelength and frequency
- photon energy
- the elecromagnetic spectrum
- the optical reigon
- radiation risks
- can electromagnetic radiation cause cancer
- protective mechanisms
- light
- the optical spectrum
- light sources
- various sources of radiation
- natural sources of radiation
- man-made light sources
- the light emmiting diode (LED)
- flash lamps
- the laser
- laser design
- practical lasers
- the properties of laser light coherence
- interference
- laser beam characteristics
- polarisation
- output power
- continuous and pulsed lasers
- the peak power value
- average power output
- power density
- light distribution
- beam divergence
- collimation
- risk of eye injury
- decisive factors in the risk of eye injury
- the laser instrument
- properties of some laser types
- description of common surgical laser types
- the CO2 laser (carbon dioxide laser)
- carbon dioxide lasers in surgery
- carbon dioxide lasers in dental applications
- the Nd:YAG laser
- Nd:YAG lasers in surgery
- Nd:YAG lasers in dentistry
- erbium lasers in dentistry
- "strong" diode lasers in dentistry
- the KTP laser
- Q-switching
- Theraputic Lasers
- the first generation 1975-85
- the second generation 1985-95
- the third generation 1995-2005
- the fourth generation 2005 and onwards
- what is a good laser therapy instrument
- the basic instrument
- sales tricks
- high power-low power
- laser or LED
- high or low price
- penetration of light into tissue
- "a story of a young scientist"
- the wavelength
- how deep does light penetrate into tissue?
- Biostimulation
- history
- a few words on mechanisms
- photoreceptors
- what parameters to use
- laser parameters
- whitch wavelength?
- output power
- average output power
- power density
- energy density
- the dose
- treatment dose
- calculation of doses
- dose ranges
- calculation of treatment time for a desired dose
- "reay reckoner"
- dose per point
- pulsed or continuous light
- pulse repetition rate (PRP)
- patient parameters
- treatment area
- treatment intervals
- pre- or postoperative treatment
- treatment method parameters
- local treatment
- shallow problems
- deeper problems
- treating inside the body
- systemic treatments
- acccupuncture
- trigger points
- spinal processes
- dermatome
- blood irradiation
- irradiation of lymph nodes
- irradiation of ganglions
- combo treatment
- interaction with medication
- other considerations
- what about collimation?
- depth of penetration, greatest active depth
- factors that reduce penetration
- tissue compression
- how deep does the light penetrate?
- laser light irradiation through clothes
- the importance of tissue and cell condition
- the importance of ambient light
- in vitro/ in vivo
- laser therapy with high output lasers
- laser therapy with carbon dioxide lasers
- laser therapy with Nd:YAG lasers
- laser therapy with ruby lasers
- laser therapy with Er:YAG lasers
- laser therapy with surgical diode lasers
- risks and side effects
- the importance of correct diagnose
- cancer
- cytogentic effects?
- a false picture of health
- tiredness
- pain reaction
- do high doses of laser therapy damage tissue?
- is it only an effect of temperature?
- protection against radiation injury
- how to measure effects of laser therapy
- thermography
- magnetic resonance imaging
- high resolution digitized ultrasound B-scan
- tensile strength
- other objective methods
- does it have to be a laser?
- FDA (Food and Drug Administration)
- how well documented?
- confused?
- the funding research
- as time goes by
- Medical indications
- who and what can be treated?
- acne
- allergy
- antibiotic resistance
- arteriosclerosis
- arthritis
- asthma
- blood preservation
- blood pressure
- bone regeneration
- burning mouth syndrome
- cancer
- cardiac conditions
- carpal tunnel syndrome
- cerebral palsy
- crural and venous ulcers
- delayed onset muscular soreness (DOMS)
- depression, psychosomatic problems
- diabetes
- duodenal/gastric ulcer
- epicondylitis
- erythema multiform major
- fibrositis/fribomyalgia
- headache/migraine
- heamorrhoids
- herpes simplex
- immune system modulation
- inflammation
- inner ear conditions
- laryngitis
- lichen
- low back pain
- mastitis
- microcirculation
- morbus sluder
- mucositis
- muscle regeneration
- mycosis
- nerve conduction
- nerve regeneration and function
- oedema
- ophthalmic problems
- pain
- periostitis
- plantar fasciitis
- salivary glands
- sinuitis
- spinal cord injuries
- snake bites
- sports injuries
- stem cells
- stroke, irradiation of the brain
- tendinopathies
- tinnitus, vertigo, meniere's disease
- tonsillitis
- trigeminal neuralgia
- thrombophlebitis
- tuberculosis
- urology
- warts
- wiplash-assosiated dissorders
- vitiligo
- womens' health
- wound healing
- zoster
- idications in the pipeline
- alzheimer's disease
- botox failures
- cellulites
- cholesterol reduction
- complex reigonal pain syndrom (CRPS)
- eczema
- erectile dysfunction
- familiar amyotrophic lateral sclerosis (FALS)
- glomerulonephritis
- obesity
- orofacial granulomatosis
- Parkinson's disease
- post-mestrual stress
- pemphigus vulgaris
- sleeping disorders
- withdrawal periods
- wrinkles
- consumer lasers
- Dental LPT
- the dental laser literature
- on which patients can LPT be used?
- dental indications
- alveolitis
- anaesthetics
- aphthae
- bleeding
- bisphosphonate related osteonecrosis of the jaw
- caries
- dentitio dificilis (pericoronitis)
- endodontics
- extraction
- gingivitus
- herpes zoster
- hypersensitive dentine
- implantology
- leukoplakia
- lingua geographica (glossitis)
- lip wounds
- nausea
- nerve injury
- orthodontics
- mild dental pain
- paediatric dental treatment
- periodontics
- prosthetics
- root fractures
- secondary dentine formations
- temperature caveats
- toemporo-mandibular disorders (TMD)
- TMD and endodontics
- other dental laser applications
- dental pohoto dynamic therapy
- composite curing
- deminerallisation
- tooth bleaching
- caries detection
- lasers as a diagnostic tool
- case reports
- Non Coherent Light Sources
- Veterinary Use
- case reports
- Contra Idications
- pacemakers
- pregnancy
- epilepsy
- thyroid gland
- children
- cancer
- haemophilia
- irradiation of the brain
- radiation therapy patients
- diabetes
- tatoos
- light sensitivity
- Coherence
- the role of coherence in laser phototherapy
- itroduction
- summary
- Dose and Intensity
- basics about energy
- output power
- power density
- the laser beam
- the laser probe
- pulsed lasers
- energy density
- treatment dose
- the dose does not demend on the intensity
- dose per point
- more about treatment technique
- The Mechanisms
- are biostimulative effects laser specific?
- is it possible to prove that laser therapy doesn't work?
- comparisons between coherent and non-coherent light
- what is the importance of the length of coherence
- hode's hamburger
- hode's big burger
- abrahamson's apple
- moonlight
- how deep does light penetrate tissue?
- bright light phototherapy
- similarities and differences
- possible primary mechanisms
- polarisation effects
- what characterises the light in a laser speckle
- porphyrins and polarised light
- cell cultures and tissue have different optical properties
- tthe effect of heat development in the tissue
- macroscopic heating
- the microscopic heat effect
- mechanical forces
- excitation effects
- primary reactions due to excitation
- secondary reactions due to cell signaling
- flourescence-luminescence
- multi-photon effects
- llasting effects in tissue
- non-linear optical effects
- opto-acoustic waves
- secondary mechanisms
- effects on pain
- effects on blood circulation
- stimulatory and regulatory mechanisms
- effects on the immune system
- other interesting possibilities
- summary of mechanisms
- diagnostics with therapeutic lasers
- photodynamic therapy - PDT
- other medical uses of lasers
- A Guide for Scientific Work
- methodology of a trial
- parameters
- technical parameters
- treatment parameters
- medical parameters
- closer description of the technical parameters
- name of instrument (producer)
- laser type and wavelength
- laser beam characteristics
- number of sources
- beam delivery system
- output power
- power density at probe aperture
- calibration of the instrument
- closer description of the treatment parameters
- treatment area
- dose: energy density
- dose per treatment and total dose
- intensity: power density
- treatment method
- treatment distance (spot size), type of movement, scanning
- sites of treatment
- number of treatment sessions
- frequency of treatment sessions
- closer description of the medical parameters
- description of the problem to be treated
- patients (number, age, sex)
- exclusion criteria
- inclusion criteria
- condition of patient
- pre-, parallel-, or post-medication
- treated with other methods before
- drop-out rates
- follow up
- outcome measures
- statistical analysis
- economy
- gallium-alluminium and all that
- recommendations of WALT - the world assosiation for laser therapy
- The Laser Phototherapy Literature
- the importance of reporting all laser parameters - even in the abstract
- diclofenac, dexamethasone or laser phototherapy?
- another pithole in LPT research
- database of abstracts of reviews of effects (DARE)
- the wound healing contradiction
- wikipedia
- poor documentation - compared to what?
- LPT equipment and the future
- english language books od LPT:
- books in other languages, with ISBN
- laser phototherapy journals
- information for your patient
Original Source: http://www.coldlasers.org/lllt-books/
Performance Chiropractic and Wellness: The Complete A-Z Manual for Low Level Laser Therapy 5th edition
Jerome Rerucha B.S., C.S.C.S., D.C. - 2015 (Book) 4332
Dr Rerucha is on the cutting edge at documenting how different pulsing frequencies can be used for different stilulatory effects. He works mainly with Erchonia.
View Resource

The Biological Basics of Low Level Laser Light Therapy
- summary
- introduction
- Alexander Gurwitsch: cells emit light
- non-linear dynamics
- introducing quantum physics
- itroduction to quantum biology
- quantum coherence in biology
- biological coherence and the sensitivity of living systems
- Fritz Albert Popp: biophotons
- Guenther Albreecht-Buehler: cells respont to light
- Mae-Wan Ho: visualizing coherence
- conclusions
Therapeutic Laser Applications
- how does low level laser therapy work?
- what are the advantages over other modes of therapy?
- cliniclal use of low level laser therapy
- abstract submitted to laser and surgury medicine
- background and objective
- methods
- results
- conclusion
- safety considerations
- eye considerations
- pace makers and other implanted devices
- pregnancy
- excessive toxicity
- preface to treatment section
Nerver Roots
- flexion and extension
- lateral flexion
- rotation
- MRT (muscle response testing) through ROM of cervical spine
- shoulder
- neurological level
- C5
- C6
- C7
- C8
- T1
- S1
- L5
- L4
- L3
- L3-L5
- L2-L4
- L1-L3
- low back
Top Ten Laser Protocols
- organ / glands / tissue
- acute injury (shock)
- pain
- lymphatic protocol
- detox protocol
- immune protocol
- hormone protocol
- basic cranial nerve
- tissue memory
- trauma preparation protocol
A-Z Laser Protocols
- abdominal cramping
- abdominal inflammation/pain
- abrasions
- abscess
- achilles tear / strain (partial only; not rupture)
- acidosis (hyperacidity
- acid reflux
- acne
- acute injury
- adenoids
- (ADD) atention deficit disorder and hyperactivity disorder (ADHD)
- Addiction
- addison's disease
- adhesions
- adhesive capsulitis
- adrenal
- aids
- allergies
- alopecia
- alpha waves
- alzheimer's
- amenorrhea
- amoebas
- amyotrophic lateral sclerosis / lou gehrig's disease / motor neuron
- amnesia
- anemia
- anger
- angina
- anosmia (loss of smell)
- anxiety appendicitis
- arrhythmias
- arteries / arteriosclerosis
- arthritis
- asthma
- ataxia
- athlete's foot
- atrophy
- backache / back pain
- bacteria
- bed sores
- bedwetting
- bell's palsy
- beta waves
- bites
- bladder
- bleeding gums
- bloating
- blood pressure (high)
- blood pressure (low)
- blood sugar balance
- boils
- bone
- bowel
- bradycardia
- brain
- breast augmentation
- bronchitis
- bruises
- buerger's disease
- bunions
- burns
- burns (second degree)
- bursitis
- calcium deposits or formations
- candida
- canker sores
- capsulitis
- carpal tunnel syndrome
- cartilage
- cataracts
- chemical peels / resurfacing
- chest pain
- chicken pox (herpes zoster / varicella)
- cholecystitis
- cholelithiasis
- chronic fatigue
- chronic pain
- circulation
- cirrhosis
- cold sores (herpes simplex 1)
- colds and flu
- colitis
- concussion
- confusion
- congestion
- congestive heart falure (CHF)
- conjunctivitis (pink eye)
- costipation
- cramps (muscle)
- cranial nerves (general)
- cranial nerves VIII
- crepitus
- crohn's disease
- cuts
- cushing's syndrome
- cytomegalovirus (herpes syndrome V)
- deer tick
- delta waves
- depression
- dermatitis
- detoxification
- diabetes
- diabetic neuropathy
- diabetic ulcers
- digestion
- dim vision
- disc herniation
- dizziness
- dupuytren's contracture
- dyslexia
- ear ache
- ear infection
- eczema
- edema
- emotional stress
- emphysema
- emulsification of fat
- endometriosis
- epistaxis
- epstein - barr virus
- esophagitis
- exercise recovery
- eye conditions
- facet syndrome
- facial paralysis
- fever
- fever blisters
- fibromyalgia
- flu
- food intolerance
- food poisoning
- foot fungus
- fracture
- fungus
- gait
- gallbladder (general)
- gallbladder (stones)
- ganglion cyst
- general musculoskeletal
- gerd
- gingivitis
- glaucoma
- goiter
- gout
- gums
- headache
- heart
- heartburn
- hearing difficulty
- hemorrhoids
- hepatitis A
- hepatitis B
- hepatitis C
- hernia
- herpes simplex
- herpes zoster (chickenpox / varicella)
- HIV
- hives
- hoarseness
- hormone balance
- hot flashes
- human papilloma virus (HPV)
- hyperactivity
- hyper/hypo-tension
- hyper/hypo-thyroid
- hyper/hypo-gycemia
- impotence
- immune enhancement
- incontinence
- indigestion
- infection
- inflammatory bowel disease
- inflammation
- influenza
- injuries
- insect bites
- irritable bowel syndrome
- ischemia
- jaundice
- joints
- keloid
- kidney
- kidey stones
- large intestine
- laryngitis
- ligament
- liposuction
- liver (balace and support)
- loss of smell (anosmia)
- loss of taste
- low back pain
- lungs
- lyme disease
- lymphadentis
- lymphatic
- macular degeneration
- memory problems
- meniere's disease
- meniscus sprain (grade 1)
- menopause
- mensturation
- mental fatigue
- meridian balance 15
- migraine
- motion sickness
- multiple sclerosis
- muscle
- muscle spasm
- myocardial inrarction
- nerve root
- neurogenic inflammation
- neuropathy
- nervousness
- nose bleed
- numbness
- nystagmus
- ocular motility disorders
- ocular nerve
- olfactory nerve
- osgood-schlatter disease
- otitis
- pain
- pain (chronic)
- pain (general)
- injury related pain (localized)
- pain (acute injury)
- pancreas
- parasite
- parasympathetic facilitazation
- paresthesia (numbness)
- periodontal disease
- pink eye (conjunctivitis)
- plantar fasciitis
- pneumonia
- polycystic kidney diseases
- polycystic ovary
- post operative scar revision
- post operative wound healing / pain
- post traumatic stress disorder (PTSD)
- postnasal drip
- premenstral syndrome (PMS)
- pre set head PL-touch
- pre-op
- prostate
- psoriasis
- punctures
- rash
- reflex sympathetic dystrophy (RSD)
- renal problems
- respiratory problems
- restless leg syndrome
- retinitis pigmentosa
- rheumatism
- ringworm
- road rash
- scar tissue
- sciatica
- sedation
- seizures
- shingles
- sinusitis
- skin
- sleep apnea
- small intesine
- smell - lack of
- sore throat
- soreness
- spasm
- spider veins
- spleen
- sprains
- spurs
- standars (neurological) setting
- stanard (up-regulation) setting
- staph infection
- stings
- stomach ulcer
- strep infections
- stress
- stroke
- sty
- subluxation
- sunburns
- swimmer's ear
- swollen ankles
- sympathetic calming
- tachycardia
- taste - lack of
- teeth
- tendonmyopathy (tendonitis)
- tension headaches
- theta waves
- thoratic outlet syndrome
- throat
- thrush
- thyroid (hyper)
- thyroid (hypo)
- tinnitus
- TMJ
- toenail fungus
- tonsilitis
- toothache
- ulcer
- ulcerative colotis
- up-regulation
- urinary tract infection
- varicose veins
- veins
- venereal warts
- viral infections
- voice
- vomiting
- water retention
- watery discharge from eye
- warts
- wounds
- yeast
Original Source: http://www.coldlasers.org/lllt-books/
Mechanisms and applications of the anti-inflammatory effects of photobiomodulation
Michael R Hamblin - PMC 2017 Jul 24 (Publication) 4519
Chronic diseases of the modern age involving systemic inflammation such as type II diabetes, obesity, Alzheimer's disease, cardiovascular disease and endothelial dysfunction are again worth investigating in the context of PBM.
View Resource
Abstract
Photobiomodulation (PBM) also known as low-level level laser therapy is the use of red and near-infrared light to stimulate healing, relieve pain, and reduce inflammation. The primary chromophores have been identified as cytochrome c oxidase in mitochondria, and calcium ion channels (possibly mediated by light absorption by opsins). Secondary effects of photon absorption include increases in ATP, a brief burst of reactive oxygen species, an increase in nitric oxide, and modulation of calcium levels. Tertiary effects include activation of a wide range of transcription factors leading to improved cell survival, increased proliferation and migration, and new protein synthesis. There is a pronounced biphasic dose response whereby low levels of light have stimulating effects, while high levels of light have inhibitory effects. It has been found that PBM can produce ROS in normal cells, but when used in oxidatively stressed cells or in animal models of disease, ROS levels are lowered. PBM is able to up-regulate anti-oxidant defenses and reduce oxidative stress. It was shown that PBM can activate NF-kB in normal quiescent cells, however in activated inflammatory cells, inflammatory markers were decreased. One of the most reproducible effects of PBM is an overall reduction in inflammation, which is particularly important for disorders of the joints, traumatic injuries, lung disorders, and in the brain. PBM has been shown to reduce markers of M1 phenotype in activated macrophages. Many reports have shown reductions in reactive nitrogen species and prostaglandins in various animal models. PBM can reduce inflammation in the brain, abdominal fat, wounds, lungs, spinal cord.
2.1. Cytochrome c oxidase in mitochondria
Cytochrome c oxidase (CCO) is unit IV in the mitochondrial electron transport chain. It transfers one electron (from each of four cytochrome c molecules), to a single oxygen molecule, producing two molecules of water. At the same time the four protons required, are translocated across the mitochondrial membrane, producing a proton gradient that the ATP synthase enzyme needs to synthesize ATP. CCO has two heme centers (a and a3) and two copper centers (CuA and CuB). Each of these metal centers can exist in an oxidized or a reduced state, and these have different absorption spectra, meaning CCO can absorb light well into the NIR region (up to 950 nm) [9]. Tiina Karu from Russia was the first to suggest [10,11], that the action spectrum of PBM effects matched the absorption spectrum of CCO, and this observation was confirmed by Wong-Riley et al in Wisconsin [12]. The assumption that CCO is a main target of PBM also explains the wide use of red/NIR wavelengths as these longer wavelengths have much better tissue penetration than say blue or green light which are better absorbed by hemoglobin. The most popular theory to explain exactly why photon absorption by CCO could led to increase of the enzyme activity, increased oxygen consumption, and increased ATP production is based on photodissociation of inhibitory nitric oxide (NO) [13]. Since NO is non-covalently bound to the heme and Cu centers and competitively blocks oxygen at a ratio of 1:10, a relatively low energy photon can kick out the NO and allow a lot of respiration to take place [14].
2.2. Light gated ion channels and opsins
More recently it has become apparent that another class of photoreceptors, must be involved in transducing cellular signals, particularly responding to blue and green light. Thee photoreceptors have been proposed to be members of the family of light-sensitive G-protein coupled receptors known as opsins (OPN). Opsins function by photoisomerization of a cis-retinal co-factor leading to a conformational change in the protein. The most well known opsin is rhodopsin (OPN1), which is responsible for mediating vision in the rod and cone photoreceptor cells in the mammalian retina. There are other members of the opsin family (OPN2-5), which are expressed in many other tissues of the body including the brain [15]. One of the best-defined signaling events that occurs after light-activation of opsins, is the opening of light-gated ion channels such as members of the transient receptor potential (TRP) family of calcium channels [16]. TRP channels are now known to be pleiotropic cellular sensors mediating the response to a wide range of external stimuli (heat, cold, pressure, taste, smell), and involved in many different cellular processes [17]. Activation of TRP causes non-selective permeabilization (mainly of the plasma membrane) to calcium, sodium and magnesium [18]. It is now known that TRP channel proteins are conserved throughout evolution and are found in most organisms, tissues, and cell-types. The TRP channel superfamily is now classified into seven related subfamilies: TRPC, TRPM, TRPV, TRPA, TRPP, TRPML, and TRPN [19]. Light-sensitive ion channels are based on an opsin chromophore (isomerization of a cis-retinal molecule to the trans configuration) as illustrated in Drusophila photoreceptors [20].
We have shown that blue or green light (but not red or 810 nm NIR) increased intracellular calcium in adipose derived stem cells, that could be blocked by ion channel inhibitors [5].
2.3. Flavins and flavoproteins
There is another well-known family of biological chromophores called cryptochromes. These proteins have some sequence similarity to photolyases [21], which are blue light responsive enzymes that repair DNA damage in bacteria caused by UV exposure [22]. Cryptochromes rely on a flavin (flavin adenine dinucleotide, FAD) or a pterin (5,10-methenyltetrahydrofolic acid) to actually absorb the light (again usually blue or green). Cryptochromes have been studied mainly in plants and insects. Recent evidence has emerged that mammalian cryptochromes are important in regulation of the circadian clock. It is thought that human cryptochromes (CRY1 and CRY2) send signals via part of the optic nerve to the suprachiasmatic nucleus (SCN) in the brain, which is the master regulator of the CLOCK system to entrain biological responses to the light-dark cycle [23]. However the situation is complicated because retinal ganglion cells containing melanopsin (OPN4) are also involved in photoentrainment [24]. Studies are still ongoing to investigate this redundancy [25].
It should be emphasized that compared to CCO and mitochondria, evidence is still emerging concerning the extent to which opsins, cryptochomes and light-gated ion channels (which may be widely expressed in many different cell types) could be responsible for PBM effects. If their role is significant it is likely to be in the blue and green spectral regions. Further research will be necessary to explore their role in anti-inflammatory effects, wound healing and tissue regeneration.
2.4. Water as a chromophore and heat-gated ion channels
Since the biological effects of light continue to be observed, as the wavelength increases in the infra-red region (>1000 nm), beyond those known to be absorbed by CCO, it is now thought likely that an alternative chromophore must be responsible. The obvious candidate for this alternative chromophore is water molecules whose absorption spectrum has peaks at 980 nm, and also at most wavelengths longer than 1200 nm. Moreover, water is by the far the most prevalent molecule in biological tissue (particularly considering its low molecule weight = 18). At present the proposed mechanism involves selective absorption of IR photons by structured water layers (also known as interfacial water) [26] or water clusters [27], at power levels that are insufficient to cause any detectable bulk-heating of the tissue. A small increase in vibrational energy by a water cluster formed in or on a sensitive protein such as a heat-gated ion channel, could be sufficient to perturb the tertiary protein structure thus opening the channel and allowing modulation of intracellular calcium levels [28]. Pollack has shown that interfacial water can undergo charge separation when it absorbs visible or NIR light [29]. This charge separation (equivalent to localized pH changes) could affect the conformation of proteins [30]. It has also been suggested that PBM could reduce the viscosity of interfacial water within the mitochondria, and allow the F0F1 ATP synthase, which rotates as a nanomotor to turn faster [31]. It should be noted here that the first regulatory approvals of PBM were gained as a 510 K device “equivalent to an non-heating IR lamp” [32]. While the involvement of water as a chromophore may still be considered hypothetical it is difficult to think of another explanation for the beneficial of PBM at wavelengths between 1000 nm all the way to 10,000 nm (carbon dioxide laser).
3.1. PBM increases ROS in normal cells
When PBM stimulates CCO activity in normal healthy cells, the resulting increase in mitochondrial membrane potential (MMP) above normal baseline levels, leads to a brief and rather modest increase in generation of reactive oxygen species (ROS) [33]. However this brief burst of ROS caused by 3 J/cm2 of 810 nm laser (Figure 2A) was shown to be sufficient to activate the redox-sensitive transcription factor, NF-kB in embryonic fibroblasts [34] (Figure 2B). Addition of the anti-oxidant N-acetyl-cysteine to the cells could block the NK-kB activation (Figure 2C), but not the increase in cellular ATP caused by the mitochondrial stimulation (Figure 2D). In primary cultured cortical neurons [35], 810 nm laser produced a biphasic dose response in ATP production (Figure 3A) and MMP (Figure 3B) with a maximum at 3 J/cm2. At a high dose (30 J/cm2) the MMP was actually lowered below baseline. Interestingly the dose-response curve between fluence (J/cm2) and ROS production showed two different maxima (Figure 3C). One of these maxima occurred at 3 J/cm2 where the MMP showed its maximum increase. The second maximum in ROS production occurred at 30 J/cm2 where the MMP had been reduced below baseline. At a value between these two fluences (10 J/cm2) a dose at which the MMP was approximately back to baseline, there was not much ROS generation. These data are very good examples of the “biphasic dose response” or “Arndt-Schulz curve” which is often discussed in the PBM literature [7,8].
Thus it appears that ROS can be generated within mitochondria when the MMP is increased above normal values and also when it is decreased below normal values. It remains to be seen whether these two kinds of PBM-generated ROS are identical or not. One intriguing possibility is that whether the ROS generated by PBM is beneficial or detrimental may depend on the rate at which it is generated. If superoxide is generated in mitochondria at a rate that allows superoxide dismutase (SOD) to detoxify it to hydrogen peroxide, then the uncharged H2O2 can diffuse out of the mitochondria to activate beneficial signaling pathways, while if superoxide is generated at a rate or at levels beyond the ability of SOD to deal with it, then the charged superoxide may build up inside mitochondria and damage them.
3.2. PBM reduces ROS in oxidative stressed cells and tissues
Notwithstanding, the ability of PBM to produce a burst of ROS in normal cells, it is well-accepted that PBM when as a treatment for tissue injury or muscle damage is able to reduce markers of oxidative stress [36,37,38]. How can these apparently contradictory findings be reconciled? A study attempted to answer this question [39]. Primary cultured cortical neurons were treated with one of three different interventions, all of which were chosen from literature methods of artificially inducing oxidative stress in cell culture. The first was cobalt chloride (CoCl2), which is used as a mimetic for hypoxia and works by a Fenton reaction producing hydroxyl radicals [40]. The second was direct treatment with hydrogen peroxide. The third was treatment with the mitochondrial complex I inhibitor, rotenone [41]. All three of these different treatments increased the intracellular mitochondrial ROS as judged by Cell-Rox Red (Figure 4A), and at the same time lowered the MMP as measured by tetramethyl-rhodamine methyl ester (TMRM) (Figure 4B). PBM (3 J/cm2 of 810 nm laser) raised the MMP back towards baseline, while simultaneously reducing the generation of ROS in oxidatively stressed cells (while slightly increasing ROS in normal cells). In control cells (no oxidative stress), PBM increased MMP above baseline and still produced a modest increase in ROS.
Since most laboratory studies of PBM as a therapy have looked at various animal models of disease or injury, it is not surprising that most workers have measured reduction in tissue markers of oxidative stress (TBARS) after PBM [36,42]. There have been a lot of studies looking at muscles. In humans, especially in athletes, high-level exercise produces effects in muscles characterized by delayed-onset muscle soreness, markers of muscle damage (creatine kinase), inflammation and oxidative stress.
One cellular study by Macedo et al [43] used muscle cells isolated from muscular dystrophy mice (mdx LA 24) and found that 5 J/cm2 of 830 nm increased the expression levels of myosin heavy chain, and intracellular [Ca2+]i. PBM decreased H2O2 production and 4-HNE levels and also GSH levels and GR and SOD activities. The mdx cells showed significant increase in the TNF-α and NFκB levels, which were reduced by PBM.
While it is highly likely that the effects of PBM in modulating ROS are involved in the anti-inflammatory effects of PBM, it would be dangerous to conclude that that is the only explanation. Other signaling pathways (nitric oxide, cyclic AMP, calcium) are also likely to be involved in reduction of inflammation.
As mentioned above we found [34] that PBM (3 J/cm2 of 810 nm laser) activated NF-kB in embryonic fibroblasts isolated from mice that had been genetically engineered to express firefly luciferase under control of an NF-kB promoter. Although it is well-known that NF-kB functions as a pro-inflammatory transcription factor, but on the other hand it is also well known that in clinical practice or in laboratory animal studies) PBM has a profound anti-inflammatory effect in vivo. This gives rise to another apparent contradiction that must be satisfactorily resolved.
4.2. PBM reduces levels of pro-inflammatory cytokines in activated inflammatory cells
Part of the answer to the apparent contradiction highlighted above, was addressed in a subsequent paper [44]. We isolated primary bone marrow-derived dendritic cells (DCs) from the mouse femur and cultured them with GM-CSF. When these cells were activated with the classical toll-like receptor (TLR) agonists, LPS (TLR4) and CpG oligodeoxynucleotide (TLR9), they showed upregulation of cell-surface markers of activation and maturation such as MHC class II, CD86 and CD11c as measured by flow cytometry. Moreover IL12 was secreted by CpG-stimulated DCs. PBM (0.3 or 3 J/cm2 of 810 nm laser) reduced all the markers of activation and also the IL12 secretion. Figure 5.
Yamaura et al [45] tested PBM (810 nm, 5 or 25 J/cm2) on synoviocytes isolated from rheumatoid arthritis patients. They applied PBM before or after addition of tumor necrosis factor-α (TNF-α). mRNA and protein levels of TNF-α and interleukins (IL)-1beta, and IL-8 were reduced (especially by 25 J/cm2).
Hwang et al [46] incubated human annulus fibrosus cells with conditioned medium obtained from macrophages (THP-1 cells) containing proinflammatory cytokines IL1β, IL6, IL8 and TNF-α. They compared 405, 532 and 650 nm at doses up to 1.6 J/cm2. They found that all wavelengths reduced IL8 expression and 405 nm also reduced IL6.
The “Super-Lizer” is a Japanese device that emits linear polarized infrared light. Imaoka et al [47] tested it against a rat model of rheumatoid arthritis involving immunizing the rats with bovine type II collagen, after which they develop autoimmune inflammation in multiple joints. The found reductions in IL20 expression in histological sections taken from the PBM-treated joints and also in human rheumatoid fibroblast-like synoviocyte (MH7A) stimulated with IL1β.
Lim et al [48] studied human gingival fibroblasts (HGF) treated with lipopolysaccharides (LPS) isolated from Porphyromonas gingivalis. They used PBM mediated by a 635 nm LED and irradiated the cells + LPS directly or indirectly (transferring medium from PBM treated cells to other cells with LPS). Both direct and indirect protocols showed reductions in inflammatory markers (cyclooxygenase-2 (COX2), prostaglandin E2 (PGE2), granulocyte colony-stimulating factor (GCSF), regulated on activated normal T-cell expressed and secreted (RANTES), and CXCL11). In the indirect irradiation group, phosphorylation of C-Raf and Erk1/2 increased. In another study [49] the same group used a similar system (direct PBM on HGF + LPS) and showed that 635 nm PBM reduced IL6, IL8, p38 phosphorylation, and increased JNK phosphorylation. They explained the activation of JNK by the growth promoting effects of PBM. Sakurai et al reported [50] similar findings using HGF treated with Campylobacter rectus LPS and PBM (830 nm up to 6.3 J/cm2) to reduce levels of COX2 and PGE2. In another study [51] the same group showed a reduction in IL1β in the same system.
4.3. Effects of PBM on macrophage phenotype
Another very interesting property of PBM is its ability to change the phenotype of activated cells of the monocyte or macrophage lineage. These cells can display two very different phenotypes depending on which pathological situation the cells are faced with. The M1 phenotype (classically activated) applies to macrophages that are faced with a situation in which bacteria or other pathogens need to be killed, or alternatively tumor cells need to be destroyed. Inducible nitric oxide synthase is a hallmark of the M1 phenotype and nitric oxide secretion is often measured. On the other hand the M2 phenotype (alternatively activated) applies to macrophages that are involved in disposal of cellular or protein debris and stimulation of healing by angiogenesis. The M2 phenotype produces arginase, an enzyme that inhibits NO production and allows them to produce ornithine, a precursor of hydroxyproline and polyamines [52]. The markers of these two phenotypes of activated macrophage have some aspects in common, but also show many aspects that are very different [53]. It should be noted that this concept of M1 and M2 activation states, applies to other specialized macrophage type cells that are resident in different tissues, such as microglia in brain [54], alveolar macrophages in lung [55], Kuppfer cells in liver [56], etc.
Fernandes et al used J774 macrophage-like cells activated with interferon-γ and LPS to produce a MI phenotype and compared 660 nm and 780 nm laser. They found that both wavelengths reduced TNF-α, COX-2 and iNOS expression, with the 780 nm being somewhat better [57]. Silva et al used RAW264.7 macrophages to test two wavelengths (660 nm and 808 nm) at a range of fluences (11-214 J/cm2) [58]. They found increases in NO release with 660 nm at the higher fluences. von Leden et al carried out an interesting study looking at the effects of PBM on microglia and their interaction with cortical neurons [59]. They used both primary microglia isolated from mouse brains and the BV2 mouse microglial cell line and compared four fluences (0.2, 4, 10, and 30 J/cm2, at 808 nm. Fluences between 4 and 30 J/cm2 induced expression of M1 markers in microglia. Markers of the M2 phenotype, including CD206 and TIMP1, were observed at lower energy densities of 0.2–10 J/cm2. In addition, co-culture of PBM or control-treated microglia with primary neuronal cultures demonstrated a dose-dependent effect of PBM on microglial-induced neuronal growth and neurite extension. This suggests that the benefits of PBM on neuroinflammation may be more pronounced at lower overall doses. The same group went on to show that M1 activated macrophages receiving PBM (660 nm laser) showed significant decreases in CCL3, CXCL2 and TNFα mRNA expression 4 h after irradiation [60]. However, 24 h after irradiation, M1 macrophages showed increased expression of CXCL2 and TNFα genes. M1 activated macrophages irradiated with 780 nm showed a significant decrease in CCL3 gene expression 4h after irradiation. These data could explain the anti-inflammatory effects of LLLT in wound repair.
This section will cover some of the most important medical indications where PBM has been shown in laboratory studies to be effective (at least partly) by its pronounced anti-inflammatory effects. Figure 6 shows a graphical summary of the anti-inflammatory applications of PBM in experimental animal models.
5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.
5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.

5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.
5.9. Abdominal fat
Yoshimura et al [87] looked at a mouse model of obesity and type 2 diabetes [87]. Four weeks old male adult C57BL/6 mice were fed a hypercaloric high-fat diet (40% calories derived from fat) for eight weeks to induce obesity and hyperglycemia. Over a period of four weeks mice were exposed to six irradiation sessions using an 843 nm LED (5.7 J cm−2, 19 mW cm−2). Non-irradiated control mice had areas of inflammation in their abdominal fat almost five times greater than the PBM group. The PBM group had significantly lower blood glucose levels 24 hours after the last session.
Amongst the many hundreds of reports of clinical applications of PBMT, we will highlight a few here, which seem to be especially relevant to inflammation, and inflammatory disorders.
6.1. Achilles tendinopathy
Bjordal et al in Norway carried out a randomized, placebo controlled trial of PBM (904 nm, 5.4 J per point, 20 mW/cm2) for activated Achilles tendinitis [88]. In addition to clinical assessment, they used microdialysis measurement of peritendinous prostaglandin E2 concentrations. Doppler ultrasonography measurements at baseline showed minor inflammation shown by increased intratendinous blood flow, and a measurable resistive index. PGE2 concentrations were significantly reduced with PBM vs placebo. The pressure pain threshold also increased significantly.
6.2. Thyroiditis
Chavantes and Chammas in Brazil have studied PBM for chronic autoimmune thyroiditis. An initial pilot trial [89] used 10 applications of PBM (830 nm, 50 mW, 38–108 J/cm2), twice a week, using either the punctual technique (8 patients) or the sweep technique (7 patients). Patients required a lower dosage of levothyroxine, and showed an increased echogenicity by ultrasound. The next study [90] was a randomized, placebo-controlled trial of 43 patients with a 9-month follow-up. In addition to improved thyroid function they found reduced autoimmunity evidenced by lower thyroid peroxidase antibodies (TPOAb), and thyroglobulin antibodies (TgAb). A third study [91] used color Doppler ultrasound to show improved normal vascualrization in the thyroid parenchyma. Finally [92] they showed a statistically significant increase in serum TGF-β1 levels 30 days post-intervention in the PBM group, thus confirming the anti-inflammatory effect. Recently a long-term follow up study of these thyroiditis patients (6 years later) was presented showing that PBM was safe in the long term and demonstrated lasting benefits [93].
6.3. Muscles
PBM for muscles aims to benefit athletic performance and training, to reduce delayed onset muscle soreness (DOMS), as well as to ameliorate signs of muscle damage (creatine kinase) after intense or prolonged exercise. Moreover PBM can also be used to treat frank muscle damage caused by muscle strains or trauma. The International Olympic Committee and the World Anti-Doping Agency cannot ban light therapy for athletes considering (1) the intensity is similar to sunlight, and (2) there is no forensic test for light exposure. There have been several clinical trials carried out in Brazil in athletes such as elite runners [94], volleyball players [95] and rugby players [96]. Ferraresi et al conducted a case-controlled study in a pair of identical twins [97]. They used a flexible LED array (850 nm, 75 J, 15 sec) applied to both quadriceps femoris muscles (real to one twin and sham to the other) immediately after each strength training session (3 times/wk for 12 weeks) consisting of leg press and leg extension exercises with load of 80% and 50% of the 1-repetition maximum test, respectively. PBM increased the maximal load in exercise and reduced fatigue, creatine kinase, and visual analog scale (DOMS) compared to sham. Muscle biopsies were taken before and after the training program and showed that PBM decreased inflammatory markers such as interleukin 1β and muscle atrophy (myostatin). Protein synthesis (mammalian target of rapamycin) and oxidative stress defense (SOD2, mitochondrial superoxide dismutase) were up-regulated.
6.4. Psoriasis
Psoriasis is a chronic autoimmune skin disease. Psoriasis is characterized by the abnormally excessive and rapid growth of keratinocytes (instead of being replaced every 28–30 days as in normal skin, in psoriatic skin they are replaced every 3–5 days). This hyperproliferation is caused by an inflammatory cascade in the dermis involving dendritic cells, macrophages, and T cells secreting TNF-α, IL-1β, IL-6, IL-17, IL-22, and IL-36γ [98]. PBM has been used for psoriasis because of its anti-inflammatory effects, which is a different approach from UV phototherapy which tends to kill circulating T-cells. Ablon [99] tested PBM using LEDs (830 nm, 60 J/cm2 and 633 nm, 126 J/cm2) in two 20-min sessions over 4 or 5 weeks, with 48 h between sessions in 9 patients with chronic treatment-resistant psoriasis. Clearance rates at the end of the follow-up period ranged from 60% to 100%. Satisfaction was universally very high.
Choi et al [100] tested PBM in case report of a patient with another inflammatory skin disease called acrodermatitis continua, who also had a 10-yr history of plaque psoriasis on her knees and elbows. As she was pregnant and not suited for pharmacological therapy, she received treatment with PBM (broad-band polarized light, 480–3,400 nm, 10 J/cm2). In two weeks (after only 4 treatments), the clinical resolution was impressive and no pustules were found. Topical methylprednisolone aceponate steroid cream was switched to a moisturizer, and she was treated twice or once a week with PBM until a healthy baby was delivered.
6.5. Arthritis
As can be seen from the animal studies section, arthritis is one of the most important clinical indications for PBM [101,102]. The two most common forms of arthritis are osteoarthritis (degenerative joint disease that mostly affects the fingers, knees, and hips) and rheumatoid arthritis (autoimmune joint inflammation that often affects the hands and feet). Osteoarthritis (OA) affects more than 3.8% of the population while rheumatoid arthritis (RA) affects about 0.24%. Both types have been successfully treated with PBM. Cochrane systematic reviews found for good evidence for its effectiveness in RA [103], and some evidence in the case of OA [104]. Most clinical studies have used pain scales and range of movement scores to test the effectiveness, rather than measures of inflammation which are difficult to carry out in human subjects.
Barabas and coworkers [105] made an attempt by testing PBM on ex vivo samples of synovial tissue surgically removed from patients receiving knee joint replacement. Synovial membrane samples received exposure to PBM (810 nm, 448 mW, 25 J/cm2, 1 cm2 area). PBM caused an increase in mitochondrial heat shock protein 1 60 kD, and decreases in calpain small subunit 1, tubulin alpha-1C, beta 2,vimentin variant 3, annexin A1, annexin A5, cofilin 1,transgelin, and collagen type VI alpha 2 chain precursor all significantly decreased compared to the control
6.6. Alopecia areata
Alopecia areata (AA) is one of the three common types of hair loss, the other two being androgenetic alopecia (AGA, male pattern baldness) and chemotherapy induced alopecia. AA is a common autoimmune disease resulting from damage caused to the hair follicles (HFs) by T cells. Evidence of autoantibodies to anagen stage HF structures is found in affected humans and experimental mouse models. Biopsy specimens from affected individuals demonstrate a characteristic peri- and intrafollicular inflammatory infiltrate around anagen-stage HFs consisting of activated CD4 and CD8 T lymphocytes [106]. PBM is an excellent treatment for hair loss in general and AGA in particular [107,108]. Yamazaki et al [109] reported the use of the “Super-Lizer” delivering linear-polarized light between 600–1600 nm at a power of 1.26 W to the areas of hair loss on the scalp (4-s pulses delivered at 1-s intervals for 3 min every 1 or 2 weeks until hair growth was observed). Regrowth of vellus hairs was achieved on more than 50% ofthe involved areas in all 15 cases. The frequency of irradiation until regrowth ranged from one to 14 times and the duration of SL treatment was 2 weeks to 5 months.
7. Conclusion and Future Studies
The clinical applications of PBM have been increasing apace in recent years. The recent adoption of inexpensive large area LED arrays, that have replaced costly, small area laser beams with a risk of eye damage, has accelerated this increase in popularity. Advances in understanding of PBM mechanisms of action at a molecular and cellular level, have provided a scientific rationale for its use for multiple diseases. Many patients have become disillusioned with traditional pharmaceutical approaches to a range of chronic conditions, with their accompanying distressing side-effects and have turned to complementary and alternative medicine for more natural remedies. PBM has an almost complete lack of reported adverse effects, provided the parameters are understood at least at a basic level. The remarkable range of medical benefits provided by PBM, has led some to suggest that it may be “too good to be true”. However one of the most general benefits of PBM that has recently emerged, is its pronounced anti-inflammatory effects. While the exact cellular signaling pathways responsible for this anti-inflammatory action are not yet completely understood, it is becoming clear that both local and systemic mechanisms are operating. The local reduction of edema, and reductions in markers of oxidative stress and pro-inflammatory cytokines are well established. However there also appears to be a systemic effect whereby light delivered to the body, can positively benefit distant tissues and organs.
There is a lot of scope for further work on PBM and inflammation. The intriguing benefits of PBM on some autoimmune diseases, suggests that this area may present a fertile area for researchers. There may be some overlap between the ability of PBM to activate and mobilize stem cells and progenitor cells, and its anti-inflammatory action, considering that one of the main benefits of exogenous stem cell therapy has been found to be its anti-inflammatory effect. The versatile benefits of PBM on the brain and the central nervous system, encourages further study of its ability to reduce neuroinflammation. Chronic diseases of the modern age involving systemic inflammation such as type II diabetes, obesity, Alzheimer's disease, cardiovascular disease and endothelial dysfunction are again worth investigating in the context of PBM.
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5523874/
Treatment of Neurodegeneration: Integrating Photobiomodulation and Neurofeedback in Alzheimer's Dementia and Parkinson's: A Review
Marvin H Berman, Trent W Nichols - (Publication) 4529
View Resource
Objective: A review of photobiomodulation (PBM) in Alzheimer's dementia is submitted. The addition of PBM in neurodegenerative diseases is a dual modality that is at present gaining traction as it is safe, antiviral, and anti-inflammatory for treating neurodegeneration with photons that stimulate mitochondria increasing adenosine triphosphate and proteasomes increasing misfolded protein removal. Neurofeedback provides neural plasticity with an increase in brain-derived nerve factor mRNA and an increase in dendrite production and density in the hippocampus coupled with overall growth in dendrites, density, and neuronal survival. Background: Alzheimer's disease pathophysiology is the accumulation of hyperphosphorylated tau protein neurofibrillary tangles and subsequently amyloid-beta plaques. PBM and neurobiofeedback (NBF)address the multiple gene expression and upregulation of multiple pathogenic pathway inflammation, reactive oxidative stress, mitochondrial disorders, insulin resistance, methylation defects, regulation of neuroprotective factors, and regional hypoperfusion of the brain. There is no human evidence to suggest a clinical therapeutic benefit from using consistent light sources while significantly increasing safety concerns. Methods: A PBM test with early- to mid-Alzheimer's was reported in 2017, consisting of a double-blind, placebo-controlled trial in a small pilot group of early- to mid-dementia subjects under Institutional Review Board (IRB)-approved Food and Drug Administration (FDA) Clinical Trial. Results: PBM-treated subjects showed that active treatment subjects tended to show greater improvement in the functioning of the executive: clock drawing, immediate recall, practical memory, and visual attention and task switching (Trails A&B). A larger study using the CerebroLite helmet in Temple Texas again of subjects in a double-blind, placebo-controlled IRB-approved FDA Clinical Trial demonstrated gain in memory and cognition by increased clock drawing. Conclusions: Next-generation trials with the Cognitolite for Parkinson's disease subjects will incorporate the insights regarding significant bilateral occipital hypocoherence deficits gained from the quantitative EEG analyses. Future applications will integrate noninvasive stimulation delivery, including full-body and transcranial and infrared light with pulsed electromagnetic frequencies.
Original Source: https://pubmed.ncbi.nlm.nih.gov/31647776/
Low-level laser therapy ameliorates disesase progression in a mouse model of multiple sclerosis.
Elaine D. Goncalves, Priscila S. Souiza, Vicente Lieberknecht, Giulia S. P. Fidelis, Rafael I. Barbosa, Paulo C. L. Silveria, Ricardo A. de Pinho, Rafael C. Dutra - Taylor & Francis Online 12/2015 (Publication) 4484
View Resource
Abstract
Multiple sclerosis (MS) is an autoimmune demyelinating inflammatory disease characterized by recurrent episodes of T cell-mediated immune attack on central nervous system (CNS) myelin, leading to axon damage and progressive disability. The existing therapies for MS are only partially effective and are associated with undesirable side effects. Low-level laser therapy (LLLT) has been clinically used to treat inflammation, and to induce tissue healing and repair processes. However, there are no reports about the effects and mechanisms of LLLT in experimental autoimmune encephalomyelitis (EAE), an established model of MS. Here, we report the effects and underlying mechanisms of action of LLLT (AlGaInP, 660 nm and GaAs, 904 nm) irradiated on the spinal cord during EAE development. EAE was induced in female C57BL/6 mice by immunization with MOG35–55 peptide emulsified in complete Freund’s adjuvant. Our results showed that LLLT consistently reduced the clinical score of EAE and delayed the disease onset, and also prevented weight loss induced by immunization. Furthermore, these beneficial effects of LLLT seem to be associated with the down-regulation of NO levels in the CNS, although the treatment with LLLT failed to inhibit lipid peroxidation and restore antioxidant defense during EAE. Finally, histological analysis showed that LLLT blocked neuroinflammation through a reduction of inflammatory cells in the CNS, especially lymphocytes, as well as preventing demyelination in the spinal cord after EAE induction. Together, our results suggest the use of LLLT as a therapeutic application during autoimmune neuroinflammatory responses, such as MS.
Introduction
Multiple sclerosis (MS) is an inflammatory chronic autoimmune and neurodegenerative disorder of the human central nervous system (CNS), in which encephalitogenic Th1 and Th17 lymphocytes induce a response against components of myelin [1–3 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747
Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145
Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407 ]. Inflammation, gliosis and axonal injury are additional prominent neuropathological characteristics, as is the clinical evolution from intermittent attacks to slow, steady progressive worsening [4 Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, some evidence points to an important role for nitric oxide (NO) in the pathogenesis of MS and to its contribution to the various facets of the disorder, including inflammation, oligodendrocytes injury, changes in synaptic transmission, axonal degeneration and neuronal death [5 Smith, K. J., and H. Lassmann. 2002. The role of nitric oxide in multiple sclerosis. Lancet Neurol. 1: 232–241[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
Patients with MS typically present between the ages of 20 and 40 years, with affected women outnumbering men 2:1, and the progressive phase of disease manifests at any time between 5 and 35 years after onset [4 Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. MS causes a multitude of symptoms, including visual disturbances, spasticity, weakness, impairment of walking, coordination difficulties, tremor/ataxia, sensory problems and bladder disturbances [1 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747[Crossref], [PubMed], [Web of Science ®], [Google Scholar],6 McFarland, H. F., and R. Martin. 2007. Multiple sclerosis: a complicated picture of autoimmunity. Nat. Immunol. 8: 913–919[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, “invisible” symptoms, such as fatigue, neuropathic pain and cognitive deficits, are also common [7–9 Shi, J., C. B. Zhao, T. L. Vollmer, et al. 2008. APOE epsilon 4 allele is associated with cognitive impairment in patients with multiple sclerosis. Neurology 70: 185–190
Rao, S. M., G. J. Leo, L. Bernardin, and F. Unverzagt. 1991. Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41: 685–691
Rao, S. M. 1995. Neuropsychology of multiple sclerosis. Curr. Opin. Neurol. 8: 216–220 ] and are detectable even before a definitive diagnosis of MS is made. These sensorial, cognitive and emotional symptoms related to MS strongly impact on family, social and work activities, as well as their quality of life [10 Engel, C., B. Greim, and U. K. Zettl. 2007. Diagnostics of cognitive dysfunctions in multiple sclerosis. J. Neurol. 254: II30–II34[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. MS manifests in several forms, like as: (i) clinically isolated syndrome (CIS) is the first manifestation of MS-like signs and symptoms, usually followed by another attack at which a clinical diagnosis of MS is made; (ii) relapsing remitting MS (RRMS), which is characterized by sudden relapses punctuated by short- or long-term remissions; (iii) secondary progressive MS (SPMS), which has a progressive course resulting in severe, irreversible debilitation and (iv) primary progressive MS (PPMS), which is a progressive type of MS without an initial relapsing and remitting period [11 Confavreux, C., and S. Vukusic. 2006. Natural history of multiple sclerosis: a unifying concept. Brain J. Neurol. 129: 606–616[Crossref], [Web of Science ®], [Google Scholar]]. The economic cost of MS associated with relapses and subsequent disability is considerable. For instance, a multicenter study initially carried out in five European countries examined the costs associated with MS, and the annual cost for those with expanded disability status scale (EDSS) ≤3 ranged from E 13.534 to E 22.561 increasing to E 28.524–E 43.984 for EDSS 4–6.5 and E 39.592–E 65.395 for EDSS ≥7 [12 Karampampa, K., A. Gustavsson, C. Miltenburger, and B. Eckert. 2012. Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from five European countries. Mult. Scler. 18: 7–15[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and loss of earnings was the biggest contributor to indirect costs [13 O'Connell, K., S. B. Kelly, E. Fogarty, et al. 2014. Economic costs associated with an MS relapse. Mult. Scler. Relat. Disord. 3: 678–683[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In this context, phototherapy, especially laser, has been widely used in research of different tissues, such as tendons, nerves, skin tissue, bones, muscles and CNS [14–18 Baroni, B. M., R. Rodrigues, B. B. Freire, et al. 2015. Effect of low-level laser therapy on muscle adaptation to knee extensor eccentric training. Eur. J. Appl. Physiol. 115: 639–647
Barbosa, R. I., A. M. Marcolino, R. R. de Jesus Guirro, et al. 2010. Comparative effects of wavelengths of low-power laser in regeneration of sciatic nerve in rats following crushing lesion. Lasers Med. Sci. 25: 423–430
Batista, J. D., S. Sargenti-Neto, P. Dechichi, et al. 2015. Low-level laser therapy on bone repair: is there any effect outside the irradiated field? Lasers Med. Sci. 30: 1569–1574
Allahverdi, A., D. Sharifi, M. A. Takhtfooladi, et al. 2015. Evaluation of low-level laser therapy, platelet-rich plasma, and their combination on the healing of Achilles tendon in rabbits. Lasers Med. Sci. 30: 1305–1313
Hartzell, T. L., R. Rubinstein, and M. Herman. 2012. Therapeutic modalities – an updated review for the hand surgeon. J. Hand Surg. 37: 597–621 ].
Low-level laser therapy (LLLT) has been considered as an adjuvant clinical treatment [19 Carrasco, T. G., M. O. Mazzetto, R. G. Mazzetto, and W. MestrinerJr. 2008. Low intensity laser therapy in temporomandibular disorder: a phase II double-blind study. Cranio 26: 274–281[PubMed], [Google Scholar],20 Gavish, L., L. S. Perez, P. Reissman, and S. D. Gertz. 2008. Irradiation with 780 nm diode laser attenuates inflammatory cytokines but upregulates nitric oxide in lipopolysaccharide-stimulated macrophages: implications for the prevention of aneurysm progression. Lasers Surg. Med. 40: 371–378[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and its photomodulating, analgesic and direct interference effects on the neuroinflammatory process have drawn the attention of many researchers. LLLT can modulate a broad-spectrum of cellular processes, including: (i) protection from cell and tissue death; (ii) stimulation of healing and repair of injuries and (iii) reduction of pain, swelling and inflammation [21 Chung, H., T. Dai, S. K. Sharma, et al. 2012. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 40: 516–533[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. It has been reported that the effects of laser irradiation – called photobiomodulation – are related to light fluence [22 Wang, F., T. S. Chen, D. Xing, et al. 2005. Measuring dynamics of caspase-3 activity in living cells using FRET technique during apoptosis induced by high fluence low-power laser irradiation. Lasers Surg. Med. 36: 2–7[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Evidence suggests that red or near-infra-red light (at wavelengths that can penetrate tissue) is absorbed by mitochondrial chromophores, especially cytochrome c oxidase, leading to increased cellular respiration and ATP formation, and modulation of oxidative stress and NO production that together lead to the activation of signaling pathways and gene transcription [23 Chen, A. C., P. R. Arany, Y. Y. Huang, et al. 2011. Low-level laser therapy activates NF-kB via generation of reactive oxygen species in mouse embryonic fibroblasts. PLoS One 6: e22453[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The effect of LLLT on the brain has also been extensively investigated. Transcranially applied LLLT has been shown to have beneficial effects on Alzheimer’s disease (AD) mouse models, and on rats and rabbits post-stroke [24–26 Oron, A., U. Oron, J. Chen, et al. 2006. Low-level laser therapy applied transcranially to rats after induction of stroke significantly reduces long-term neurological deficits. Stroke 37: 2620–2624
De Taboada, L., J. Yu, S. El-Amouri, et al. 2011. Transcranial laser therapy attenuates amyloid-beta peptide neuropathology in amyloid-beta protein precursor transgenic mice. J. Alzheimers Dis. 23: 521–535
Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436 ]. Furthermore, LLLT-regulated microglial function through Src kinase – a non-receptor tyrosine kinase that is activated by oxidative events [27 Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] – and reduced long-term neurological deficits after traumatic brain injury (TBI) [28 Xuan, W., F. Vatansever, L. Huang, et al. 2013. Transcranial low-level laser therapy improves neurological performance in traumatic brain injury in mice: effect of treatment repetition regimen. PLoS One 8: e53454[Crossref], [PubMed], [Web of Science ®], [Google Scholar],29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. However, there have been no reports about the effects of irradiation on the autoimmune neuroinflammatory diseases, such as MS. Therefore, in the present study, we investigated the therapeutic potential of LLLT on experimental autoimmune encephalomyelitis (EAE) disease progression, an established model of MS. Most importantly, we attempted to elucidate some of the mechanisms through which LLLT modulates the pro-inflammatory environment of CNS.
Methods
Experimental animals
Experiments were conducted using female C57BL/6 mice (6–10 weeks of age). The mice were kept in groups of four to six animals per cage, maintained under controlled temperature (22 ± 1 °C) with a 12-h light/dark cycle (lights on at 07:00 h), and were given free access to food and water. All procedures used in the present study followed the Guide for the Care and Use of Laboratory Animals (NIH publication no. 85–23) and were approved by the Animal Ethics Committee of the Universidade Federal de Santa Catarina (CEUA-UFSC, protocol number PP00956) and Universidade do Extremo Sul Catarinense (CEUA-UNESC, protocol number 042/2014-1).
EAE induction and clinical evaluation
Active EAE was induced by subcutaneous immunization with 200 µg of myelin oligodendrocytes glycoprotein (MOG) peptide, amino acids 35–55 and 500 µg Mycobacterium tuberculosis extract H37Ra in complete Freund’s adjuvant oil, as previously described [30 Stromnes, I. M., and J. M. Goverman. 2006. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1: 1810–1819[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. All animals were also injected intraperitoneally on days 0 and 2 with 300 ng of Pertussis toxin. Non-immunized (naive) and EAE non-irradiated animals were used as controls. Mice were monitored and scored daily for clinical disease severity according to the standard 0–5 EAE grading scale: 0, unaffected; 1, tail limpness; 2, failure to right upon attempt to roll over; 3, partial paralysis; 4, complete paralysis and 5, moribund. The mean of the clinical scores and body weight (a parameter of health) of all mice within a given treatment group was determined daily, thereby yielding the mean clinical score and body weight change for that treatment group.
Laser treatment
The animals were randomly divided into four groups: (I) not immunized and untreated – naïve group; (II) immunized and untreated – EAE group; (III) immunized and treated with AlGaInP LLLT (660 nm) and (IV) immunized and treated with GaAs LLLT (904 nm). A two-laser diode (Ibramed™, São Paulo, Brazil) was used with the following parameters: (i) 660-nm wavelength (AsGaInP), mean power of 30 mW, continuous regime and beam area of 0.06 cm2. The laser irradiation was delivered with a fluency of 10 J/cm2 and energy of 0.6 J, with exposure time of 20 s for each position; (ii) 904-nm wavelength (GaAs), peak power of 70 W, pulsed regime (time of pulse 60 ns) and beam area of 0.10 cm2. The laser irradiation was delivered with a fluency of 3 J/cm2. The animals were irradiated during 30 days (starting on day 0 until day 30 post-immunization), with a total of six position of irradiation per day – laser radiation was timed to contact in six points located 0.5 cm distance between the points. The laser focus was positioned on the spinal cord at an angle of 90° to the skin according to a contact-point technique, and the gauging of the laser emission was conducted before and after completion of the experiments.
Biochemical assays
Nitric oxide
NO release was quantified using the Griess assay [31 Pang, Q., X. Hu, X. Li, et al. 2015. Behavioral impairments and changes of nitric oxide and inducible nitric oxide synthase in the brains of molarless KM mice. Behav. Brain Res. 278: 411–416[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. After EAE induction, the production of NO was determined by an assay for nitrite. Eight mice of each group were euthanized, and the inguinal lymph nodes, spinal cords and spleen were extracted. The lymph node, spinal cord and spleen were rapidly separated on an ice plate and weighed. The samples were incubated with Griess reagent (1% sulfanilamide in 0.1 mol/L HCl and 0.1% N-(1-naphthyl) ethylenediamine dihydrochloride) at room temperature for 10 min, and optical density of the assay samples was measured spectrophotometrically at 540 nm.
Oxidative damage to lipids
The levels of 2-thiobarbituric acid-reactive species (TBARS) are expressed as malondialdehyde (MDA) equivalents, as previously described [32 Draper, H. H., and M. Hadley. 1990. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186: 421–431[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Briefly, spinal cord and inguinal lymph nodes were mixed with 1 mL of 10% trichloroacetic acid and 1 mL of 0.67% thiobarbituric acid. Subsequently, the mixture was heated in a boiling water bath for 15 min. The amount of TBARS was determined by measuring absorbance at 532 nm, and the results are given in nanomoles of TBARS per milligram of protein.
Glutathione (GSH) levels
Samples of spinal cord were collected and maintained at −80 °C for at least 48 h. The sample was homogenized with 200 μL of 0.02 M EDTA. The homogenate was mixed with 25 μL of 10% trichloroacetic acid, and was homogenized three times over 15 min, followed by centrifugation (15 min× 1500g × 4 °C). The supernatant was added to 200 μL of 0.2 M TRIS buffer, pH 7.4 and 500 μM DTNB. Color development resulting from the reaction between DTNB and thiols reached a maximum in 5 min and was stable for more than 30 min. Absorbance was read at 412 nm after 10 min. A standard GSH curve was formed. The results are expressed as GSH per mg of protein [33 Borghi, S. M., A. C. Zarpelon, F. A. Pinho-Ribeiro, et al. 2014. Role of TNF-alpha/TNFR1 in intense acute swimming-induced delayed onset muscle soreness in mice. Physiol. Behav. 128: 277–287[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
Histopathological examination and assessment
For histopathological analysis, 30 days after EAE induction, animals were sacrificed and each portion of the lumbar spinal cord (L3–L5) was removed and fixed immediately in 10% neutral formalin buffer [formalin:phosphate buffer (0.01 M, pH 7.4) = 1:1] for 24 h. The spinal cord portions were subsequently processed by routine paraffin embedding, sectioned (5 -μm thickness) and mounted on glass slides. A deparaffinization protocol was carried out through a xylene-free method as previously described [34 Falkeholm, L., C. A. Grant, A. Magnusson, and E. Moller. 2001. Xylene-free method for histological preparation: a multicentre evaluation. Lab. Invest. 81: 1213–1221[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Hematoxylin–eosin (H&E)- or luxol fast blue (LFB)-stained slides were observed for immune cell infiltration and demyelination area, respectively. The settings used for image acquisition were identical for both control and experimental tissues, and representative images are presented. Four ocular fields per section (six to nine mice per group) were captured and a threshold optical density that best discriminated the nuclear staining of inflammatory cells (hematoxylin-eosin) or myelin (luxol fast blue) was obtained using NIH ImageJ 1.36 b imaging software (NIH, Bethesda, MD) and applied to all experimental groups The total pixel intensity was determined, and the data are expressed as optical density (O.D.).
ELISA assay
Spinal cord segments were homogenized in phosphate buffer containing 0.05% Tween® 20, 0.1 mM phenylmethylsulphonyl fluoride, 0.1 mM benzethonium chloride, 10 mM EDTA and 20 UI aprotinin A. The homogenate was centrifuged at 3000g for 10 min, and the supernatants were stored at −70 °C until further analysis. IFN-γ, IL-17 and IL-1β levels were estimated with ELISA kits from R&D Systems (Minneapolis, MN) according to the manufacturer’s recommendations.
Drugs and reagents
Pertussis toxin, phosphate-buffered saline (PBS) and complete Freund’s adjuvant oil were all purchased from Sigma Chemical Co. (St. Louis, MO). The MOG35–55 peptide (MEVGWYRSPFSRVVHLYRNGK) was obtained from EZBiolab (Carmel, IN) and the M. tuberculosis extract H37Ra from Difco Laboratories (Detroit, MI). The anti-mouse-IL-17, IFN-γ, IL-1β DuoSet kits were obtained from R&D Systems (Minneapolis, MN). Other reagents were all of analytical grade and were obtained from different commercial sources.
Statistical analysis
Results are presented as means ± SEM of measurements made on 6–9 mice per group per experiment, and are representative of one/two independent experiments without overlapping samples by evaluating the reproducibility of these results. One-way ANOVA followed by the Newman–Keuls test was used to compare the groups at each time-point when the parameters were measured at different times after the EAE induction. p values less than 0.05 (p < 0.05) were considered significant. The statistical analyses were performed using GraphPad Prism 4 Software (GraphPad Software Inc., San Diego, CA).
Results
LLLT alleviates symptoms and delays disease onset in EAE mice
C57BL/6 mice immunized with MOG35–55 developed EAE clinical symptoms after 7 days and reached a maximum mean clinical on day 30, when the incidence of clinical EAE was 100% and the average score was around 3.5 ± 0.5 (Figure 1A and Table 1). To test the prophylactic efficacy of laser during EAE, treatment starts from day 0 of induction. Compared with the untreated EAE group, AlGaInP 10 J/cm2 or GaAs 3 J/cm2 treatment significantly delayed disease onset (p < 0.001; Table 1) and decreased disease severity as measured by the mean maximal clinical score (2.0 ± 0.2 and 2.5 ± 0.5, respectively), with inhibition of 68 ± 2% (AlGaInP 10 J/cm2, Figure 1A and B) and 54 ± 5% (GaAs 3 J/cm2) (p < 0.0001; F = 48.05), based on the area under the curve (AUC), compared with the EAE-untreated group (Figure 1A and B; Table 1).
Figure 1. Low-level laser therapy attenuates the EAE disease process in C57BL/6 mice. Active EAE was induced in C57BL/6 mice by immunization with MOG35–55 on day 0. The clinical score (A), area under the curve (AUC) (B), body weight change (C) and delta (Δ) body weight gain or loss at the peak of disease (day 30 post-induction) (D) were evaluated in the naive group, the control group (EAE), in mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and in mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after immunization. The clinical symptoms were scored every day in a blinded manner and are expressed as the mean clinical score or as the AUC. Data points are presented as the mean ± SEM. Values of ##p < 0.001 versus naive group and **p < 0.001 versus EAE group (one-way ANOVA followed by post-hoc Newman–Keuls).

As previously described, animals with EAE tend to have a reduced body weight as a result of anorexia and deficient fluid uptake, which fit well with the severity of the clinical score [35 Mix, E., H. Meyer-Rienecker, and U. K. Zettl. 2008. Animal models of multiple sclerosis for the development and validation of novel therapies – potential and limitations. J. Neurol. 255: 7–14[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Next, we evaluated whether LLLT prevents the body weight change that is induced by EAE in mice. As expected, after EAE induction, a significant body weight loss was observed in the EAE mice compared with the naïve group (Figure 1C and D). Interestingly, a significant body weight gain was found in the EAE plus AlGaInP 10 J/cm2 (10 ± 2.5%; Figure 1D) group and the EAE plus GaAs 3 J/cm2 group (11 ± 3.0%; Figure 1D) (p < 0.01; F = 6.3) when compared with the EAE group.
LLLT down-regulates NO levels in the CNS and peripheral lymphoid tissue without affecting lipid peroxidation or the antioxidant defense during EAE
Excess amounts of NO are harmful for CNS function and are implicated in the pathophysiology of many neurologic diseases, such as MS, and the EAE model, in which NO is overproduced, mainly by innate immunity cells, such as macrophages and microglia [36–38 Ghasemi, M., and A. Fatemi. 2014. Pathologic role of glial nitric oxide in adult and pediatric neuroinflammatory diseases. Neurosci. Biobehav. Rev. 45: 168–182
Das, U. N. 2012. Is multiple sclerosis a proresolution deficiency disorder? Nutrition 28: 951–958
Miller, E. 2012. Multiple sclerosis. Adv. Exp. Med. Biol. 724: 222–238 ]. Thus, we investigated the effect of LLLT on the level of NO in the CNS and secondary lymphoid tissue of EAE-treated and untreated animals. In agreement with clinical signs, the concentration of NO in the spinal cord of the EAE mice was significantly increased (52 ± 25 µmol/mg of protein) compared with the control animals (Figure 2A). In contrast, both AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment down-regulated the NO level in the CNS of the EAE-treated animals, with a mean of 10 ± 5 and 15 ± 10 µmol/mg of protein, respectively (Figure 2A; p < 0.01; F = 7.15). Moreover, this upregulation was attenuated with LLLT (AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment) in the spleen tissue after EAE induction (p < 0.05 and p < 0.01 versus the healthy group; Figure 2C). However, compared with the untreated EAE group, LLLT did not significantly modulate NO in the lymph node (Figure 2B). In addition, LLLT failed to inhibit lipid peroxidation (Figure 3A and B; p < 0.08; F = 2.80 and p < 0.7; F = 0.38) or to restore the antioxidant defense (Figure 3C and D; p < 0.31; F = 1.28 and p < 0.45; F = 0.91) after EAE induction in the spinal cord and lymph node, respectively.
Figure 2. Low-level laser therapy selectively inhibits NO level in the CNS and peripheral lymphoid tissue of EAE mice. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA. The spinal lumbar cords (A), inguinal lymph nodes (B) and spleen (C) were obtained from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after immunization. The NO production was analyzed using the Griess assay. Data are presented as means ± SEM of 6–9 mice per group and are representative of two independent experiments. #p < 0.05 versus naïve group and **p < 0.001 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

Figure 3. Low-level laser therapy ameliorates EAE without affecting lipid peroxidation or the antioxidant defense. Animals were immunized with MOG35–55 peptide/CFA and pertussis toxin. Lumbar spinal cord and inguinal lymph node samples were collected from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after EAE induction for the determination of TBARS (panels A and B) and GSH (panels C and D) levels, respectively. Results are presented as means ± SEM of 6–9 mice/group, and are representative of two separate experiments.

LLLT limits the infiltration of immune cells to the CNS
The hallmark of EAE disease is the infiltration of inflammatory cells into the CNS, leading to neuronal and oligodendrocyte damage [39 Bogie, J. F., P. Stinissen, and J. J. Hendriks. 2014. Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol. 128: 191–213[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Therefore, we aimed to determine the effect of LLLT on the infiltration of inflammatory cells into the CNS after EAE induction. As shown in Figure 4, no inflammatory foci were detected in the naïve lumbar spinal cord; however, the untreated EAE mice showed profound infiltration of immune cells into the CNS, particularly in the white matter region (Figure 4A and B). Interestingly, treatment with AlGaInP 10 J/cm2 significantly reduced the infiltration of these inflammatory cells into the CNS (Figure 4A and B; p < 0.02; F = 4.36). In contrast, treatment with GaAs 3 J/cm2 only resulted in a moderate inhibition (Figure 4).
Figure 4. Low-level laser therapy blocks infiltration of mononuclear cells into the CNS during EAE pathology. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA plus pertussis toxin. At the peak of disease (day 30), animals were killed and the lumbar spinal cords from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm) were harvested for infiltration studies. Infiltration of mononuclear cells into spinal cords sections was examined by H&E staining (A), with magnification ×40, ×100 and ×400. Graphical representation of the inflammatory cells evaluated in the lumbar spinal cord (B). Specifically, four alternate 5 -µm sections (six to nine animals/group) of the white matter of the lumbar spinal cord were obtained between L4 and L6. Detail: inflammatory foci in the white matter after EAE induction. Data are presented as means ± SEM. #p < 0.05 versus naïve group and *p < 0.05 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

LLLT reduces the demyelination area
To investigate whether clinical improvement was accompanied by decreased neuropathology, we examined the demyelination area in longitudinal sections of the lumbar region of spinal cords by LFB staining 30 days post-immunization. Histological analysis of the spinal cord tissue sections from the healthy control mice showed an intact myelin sheath (Figure 5), whereas typical demyelination was observed in the EAE mice (Figure 5A and B). Again, AlGaInP 10 J/cm2 treatment remarkably attenuated CNS demyelination in the EAE mice (Figure 5A and B), while GaAs 3 J/cm2 failed to inhibit the demyelination area induced by EAE (Figure 5A and B). These data suggest the clinical relevance of LLLT, especially AlGaInP 10 J/cm2, in reducing EAE severity.
Figure 5. Low-level laser therapy inhibits CNS demyelination during EAE development. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA plus pertussis toxin. At the peak of disease (day 30), animals were killed and the lumbar spinal cords from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm) were harvested for demyelination studies. Demyelination areas in spinal cord sections were examined by luxol fast blue (LFB) staining (A), with magnification ×40 and ×100. Graphical representation of the CNS demyelination in lumbar spinal cord (B). Specifically, four alternate 5 -µm sections (six to nine animals/group) of the white matter of the lumbar spinal cord were obtained between L4 and L6. Detail: CNS demyelination in the white matter after EAE induction. Data are presented as means ± SEM. #p < 0.05 versus naïve group and *p < 0.05 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

LLLT attenuated production of pro-inflammatory cytokines during EAE pathology
To initiate CNS inflammation, myelin-specific T cells, especially Th17 and Th1 subsets, must be activated in the periphery, gain access to the CNS and then be reactivated by central APCs presenting self-antigen, initiating a cascade of events, including the secretion of cytokines/chemokines, which recruit macrophages to the sites of T-cell activation [3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, pro-inflammatory mediators secreted by macrophages/microglia, such as IL-1β, are important for both perpetuating inflammation and contributing to CNS tissue damage in EAE [40 Kuchroo, V. K., A. C. Anderson, H. Waldner, et al. 2002. T cell response in experimental autoimmune encephalomyelitis (EAE): role of self and cross-reactive antigens in shaping, tuning, and regulating the autopathogenic T cell repertoire. Ann. Rev. Immunol. 20: 101–123[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Here, pronounced increase in IL-17, IFN-γ and IL-1β levels was observed in the spinal cord after EAE-immunization (Figure 6). AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment markedly inhibited the upregulation of IL-17 (Figure 6A), IFN-γ (Figure 6B) and IL-1β (Figure 6C) in the CNS after EAE induction.
Figure 6. Low-level laser therapy inhibits production of pro-inflammatory cytokines during EAE pathology. The spinal cord was extracted and processed to estimate the levels of IL-17 (A), IFN-γ (B) and IL-1β (C) by ELISA in the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm). Data are presented as means ± SEM of 6–9 mice per group. #p < 0.05 and ##p < 0.01 versus naïve group; *p < 0.05 and **p < 0.001 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).
Discussion
MS is the prototypic autoimmune inflammatory disorder of the CNS for which no cure is presently known. T cells have a pivotal role in orchestrating the complex cascade of events in MS, which include chronic inflammation, primary demyelination and axonal damage. The adverse events associated with the widely used IFN-β, glatiramer acetate, fingolimod, and, more recently, dimethyl fumarate justifying the search for alternative and less detrimental therapies.
Herein, we show that LLLT reduced the clinical score of EAE and delayed the disease onset through down-regulation of NO levels in the CNS and peripheral lymphoid tissue. Interestingly, a significant body weight gain was found in the EAE plus AlGaInP group and the EAE plus GaAs group, when compared with the EAE group, which could be due to the modulation of leptin levels. In fact, recently, Burduli demonstrated that the combined treatment by means of low-intensity laser irradiation is accompanied by the normalization of the plasma leptin level, suppression of the inflammatory process and a significant improvement of the quality of life of the patients suffering from rheumatoid arthritis [41 Burduli, N. N., and N. M. Burduli. 2015. [The influence of intravenous laser irradiation of the blood on the dynamics of leptin levels and the quality of life of the patients presenting with rheumatoid arthritis]. Vopr. Kurortol. Fizioter. Lech. Fiz. Kult. 92: 11–13[PubMed], [Google Scholar]]. Therefore, further experiments are required to confirm whether or not LLLT modulates the leptin pathway during the development of EAE. In addition, these beneficial effects of LLLT seem to be associated with a block of the entry of the inflammatory cells (especially lymphocytes) into the CNS, as well as immune cell migration, the demyelinating process and production of pro-inflammatory cytokines, after EAE induction (see proposed scheme in Figure 7). These results are in accord with studies in rodent models demonstrating that LLLT: (i) improves cognitive functions in the progressive stages of a mouse model of AD [26 Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]; (ii) recovers short- and long-term (56 days) neurobehavioral functions and reduces brain lesion volume after TBI [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] and (iii) reduces the delayed-type hypersensitivity reaction to ovalbumin by down-regulation of pro-inflammatory mediators [42 Oliveira, R. G., A. P. Ferreira, A. J. Cortes, et al. 2013. Low-level laser reduces the production of TNF-alpha, IFN-gamma, and IL-10 induced by OVA. Lasers Med. Sci. 28: 1519–1525[Crossref], [PubMed], [Web of Science ®], [Google Scholar],43 de Oliveira, R. G., F. M. Aarestrup, C. Miranda, et al. 2010. Low-level laser therapy reduces delayed hypersensitivity reaction to ovalbumin in Balb/C mice. Photomed. Laser Surg. 28: 773–777[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Taken together, these studies demonstrate the biological effects of LLLT with different parameters, confirming the ample therapeutic window of LLLT in different clinical conditions. In the literature, there are a large number of experimental studies with LLLT, although few parameters are described in detail, which results in the comparison and consequent understanding of the mechanisms involved being difficult. In the present study, we used two wavelengths – 660 and 904 nm. In agreement with our data, Enwemeka reported that only 30% of published papers using LLLT reveal consistent information to determine the dose, or even reported inaccurate data [44 Enwemeka, C. S. 2008. Standard parameters in laser phototherapy. Photomed. Laser Surg. 26: 411[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Thus, further studies are required to verify the effectiveness of LLLT in MS.
Figure 7. Schematic representation of low-level laser therapy (LLLT) anti-inflammatory and immunosuppressive effects in an experimental model of MS. Preventive treatment with LLLT during the induction phase of EAE, an experimental model of MS, inhibits development and progression of disease, besides neuroinflammation and demyelinating process in the CNS. Together, LLLT immunomodulatory correlates to inhibition of NO and cytokines levels in the spinal cord after EAE induction. LLLT, low-level laser therapy; EAE, experimental autoimmune encephalomyelitis; MS, multiple sclerosis; BBB, blood–brain barrier; CNS, central nervous systems; MOG, myelin oligodendrocytes glycoprotein; Th cell, T helper lymphocyte.

It has been suggested that LLLT may affect the inflammatory system, although the basis for the immunosuppressive effects of LLLT is still unknown. It is possible that LLLT irradiation changes RNA expression at the level of mRNA or protein synthesis of pro-inflammatory mediators, such as IL-2, TNF-α, IFN-γ, ICAM-1 and COX-2, as well as enhancing peripheral endogenous opioid in rats [45–47 Mafra de Lima, F., M. S. Costa, R. Albertini, et al. 2009. Low level laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat bronchi segments in E. coli lipopolysaccharide-induced airway inflammation by a NF-kappaB dependent mechanism. Lasers Surg. Med. 41: 68–74
Hagiwara, S., H. Iwasaka, A. Hasegawa, and T. Noguchi. 2008. Pre-Irradiation of blood by gallium aluminum arsenide (830 nm) low-level laser enhances peripheral endogenous opioid analgesia in rats. Anesth. Analg. 107: 1058–1063
Matsumoto, M. A., R. V. Ferino, G. F. Monteleone, and D. A. Ribeiro. 2009. Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med. Sci. 24: 195–201 ], from immune cells. In fact, recently, Mozzati et al. demonstrated that superpulsed laser irradiation blocked down-regulation of IL-1β, IL-6, IL-10 and COX-2, and that this was associated with a reduction in the inflammatory process after tooth extraction [48 Mozzati, M., G. Martinasso, N. Cocero, et al. 2011. Influence of superpulsed laser therapy on healing processes following tooth extraction. Photomed. Laser Surg. 29: 565–571[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Specific wavelengths of light trigger different inflammatory pathways of immune cells, such as antigen-presenting cells (APCs, e.g. macrophages) [49 Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], which leads to increased infiltration into the tissues. The ability of macrophages to act as phagocytes is also modulated by the application of LLLT [49 Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The ability of LLLT to drain lymphatic cells can be explained by the direct effects of laser light on the production of cytokines, because laser light can penetrate to 50 mm below the tissue surface [50 Uebelhoer, N. S., and E. V. Ross. 2008. Introduction. Update on lasers. Semin. Cutan. Med. Surg. 27: 221–226[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Accumulated evidence now suggests that in the induction phase of EAE and MS disease (day 0–day 7), encephalitogenic T cells in the periphery become activated by a viral or another infectious antigen [1 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Here, we hypothesize that LLLT applied during the induction phase of EAE increased phagocytic activity, and thus reduced antigen presentation in draining lymphatic cells and consistently inhibited activation of encephalitogenic Th1 and Th17 cells during the presentation of myelin antigens in peripheral lymphoid organs. Consequently, these cells failed to differentiate, proliferate and migrate to the CNS effectively, an effect that abrogated the development of EAE. In agreement with our data, Farfara et al. showed that laser-induced CD11b-positive phagocytotic monocyte cells were associated with a significant reduction of brain amyloid load following a short period of treatment [26 Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
After peripheral activation, CD4+ T cells effectively enter the subarachnoid space by crossing the blood-cerebrospinal fluid (CSF) barrier in either the choroid plexus or the meningeal venules [2 Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145[Crossref], [PubMed], [Web of Science ®], [Google Scholar],3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, inside the CNS, the T cells are re-activated by MHC class II-expressing microglia, which express myelin epitopes [3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. These T cells are capable of producing pro-inflammatory mediators, such as cytokines and reactive oxygen species (ROS). The level of ROS, especially NO, is enhanced in MS [51 Koch, M., G. S. Ramsaransing, A. V. Arutjunyan, et al. 2006. Oxidative stress in serum and peripheral blood leukocytes in patients with different disease courses of multiple sclerosis. J. Neurol. 253: 483–487[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] and consequently causes increased permeability of the blood–brain barrier (BBB) [52 Kuhlmann, C. R., R. Tamaki, M. Gamerdinger, et al. 2007. Inhibition of the myosin light chain kinase prevents hypoxia-induced blood–brain barrier disruption. J. Neurochem. 102: 501–507[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Similarly, opening of the BBB and oxidative stress are known to be involved in the pathogenesis of EAE, the animal model of MS [53 van Horssen, J., G. Schreibelt, J. Drexhage, et al. 2008. Severe oxidative damage in multiple sclerosis lesions coincides with enhanced antioxidant enzyme expression. Free Radic. Biol. Med. 45: 1729–1737[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In this study, we extended and enriched these findings by demonstrating that LLLT inhibited the NO level in the CNS and peripheral lymphoid tissue, especially, spleen after EAE induction. On the other hand, LLLT did not modulate the production of NO in the inguinal lymph nodes, which could be due to either the EAE mice having less NO-producing cells or to a decreased NO-producing capability on a per cell basis in the regional lymph nodes. Thus, future studies will need to clarify this hypothesis, as well as to investigate whether LLLT could modulate, directly, these cells in lymph nodes after EAE induction. Interestingly, the beneficial effect of LLLT can be partially explained based on the rapid elevation of ATP content, as previously demonstrated after laser irradiation in the ischemic heart [54 Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Furthermore, increases in total antioxidants, angiogenesis, heat-shock protein content and anti-apoptotic activity following LLLT were previously found for ischemic heart and skeletal muscles [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar],54 Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301[Crossref], [PubMed], [Web of Science ®], [Google Scholar],55 Avni, D., S. Levkovitz, L. Maltz, and U. Oron. 2005. Protection of skeletal muscles from ischemic injury: low-level laser therapy increases antioxidant activity. Photomed. Laser Surg. 23: 273–277[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and can be suggested as possible processes that are also attenuated by LLLT in the EAE model.
Additionally, much attention has been paid to therapeutic strategies aimed at controlling microglia-mediated neurotoxicity. Recently, it has been debated whether He–Ne (632.8 nm) LLLT can activate a number of signaling pathways, including MAPK/ERK, Src, Akt and RTK/PKCs signaling pathways [56 Zhang, J., D. Xing, and X. Gao. 2008. Low-power laser irradiation activates Src tyrosine kinase through reactive oxygen species-mediated signaling pathway. J. Cell. Physiol. 217: 518–528[Crossref], [PubMed], [Web of Science ®], [Google Scholar],57 Zhang, L., D. Xing, X. Gao, and S. Wu. 2009. Low-power laser irradiation promotes cell proliferation by activating PI3K/Akt pathway. J. Cell. Physiol. 219: 553–562[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. A study by Song et al. employed a microglial activation model (BV2 cells plus lipopolysaccharide) and evaluated the LLLT-induced neuroprotective effect. They found that LLLT prevents Toll-like receptor (TLR)-mediated pro-inflammatory responses in microglia, characterized by down-regulation of pro-inflammatory cytokine expression and NO production [27 Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Results reported here indicate, for the first time to our knowledge, that LLLT preventive treatment produced a marked reduction in inflammatory cell recruitment into the spinal cord and effectively prevented demyelination areas in the EAE mice. According to our data, the neuroinflammatory process results in neuronal injury that may impair function in the CNS, and these results suggest a neuroprotective effect of LLLT, which can be observed in terms of reduced EAE development and severity of clinical scores.
The present study also indicates a possible preferable mode of laser use for LLLT application after EAE immunization. The 660-nm wavelength (AsGaInP 10 J/cm2) in continuous-pulse mode demonstrated a better outcome in the percentage of mice showing complete recovery compared with the 904-nm wavelength (GaAs 3 J/cm2). In agreement with our data, Oron et al. (LLLT 808 nm, GaAlAs) described the superiority of the 100-Hz laser compared to the 600-Hz frequency after closed-head injury (CHI), and suggested that this difference may be associated with a resonance effect between pulsed light and brain waves (such as α-waves and θ-waves) [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In addition, there is a higher elevation in ATP content in the rabbit brain when laser energy was applied in the 100-Hz mode compared with 600 Hz [58 Lapchak, P. A., and L. De Taboada. 2010. Transcranial near infrared laser treatment (NILT) increases cortical adenosine-5′-triphosphate (ATP) content following embolic strokes in rabbits. Brain Res. 1306: 100–105[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Thus, we can propose that the AsGaInP 10-J/cm2 mode is perhaps the preferable mode with which to obtain a beneficial effect after autoimmune and neurodegenerative diseases, such as MS. Finally, in agreement with our data, Hudson et al. used the LLLT at 808 and 980 nm (1 W/cm2), which was projected through bovine tissue samples ranging in thickness from 18 to 95 mm and power density measurements were taken for each wavelength at the various depths. Thus, the authors concluded that 808 nm of light penetrates as much as 54% deeper than 980 nm light in bovine tissue, although we have not found any data with another tissue, such as bone, skin, nerves or MS [59 Hudson, D. E., D. O. Hudson, J. M. Wininger, and B. D. Richardson. 2013. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed. Laser Surg. 31: 163–168[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, Byrnes et al. showed that LLLT at 810 nm can penetrate deep into the body and promote neuronal regeneration and functional recovery for spinal cord injury (SCI) [60 Byrnes, K. R., R. W. Waynant, I. K. Ilev, et al. 2005. Light promotes regeneration and functional recovery and alters the immune response after spinal cord injury. Lasers Surg. Med. 36: 171–185[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The noninvasive nature and almost complete absence of side effects encourage further studies in neuroscience. Usually every laser and light source has a therapeutic window, narrow or wide for a particular lesion or condition. In laser treatment side effects can be divided into: (i) immediate effects, which occur either immediately or within a few minutes or hours of laser treatment, occasionally related to improper technique and rarely related to an accident; (ii) late effects, excess fluence leads to epidermal erythema, superficial burn or deep dermal burn with incident scarring depending on the extent of injury and pigmentary changes and (iii) sequelae, which rarely occurs. Importantly, these adverse effects can be prevented or minimised by proper patient and lesion selection, proper parameter selection, test shots and stepping down on fluence [61 Patil, U. A., and L. D. Dhami. 2008. Overview of lasers. Indian J. Plastic Surg. 41: S101–S113[Crossref], [PubMed], [Google Scholar]].
In summary, the present study indicates that LLLT applied daily post-EAE induction to C57BL/6 mice markedly inhibits clinical signs, neuroinflammation and oxidative damage induced by encephalitogenic T lymphocytes and microglia in the CNS. Thus, LLLT may be a promising non-pharmacological disease-modifying therapy for the treatment of autoimmune conditions, such as MS.
References
- Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747
,

- Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145
,

- Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407
,

- Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142
,

- Smith, K. J., and H. Lassmann. 2002. The role of nitric oxide in multiple sclerosis. Lancet Neurol. 1: 232–241
,

- McFarland, H. F., and R. Martin. 2007. Multiple sclerosis: a complicated picture of autoimmunity. Nat. Immunol. 8: 913–919
,

- Shi, J., C. B. Zhao, T. L. Vollmer, et al. 2008. APOE epsilon 4 allele is associated with cognitive impairment in patients with multiple sclerosis. Neurology 70: 185–190
,

- Rao, S. M., G. J. Leo, L. Bernardin, and F. Unverzagt. 1991. Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41: 685–691
,

- Rao, S. M. 1995. Neuropsychology of multiple sclerosis. Curr. Opin. Neurol. 8: 216–220
,

- Engel, C., B. Greim, and U. K. Zettl. 2007. Diagnostics of cognitive dysfunctions in multiple sclerosis. J. Neurol. 254: II30–II34
,

- Confavreux, C., and S. Vukusic. 2006. Natural history of multiple sclerosis: a unifying concept. Brain J. Neurol. 129: 606–616
,

- Karampampa, K., A. Gustavsson, C. Miltenburger, and B. Eckert. 2012. Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from five European countries. Mult. Scler. 18: 7–15
,

- O'Connell, K., S. B. Kelly, E. Fogarty, et al. 2014. Economic costs associated with an MS relapse. Mult. Scler. Relat. Disord. 3: 678–683
,

- Baroni, B. M., R. Rodrigues, B. B. Freire, et al. 2015. Effect of low-level laser therapy on muscle adaptation to knee extensor eccentric training. Eur. J. Appl. Physiol. 115: 639–647
,

- Barbosa, R. I., A. M. Marcolino, R. R. de Jesus Guirro, et al. 2010. Comparative effects of wavelengths of low-power laser in regeneration of sciatic nerve in rats following crushing lesion. Lasers Med. Sci. 25: 423–430
,

- Batista, J. D., S. Sargenti-Neto, P. Dechichi, et al. 2015. Low-level laser therapy on bone repair: is there any effect outside the irradiated field? Lasers Med. Sci. 30: 1569–1574
,

- Allahverdi, A., D. Sharifi, M. A. Takhtfooladi, et al. 2015. Evaluation of low-level laser therapy, platelet-rich plasma, and their combination on the healing of Achilles tendon in rabbits. Lasers Med. Sci. 30: 1305–1313
,

- Hartzell, T. L., R. Rubinstein, and M. Herman. 2012. Therapeutic modalities – an updated review for the hand surgeon. J. Hand Surg. 37: 597–621
,

- Carrasco, T. G., M. O. Mazzetto, R. G. Mazzetto, and W. MestrinerJr. 2008. Low intensity laser therapy in temporomandibular disorder: a phase II double-blind study. Cranio 26: 274–281
,

- Gavish, L., L. S. Perez, P. Reissman, and S. D. Gertz. 2008. Irradiation with 780 nm diode laser attenuates inflammatory cytokines but upregulates nitric oxide in lipopolysaccharide-stimulated macrophages: implications for the prevention of aneurysm progression. Lasers Surg. Med. 40: 371–378
,

- Chung, H., T. Dai, S. K. Sharma, et al. 2012. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 40: 516–533
,

- Wang, F., T. S. Chen, D. Xing, et al. 2005. Measuring dynamics of caspase-3 activity in living cells using FRET technique during apoptosis induced by high fluence low-power laser irradiation. Lasers Surg. Med. 36: 2–7
,

- Chen, A. C., P. R. Arany, Y. Y. Huang, et al. 2011. Low-level laser therapy activates NF-kB via generation of reactive oxygen species in mouse embryonic fibroblasts. PLoS One 6: e22453
,

- Oron, A., U. Oron, J. Chen, et al. 2006. Low-level laser therapy applied transcranially to rats after induction of stroke significantly reduces long-term neurological deficits. Stroke 37: 2620–2624
,

- De Taboada, L., J. Yu, S. El-Amouri, et al. 2011. Transcranial laser therapy attenuates amyloid-beta peptide neuropathology in amyloid-beta protein precursor transgenic mice. J. Alzheimers Dis. 23: 521–535
,

- Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436
,

- Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219
,

- Xuan, W., F. Vatansever, L. Huang, et al. 2013. Transcranial low-level laser therapy improves neurological performance in traumatic brain injury in mice: effect of treatment repetition regimen. PLoS One 8: e53454
,

- Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407
,

- Stromnes, I. M., and J. M. Goverman. 2006. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1: 1810–1819
,

- Pang, Q., X. Hu, X. Li, et al. 2015. Behavioral impairments and changes of nitric oxide and inducible nitric oxide synthase in the brains of molarless KM mice. Behav. Brain Res. 278: 411–416
,

- Draper, H. H., and M. Hadley. 1990. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186: 421–431
,

- Borghi, S. M., A. C. Zarpelon, F. A. Pinho-Ribeiro, et al. 2014. Role of TNF-alpha/TNFR1 in intense acute swimming-induced delayed onset muscle soreness in mice. Physiol. Behav. 128: 277–287
,

- Falkeholm, L., C. A. Grant, A. Magnusson, and E. Moller. 2001. Xylene-free method for histological preparation: a multicentre evaluation. Lab. Invest. 81: 1213–1221
,

- Mix, E., H. Meyer-Rienecker, and U. K. Zettl. 2008. Animal models of multiple sclerosis for the development and validation of novel therapies – potential and limitations. J. Neurol. 255: 7–14
,

- Ghasemi, M., and A. Fatemi. 2014. Pathologic role of glial nitric oxide in adult and pediatric neuroinflammatory diseases. Neurosci. Biobehav. Rev. 45: 168–182
,

- Das, U. N. 2012. Is multiple sclerosis a proresolution deficiency disorder? Nutrition 28: 951–958
,

- Miller, E. 2012. Multiple sclerosis. Adv. Exp. Med. Biol. 724: 222–238
,

- Bogie, J. F., P. Stinissen, and J. J. Hendriks. 2014. Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol. 128: 191–213
,

- Kuchroo, V. K., A. C. Anderson, H. Waldner, et al. 2002. T cell response in experimental autoimmune encephalomyelitis (EAE): role of self and cross-reactive antigens in shaping, tuning, and regulating the autopathogenic T cell repertoire. Ann. Rev. Immunol. 20: 101–123
,

- Burduli, N. N., and N. M. Burduli. 2015. [The influence of intravenous laser irradiation of the blood on the dynamics of leptin levels and the quality of life of the patients presenting with rheumatoid arthritis]. Vopr. Kurortol. Fizioter. Lech. Fiz. Kult. 92: 11–13
,

- Oliveira, R. G., A. P. Ferreira, A. J. Cortes, et al. 2013. Low-level laser reduces the production of TNF-alpha, IFN-gamma, and IL-10 induced by OVA. Lasers Med. Sci. 28: 1519–1525
,

- de Oliveira, R. G., F. M. Aarestrup, C. Miranda, et al. 2010. Low-level laser therapy reduces delayed hypersensitivity reaction to ovalbumin in Balb/C mice. Photomed. Laser Surg. 28: 773–777
,

- Enwemeka, C. S. 2008. Standard parameters in laser phototherapy. Photomed. Laser Surg. 26: 411
,

- Mafra de Lima, F., M. S. Costa, R. Albertini, et al. 2009. Low level laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat bronchi segments in E. coli lipopolysaccharide-induced airway inflammation by a NF-kappaB dependent mechanism. Lasers Surg. Med. 41: 68–74
,

- Hagiwara, S., H. Iwasaka, A. Hasegawa, and T. Noguchi. 2008. Pre-Irradiation of blood by gallium aluminum arsenide (830 nm) low-level laser enhances peripheral endogenous opioid analgesia in rats. Anesth. Analg. 107: 1058–1063
,

- Matsumoto, M. A., R. V. Ferino, G. F. Monteleone, and D. A. Ribeiro. 2009. Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med. Sci. 24: 195–201
,

- Mozzati, M., G. Martinasso, N. Cocero, et al. 2011. Influence of superpulsed laser therapy on healing processes following tooth extraction. Photomed. Laser Surg. 29: 565–571
,

- Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855
,

- Uebelhoer, N. S., and E. V. Ross. 2008. Introduction. Update on lasers. Semin. Cutan. Med. Surg. 27: 221–226
,

- Koch, M., G. S. Ramsaransing, A. V. Arutjunyan, et al. 2006. Oxidative stress in serum and peripheral blood leukocytes in patients with different disease courses of multiple sclerosis. J. Neurol. 253: 483–487
,

- Kuhlmann, C. R., R. Tamaki, M. Gamerdinger, et al. 2007. Inhibition of the myosin light chain kinase prevents hypoxia-induced blood–brain barrier disruption. J. Neurochem. 102: 501–507
,

- van Horssen, J., G. Schreibelt, J. Drexhage, et al. 2008. Severe oxidative damage in multiple sclerosis lesions coincides with enhanced antioxidant enzyme expression. Free Radic. Biol. Med. 45: 1729–1737
,

- Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301
,

- Avni, D., S. Levkovitz, L. Maltz, and U. Oron. 2005. Protection of skeletal muscles from ischemic injury: low-level laser therapy increases antioxidant activity. Photomed. Laser Surg. 23: 273–277
,

- Zhang, J., D. Xing, and X. Gao. 2008. Low-power laser irradiation activates Src tyrosine kinase through reactive oxygen species-mediated signaling pathway. J. Cell. Physiol. 217: 518–528
,

- Zhang, L., D. Xing, X. Gao, and S. Wu. 2009. Low-power laser irradiation promotes cell proliferation by activating PI3K/Akt pathway. J. Cell. Physiol. 219: 553–562
,

- Lapchak, P. A., and L. De Taboada. 2010. Transcranial near infrared laser treatment (NILT) increases cortical adenosine-5′-triphosphate (ATP) content following embolic strokes in rabbits. Brain Res. 1306: 100–105
,

- Hudson, D. E., D. O. Hudson, J. M. Wininger, and B. D. Richardson. 2013. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed. Laser Surg. 31: 163–168
,

- Byrnes, K. R., R. W. Waynant, I. K. Ilev, et al. 2005. Light promotes regeneration and functional recovery and alters the immune response after spinal cord injury. Lasers Surg. Med. 36: 171–185
,

- Patil, U. A., and L. D. Dhami. 2008. Overview of lasers. Indian J. Plastic Surg. 41: S101–S113
,

-
Original Source: https://www-tandfonline-com.colorado.idm.oclc.org/doi/full/10.3109/08916934.2015.1124425?scroll=top&needAccess=true
Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease.
Farfara D1, Tuby H, Trudler D, Doron-Mandel E, Maltz L, Vassar RJ, Frenkel D, Oron U. - J Mol Neurosci. 2015 Feb;55(2):430-6. doi: 10.1007/s12031-014-0354-z. Epub 2014 Jul 4. (Publication) 401
PBMT increased conginitive ability in tests on rats.
View Resource
Intro: Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and β-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Aβ). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Aβ brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Background: Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and β-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Aβ). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Aβ brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Abstract: Abstract
Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and β-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Aβ). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Aβ brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24994540
Mitochondrial respiration as a target for neuroprotection and cognitive enhancement.
Gonzalez-Lima F1, Barksdale BR2, Rojas JC3. - Biochem Pharmacol. 2014 Apr 15;88(4):584-93. doi: 10.1016/j.bcp.2013.11.010. Epub 2013 Dec 4. (Publication) 614
View Resource
Intro: This paper focuses on brain mitochondrial respiration as a therapeutic target for neuroprotection and cognitive enhancement. We propose that improving brain mitochondrial respiration is an important future direction in research and treatment of Alzheimer's disease (AD) and other conditions associated with cognitive impairment and neurodegeneration. The central thesis is that supporting and improving brain mitochondrial respiration constitutes a promising neurotherapeutic principle, with potential applications in AD as well as in a wide variety of neuropsychological conditions. We propose three different interventional approaches to improve brain mitochondrial respiration based on (a) pharmacology, (b) photobiomodulation and (c) nutrition interventions, and provide detailed examples for each type of intervention. First, low-dose USP methylene blue is described as a pharmacological intervention that can successfully increase mitochondrial respiration and result in memory enhancement and neuroprotection. Second, transcranial low-level light/laser therapy with near-infrared light is used to illustrate a photobiomodulation intervention with similar neurometabolic mechanisms of action as low-dose methylene blue. Finally, a nutrition intervention to improve mitochondrial respiration is proposed by increasing ketone bodies in the diet. The evidence discussed for each intervention supports a fundamental neurotherapeutic strategy based on improving oxidative energy metabolism while at the same time reducing the pro-oxidant tendencies of the nervous system. Targeting brain mitochondrial respiration with these three types of interventions is proposed as part of a holistic neurotherapeutic approach to improve brain energy metabolism and antioxidant defenses. This strategy represents a promising new bioenergetics direction for treatment of AD and other neuropsychological disorders featuring cognitive impairment and neurodegeneration.
Background: This paper focuses on brain mitochondrial respiration as a therapeutic target for neuroprotection and cognitive enhancement. We propose that improving brain mitochondrial respiration is an important future direction in research and treatment of Alzheimer's disease (AD) and other conditions associated with cognitive impairment and neurodegeneration. The central thesis is that supporting and improving brain mitochondrial respiration constitutes a promising neurotherapeutic principle, with potential applications in AD as well as in a wide variety of neuropsychological conditions. We propose three different interventional approaches to improve brain mitochondrial respiration based on (a) pharmacology, (b) photobiomodulation and (c) nutrition interventions, and provide detailed examples for each type of intervention. First, low-dose USP methylene blue is described as a pharmacological intervention that can successfully increase mitochondrial respiration and result in memory enhancement and neuroprotection. Second, transcranial low-level light/laser therapy with near-infrared light is used to illustrate a photobiomodulation intervention with similar neurometabolic mechanisms of action as low-dose methylene blue. Finally, a nutrition intervention to improve mitochondrial respiration is proposed by increasing ketone bodies in the diet. The evidence discussed for each intervention supports a fundamental neurotherapeutic strategy based on improving oxidative energy metabolism while at the same time reducing the pro-oxidant tendencies of the nervous system. Targeting brain mitochondrial respiration with these three types of interventions is proposed as part of a holistic neurotherapeutic approach to improve brain energy metabolism and antioxidant defenses. This strategy represents a promising new bioenergetics direction for treatment of AD and other neuropsychological disorders featuring cognitive impairment and neurodegeneration.
Abstract: Abstract
This paper focuses on brain mitochondrial respiration as a therapeutic target for neuroprotection and cognitive enhancement. We propose that improving brain mitochondrial respiration is an important future direction in research and treatment of Alzheimer's disease (AD) and other conditions associated with cognitive impairment and neurodegeneration. The central thesis is that supporting and improving brain mitochondrial respiration constitutes a promising neurotherapeutic principle, with potential applications in AD as well as in a wide variety of neuropsychological conditions. We propose three different interventional approaches to improve brain mitochondrial respiration based on (a) pharmacology, (b) photobiomodulation and (c) nutrition interventions, and provide detailed examples for each type of intervention. First, low-dose USP methylene blue is described as a pharmacological intervention that can successfully increase mitochondrial respiration and result in memory enhancement and neuroprotection. Second, transcranial low-level light/laser therapy with near-infrared light is used to illustrate a photobiomodulation intervention with similar neurometabolic mechanisms of action as low-dose methylene blue. Finally, a nutrition intervention to improve mitochondrial respiration is proposed by increasing ketone bodies in the diet. The evidence discussed for each intervention supports a fundamental neurotherapeutic strategy based on improving oxidative energy metabolism while at the same time reducing the pro-oxidant tendencies of the nervous system. Targeting brain mitochondrial respiration with these three types of interventions is proposed as part of a holistic neurotherapeutic approach to improve brain energy metabolism and antioxidant defenses. This strategy represents a promising new bioenergetics direction for treatment of AD and other neuropsychological disorders featuring cognitive impairment and neurodegeneration.
Copyright © 2013 Elsevier Inc. All rights reserved.
Methods: Copyright © 2013 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24316434
Alzheimer's Research
Shannon Macauley - Scientific American (Publication) 4511
Combining light with sound show promise for treating Alzheimer's
View Resource
There is no cure for Alzheimer’s disease. Although a few drugs manage temporarily certain cognitive symptoms of the illness, none can stop or meaningfully slow its progression. “We really don’t have much to offer people,” says Shannon Macauley, a neuroscientist at Wake Forest School of Medicine. Virtually all new treatments have failed in clinical trials. But new research is looking beyond drugs to see what relief might come from a simple LED light and a speaker.
Bathing patients in flashing light and pulsing sounds both tuned to a frequency of 40 hertz might reverse key signs of Alzheimer’s in the brain, according to a paper published in Cell on Thursday. “I think it’s an absolutely fascinating paper to be honest,” says Macauley, who was not involved in this work. “It’s a very provocative idea. It’s noninvasive and easy and low cost, potentially, so if it were to come to fruition in humans—that’s fabulous.”
Still, all this is a big if, Macauley acknowledges. The work was done in mice with genetic alterations that doomed them to develop key symptoms and pathology of Alzheimer’s disease. One batch of mice formed neurofibrillary tangles inside their neurons—dysfunctional knots of a protein called tau that can lead to the cell’s death. Another batch of the mice developed amyloid beta plaques—sticky heaps of protein that dam the flow of communication between neurons. All the mice also had a third hallmark of the disease—irregular brain activity in the gamma range of brain waves that oscillate between 30 and 100 times a second.
In 2015 neuroscientist Li-Huei Tsai, director at The Picower Institute for Learning and Memory at Massachusetts Institute of Technology, was working on an experiment to manipulate that brain activity by flashing a white light at these mice. Like light strobes, our brains flicker. Brain waves are generated when large groups of neurons oscillate on and off together. Neurons encode our thoughts and actions and senses in this rhythmic electrical flutter. So when Tsai tuned her light to flash 40 times a second, or 40 hertz, and flickered it at the mice, their brains flickered back—generating gamma waves at a corresponding 40 hertz. Then, something unexpected happened.
When Tsai dissected the mice brains afterward, the amount of amyloid plaques and tau tangles in the mice that saw the light had plummeted. “It was the most remarkable thing,” Tsai says. “The light flicker stimulation triggers a tremendous microglia response. These are the brain’s immune cells that clear cell debris and toxic waste including amyloid. They’re impaired in Alzheimer’s disease, but [the light] seems to restore their abilities.”
This clearing-out process only happened in the visual cortex where the brain processes light information. To get these effects to penetrate deeper into the brain, she added a clicking sound like a dolphin’s chirrup that also had a 40-hertz frequency. When the mice sat in a room with both the flashing light and the droning sound for an hour day, seven days in a row, amyloid plaques and tau tangles began falling in not just the audio and visual cortices but the prefrontal cortex and the hippocampus as well. “This was one of the big jumps in the new paper,” Macauley says. “These are the learning and memory centers of the brain. And there was about a 40 or 50 percent decrease in amyloid and tau levels. It’s an absolutely impressive feat.”
That showed when Tsai put the mice through a set of cognitive tests. In one, where the mice were given a familiar and an unfamiliar object to explore, mice that didn’t get the treatment acted as though they’d never seen the familiar object. “That shows some memory problems,” Tsai says. Mice that saw the light and heard the sound spent about two thirds of the time that untreated mice did examining the familiar object. “It was unbelievable,” Tsai says. “This is the first time we’ve seen that this noninvasive stimulation can improve cognitive function. It’s not a drug or an antibody or anything, it’s just light and sound.”
One possible explanation for this is brains with Alzheimer’s have irregular, often hyperactive, neurons, says Jorge Palop, a neurologist at the University of California, San Francisco, who did not work on the study. By providing the brains with a steady and regular beat, the repeating light and sound might work as a kind of metronome for brain activity. “This could be like resetting the mice every day and correcting some of this abnormal activity that they have,” he says. “Then downstream of that are all these beneficial effects.”
Original Source: https://www.scientificamerican.com/article/an-hour-of-light-and-sound-a-day-might-keep-alzheimers-at-bay/
Nanoparticle-emitted light attenuates amyloid-β-induced superoxide and inflammation in astrocytes.
Bungart BL1, Dong L1, Sobek D2, Sun GY3, Yao G1, Lee JC4. - Nanomedicine. 2014 Jan;10(1):15-7. doi: 10.1016/j.nano.2013.10.007. Epub 2013 Nov 4. (Publication) 645
View Resource
Abstract
Alzheimer’s disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Keywords: Alzheimer’s disease, light nanomedicine, inflammation
Background
Many diseases, including AD, are characterized with aberrant cellular activities resulting from increased oxidative stress and inflammation (1, 2). In AD, Aβ when aggregated to its oligomeric form has the most cytotoxic effects (3). Toxic Aβ increases oxidative stress and triggers inflammatory responses in glial cells through increased expression of inflammatory proteins, such as interleukin-1β (IL-1β) and inducible nitric oxide synthase (iNOS) (4).
Low level light with wavelengths ranging from 632.8 to 904 nm have been reported to attenuate inflammation and oxidative stress in a number of models (5, 6). For example, we have previously demonstrated that a low level energy laser at 632.8 nm attenuated oxidative stress and inflammation in primary astrocytes induced by Aβ (5). However, the use of laser or LED light as a clinical therapy for the treatment of AD brains is limited due to the low penetration of light in tissues (7).
In this study, we demonstrate a new light delivery method by using a recently engineered nanoparticle, BRET-Qdot. This light delivery nanoparticle has been used in a number of in vivo applications (8) and exploits the F?ster resonance energy transfer from an engineered enzyme, Renilla Reniformis luciferase (Luc8) (9). When activated with its substrate, coelenterazine-h (coel-h), Luc8 is known to emit at 480 nm to a CdSe or CdTe quantum dot, which then re-emits the light at 800 nm. A typical BRET-Qdot construct incorporates 3 to 6 Luc8 molecules conjugated to the surface of CdTe quantum dots (Figure 1). Conceptually, this light delivery approach could potentially resolve difficulties in providing light from external sources to the central areas of the brain.
Methods
To test the efficacy of BRET-Qdots (Zymera, San Jose, CA, USA) as proposed, we used primary cortical rat astrocytes (Invitrogen, Grand Island, NY, USA) as in our previous study (5). The maximum fluency and duration of the BRET-Qdots’ emitted light was observed using a PIXIS 512 CCD camera (Princeton Instruments, Trenton, NJ, USA) in conjunction with a cold mirror with a 0° AOI (Edmund Optics Inc., Barrington, NJ, USA) to reflect >90% visible light. Then, the BRET-Qdots were used as the pretreatment prior to a 5 μM Aβ (American Peptide, Sunnyvale, CA, USA) exposure to astrocyte, followed by the characterizations of superoxide anion accumulation using fluorescence microscopy of dihydroethidium (DHE), and the expressions of IL-1β and iNOS using Western blot analysis. Detailed methodological information can be found in the Supplementary Materials.
Results
BRET-Qdot with coel-h attenuates Aβ-induced superoxide anion production
We test if the superoxide anion induced by Aβ can be reduced by 800 nm-emitting BRET-Qdots which preceded Aβ treatment. Figure 2 shows that exposing astrocytes to 5 μM Aβ for 2 h increased superoxide anion production by 40% as compared to the untreated control. Light pre-treatment using 66.7 pM BRET-Qdots activated with 2.5 μM coel-h reduces Aβ-induced superoxide anion production to untreated control levels. Either BRET-Qdots or coel-h individually reduces superoxide anion production on average compared to Aβ-treated cultures, but not statistically significant.
BRET-Qdot with coel-h pretreatment attenuates Aβ-induced inflammatory marker expression
Similarly to the superoxide anion experiment, we investigate if the light pretreatment with 66.7 pM BRET-Qdot activated with 2.5 μM coel-h reduce the inflammation contributed by 5 μM Aβ as measured by IL-1β and iNOS expression levels (Figure 3). Incubating astrocytes with 5 μM Aβ for 18 h increased IL-1β and iNOS expression by 30 and 50%, respectively. BRET-Qdot-based light pre-treatment lowered Aβ-induced expression of IL-1β to that of the untreated control, while Aβ-induced iNOS expression was reduced to below untreated control levels. Neither BRET-Qdot nor coel-h alone showed the same reduction in IL-1β or iNOS expression.
Discussion
Our previous study with astrocytes showed that application of 632.8 nm laser caused a decrease in oxidative stress and inflammation (5). However, red to NIR light cannot be transmitted through the scalp to the brain more than a few centimeters (7). This makes non-invasive LLLT for AD nearly impossible.
In this present study, we demonstrated the ability of a light-generating nanoparticle to attenuate Aβ-induced oxidative stress and inflammatory responses in astrocytes. The application of this light delivery method based on light-generating nanoparticles demonstrated a novel method for applying LLLT.
Some studies on the application of quantum dots in biological systems may consider the possible toxic effects of quantum dots due to their heavy metal content, nanoparticulate nature, or coating composition . Nevertheless, investigations of quantum dots in vivo have shown retention of the material in tissues from days to weeks with no major complications (10). Here we also show that exposing astrocytes to BRET-Qdots with coel-h for 2 and 12 h did not significantly lower cell viability as compared with control. (see Supplementary Materials, Fig. S2)
Results here show the advantages of light-generating nanoparticles as a concept to be further developed for in vivo light-delivery for LLLT. CdSe/CdTe quantum dots may be used further in animal models as non-toxic, NIR-emitting nanoparticles become available. In addition, even though this study only covered Aβ-induced inflammation and oxidative stress, as studied in the AD pathology, the LLLT delivered by light-generating BRET-dots may also be applied to other cellular systems and diseases involving oxidative stress and inflammation, such as diabetes (6). A light-generating nanoparticle-based therapy could potentially create the ability to offer LLLT in an in vivo setting to reduce the onset of these other pernicious illnesses.
Intro: Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Background: Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Abstract: Abstract
Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
FROM THE CLINICAL EDITOR:
Low Level Laser Therapy has already been demonstrated to mitigate amyloid-β peptide induced oxidative stress and inflammation, a key driver of Alzheimer's disease. The major issue in moving this forward from cell cultures to live animals and potentially to human subjects is light penetration depth. In this novel study, BRET-Qdots were used as an alternative near infrared light source with good efficacy, paving the way to the development of a nanomedicine platform.
Copyright © 2014 Elsevier Inc. All rights reserved.
Methods: Low Level Laser Therapy has already been demonstrated to mitigate amyloid-β peptide induced oxidative stress and inflammation, a key driver of Alzheimer's disease. The major issue in moving this forward from cell cultures to live animals and potentially to human subjects is light penetration depth. In this novel study, BRET-Qdots were used as an alternative near infrared light source with good efficacy, paving the way to the development of a nanomedicine platform.
Results: Copyright © 2014 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24200521
Quantitative In Vivo Imaging of Tissue Absorption, Scattering, and Hemoglobin Concentration in Rat Cortex Using Spatially Modulated Structured Light
David J. Cuccia, David Abookasis, Ron D. Frostig, and Bruce J. Tromberg. - 2009 (Book) 4502
View Resource
12.1. INTRODUCTION
Significant changes in blood flow or in the integrity of cerebral vessels are believed to cause cerebrovascular disease (CVD) and to contribute to dementias including Alzheimer’s disease [1]. Stroke, the most serious form of CVD, is one of the leading causes of death and adult disability worldwide. Acute treatments for stroke, however, are severely limited. Neuroprotective drugs under development show promise at halting the ischemic cascade, but as yet, no such compound has received federal approval in the United States. One of the biggest limitations to this development is the lack of understanding of the mechanisms by which cerebral vessels react to factors such as ischemia, inflammation, blood pressure changes, metabolic demands, and trauma [2]. In order to address these fundamental questions, functional brain imaging techniques such as fMRI and intrinsic signal optical imaging (ISOI) have emerged as tools to visualize and quantify cerebral hemodynamics.
In the neuroscience community, ISOI has long been used to study the organization and functional architecture of different cortical regions in animals and humans [3–5] (see other chapters in this book). Three sources of ISOI signals that affect the intensity of diffusely reflected light derive from characteristic physiologic changes in the cortex. For functional neuronal activation, these have been observed to occur over a range of timescales, including (1) light scattering changes, both fast (over 10 s of milliseconds) and slow (i.e., > ~0.5 s) (2) early (~0.5–2.5 s) absorption changes from alterations in chromophore redox status, i.e., the oxy/deoxy-hemoglobin ratio (known as the “initial dip” period), and (3), slower (~2–10 s) absorption changes due to blood volume increase (correlated with the fMRI BOLD signal). Light scattering changes have been attributed to interstitial volume changes resulting from cellular swelling, organelle swelling due to ion and water movement, capillary expansion, and neurotransmitter release [6,7]. The slower absorption factors have been demonstrated to correlate with the changes in metabolic demand and subsequent hemodynamic cascades following neuronal activation [4,8,9].
Using animal models of acute and chronic brain injury, ISOI has been used to quantify the acute hemodynamic events in response to stroke, including focal ischemia and cortical spreading depression (CSD) [10–21]. Researchers have also used ISOI to locate and quantify the spatial extent of the stroke injury, including ischemic core, penumbra, and healthy tissue zones [18,22]. CSD also plays a key role in migraine headache, and recent laser speckle imaging studies have revealed the neurovascular coupling mechanism to the transmission of headache pain [23,24].
To fully understand the underlying mechanisms in vascular changes associated with cerebrovascular diseases such as stroke, an optical imaging technique that has the capability to rapidly separate absorption from scattering effects can enhance the information content of traditional ISOI, enabling (1) more accurate quantitation of hemodynamic function, (2) isolation of the electro-chemical changes characterized by light scattering, and (3) longitudinal chronic injury studies of function where structural reorganization due to neovascularization can cause significant alterations in scattering [25,26].
Quantitative diffuse optical methods [27] such as spatially-resolved reflectance, diffuse optical spectroscopy (DOS), and tomography (DOT), and diffuse correlation spectroscopy (DCS) possess exquisite sensitivity to these functional and structural alterations associated with brain injury, and have been applied to the study of CSD [11,15,28]. DOS and DOT utilize the near-infrared spectral region (600–1000 nm) to separate and quantify the multispectral absorption (μa) and reduced scattering coefficients (μs′), providing quantitative determination of several important biological chromophores such as deoxy-hemoglobin (HbR), oxy-hemoglobin (HbO2), water (H2O), and lipids. Concentrations of these chromophores represent the direct metrics of tissue function such as blood volume fraction, tissue oxygenation, and edema. Additionally, the scattering coefficient contains important structural information about the size and density of scatterers and can be used to assess tissue composition (exctracellular matrix proteins, cell nuclei, mitochondria) as well as follow the process of tissue remodeling (wound healing, cancer progression). DOS utilizes a limited number of source-detector positions, e.g., 1–2, but often employs broadband content in temporal and spectral domains [29]. In contrast, DOT typically utilizes a limited number of optical wavelengths (e.g., 2–6) and a narrow temporal bandwidth, but forms higher resolution images of subsurface structures by sampling a large number of source-detector “views.” To achieve maximal spatial resolution, the ideal DOT design would employ thousands of source-detector pairs and wavelengths. However, several engineering considerations including measurement time and instrument complexity currently limit the practicality of this approach.
In this chapter we present the basic principles of a new, noncontact quantitative optical imaging technology, modulated imaging (MI) [30–32], and provide examples of MI performance in 2 rat models of brain injury, cortical spreading depression (CSD) and stroke. MI enables both DOS and DOT concepts with high spatial (<1 mm) and temporal resolution (<1 s) in a simple, scan-free platform. MI is capable of both separating and spatially-resolving optical absorption and scattering parameters, allowing wide-field quantitative mapping of tissue optical properties. While compatible with time-modulation methods, MI alternatively uses spatially modulated illumination for imaging of tissue constituents. Periodic illumination patterns of various spatial frequencies are projected over a large area of a sample. The diffusely reflected image is modified from the illumination pattern due to the turbidity of the sample. Typically, sine-wave illumination patterns are used. The demodulation of these spatially modulated waves characterizes the modulation transfer function (MTF) of the material, and embodies the sample optical property information.
12.2. METHODS AND INSTRUMENTATION
12.2.1. Modulated Imaging Spectroscopy
The MI instrument platform was introduced originally by Cuccia et al. [30] Based on this design, we have developed a custom multispectral near-infrared (NIR) MI spectroscopy system capable of imaging between 650 and 1000 nm. A diagram of this system is shown in Figure 12.1.
FIGURE 12.1
Modulated imaging platform. QTH—quartz tungsten halogen; L1—aspheric condenser; H—hybrid hot mirror; DMD—digital micromirror device; L2—projection lens; L3—camera lens; LCTF—liquid crystal tunable (more...)
Broadband NIR illumination is provided by an intensity-stabilized 250 W quartz-tungsten-halogen (QTH) lamp (Oriel QTH Source with Light Intensity Controller, Newport Corporation-Oriel Instruments, Stratford, Connecticut). Light is collimated and refocused with a pair of aspheric F/#0.7 optical lens systems (Oriel Aspherab). A custom-sized 3.5 in square hybrid hot mirror (Reynard Corporation, i.e., R00670-00) was placed between the lenses to limit the illumination to wavelengths below 1000 nm. Light engine optics taken from a digital projector (NEC HT1000) serve to homogenize and direct the light onto a 0.7 in digital micromirror device (DMD Discovery™ 1100 with ALP Accessory Package, ViALUX, Germany). Grayscale spatial sinusoid patterns are projected at 400 Hz using the ViALUX software development toolkit, which generates the necessary pulse-width modulation of binary sub-frames to produce a specified grayscale bit-depth (1–8 bits). Finally, a fixed focal length (f = 100 mm) projection lens illuminates the tissue at a slight angle from normal with a 15 × 25 mm illumination field. Detection was performed at normal incidence using a CRI Nuance™ camera system, which combines a 12-bit CCD camera and a liquid crystal tunable filter (LCTF; λ = 650–1100 nm, Δλ = 10 nm). To avoid specular reflection, crossed linear polarizers are used in the illumination and detection arms. For this system, the former is a 1.5 in diameter NIR linear polarizer (Meadowlark Optics, VLM-200-IR-R) placed immediately after the projection lens, and the first stage of the Nuance LCTF serves as the latter. The DMD, CCD, and LCTF are controlled via USB by a laptop computer, and synchronized using LabVIEW software (LabVIEW 8, National Instruments), enabling fast acquisition of a series of patterns with various spatial frequencies.
12.2.2. SFD Measurement, Calibration, and Modeling
A detailed description of SFD measurement, calibration, and diffusion modeling is provided by Cuccia [32]. In this work, we modeled diffuse reflectance using a transport-based White Monte Carlo (WMC) method [33,34]. Previously, we have found that compared with Monte Carlo, (1) diffusion predictions over- and underestimate low- and high-frequency diffuse reflectance, respectively, and (2) the quantitative accuracy of diffusion degrades with decreasing albedo [32]. Due to the moderate albedo of brain tissue (μs′/μa ~ 10–20), we chose to analyze all brain data with the WMC approach. This homogeneous tissue model is a significant simplification of the multilayered rat brain, and more work is necessary to accurately model this complex system. We discuss further the consequences of our simple model in Section 12.2.5.
12.2.3. Optical Property Inversion Methods
In this chapter, we use two inversion methods to calculate the absorption and reduced scattering from measurements of diffuse reflectance. When high measurement precision is desired, we use a “sweep” in spatial frequency space, producing an overdetermined set of diffuse reflectance measurements, which can be fitted to our WMC forward model predictions using least-squares minimization. This method is performed for all spatially averaged region analysis of optical properties and chromophores. When increased acquisition and/or processing speed is desired, we alternatively use a rapid two-frequency lookup table method based on cubic spline interpolation [32]. This data can be achieved with a minimal 3-phase, single frequency image set (by demodulating and averaging the images to obtain AC and DC amplitude maps, respectively). On typical personal computers this approach is capable of millions of inverse lookup calculations per second, and is therefore used to calculate all high-resolution images including time sequences. The signal-to-noise ratio (and thus the measurement precision) of either approach is limited by the data sampling, with the two-frequency method having a lower precision with the tradeoff of higher acquisition and processing speed.
12.2.4. Spectral Analysis-Chromophore Calculation
The quantitative absorption coefficient is assumed to be a linear (Beer’s law) summation of individual chromophore absorption contributions:
μa(λ)=2.303∑i=13ci?i(λ),
12.1
where ci and ?i(λ) represent chromophore concentrations and molar extinction coefficients, respectively. Using reported extinction coefficients of HbO2/HbR35 and H2O,36 we can invert Equation 12.1 and calculate tissue chromophore concentration separately at each pixel by linear least-squares fitting to the multispectral absorption images. Total hemoglobin (HbT) and oxygen saturation (StO2) can then be calculated as HbT = HbR + HbO2 and StO2= HbO 2/(HbR + HbO2) * 100, respectively.
12.2.5. Optical Property Mapping: Resolution Versus Quantitation
On a pixel-by-pixel basis, diffuse reflectance versus spatial frequency is fitted to the WMC forward model to extract the local absorption and reduced scattering optical property contrast. This process is repeated for each wavelength, resulting in multi-spectral absorption and scattering spectra at each pixel. The measured contrast from discrete absorbers and scatterers on millimeter and submillimeter spatial scales, however, will possess partial volume effects in all three spatial dimensions. This is due to the physical light transport length scales in tissue, limiting the true x-y resolution of optical property contrast to many detector pixels [32]. This phenomenon is not unique to MI, but present in all planar reflectance imaging measurements of turbid media. Absorption and scattering are calculated using a homogeneous reflectance model, extracting a locally averaged sampling of optical property contrast. Based on simulations of the tissue MTF for varying optical properties [32], we expect the resulting image resolution to scale directly with the transport length, l* = (μa + μs′)− 1, and the spatial frequency of illumination. In this chapter, we place quantitative emphasis on average optical properties and chromophores measured over a field of view that is greater than l*. Spatial maps and videos of these parameters are displayed and referred to as “contrast maps,” with the caveat that high resolution features will exhibit degraded quantitative accuracy.
12.2.6. In Vivo Rat CSD Experiments
12.2.6.1. Animal Preparation
MI spectroscopy measurements were performed on an in vivo Wistar rat model with a thinned-skull preparation. All procedures were performed in accordance with approved IACUC protocol guidelines. The animals were anesthetized, placed in a stereotaxic frame, their skulls thinned and glass coverslip applied. This preparation is described in detail by Masino et al. [37] The resulting thinned skulls allowed direct imaging of the cortex over a 5 × 7 mm field-of-view (whisker barrel cortex, centered at the C2 location). In order to investigate the sensitivity of MI toward studying acute cortical injury, we induced cortical spreading depression (CSD) by applying 1 M KCl solution to the surface of the cortex through a perforated section of skull and dura, located approximately 3 mm above the camera’s imaging field.
12.2.6.2. MI Measurement Protocol
For each of three animals, our MI measurement protocol was twofold. Prior to CSD induction, baseline spatial modulation data were acquired at 6 spatial frequencies (3-phase projections each) from 0 to 0.26 mm−1, at 10 nm intervals over the entire range between 650 and 980 nm. Depending on the wavelength, image acquisition times ranged from 200 ms to 4 s, with total spectral imaging time of approximately 30 s per spatial pattern. The entire measurement (34 wavelengths, 3 phases, 6 frequencies) was repeated three times for statistical averaging yielding an entire measurement time of approximately 30 min.
Next, rapid dynamic measurements were performed, beginning 1 min prior to K+Cl− administration. Here, a significantly reduced data set was chosen in order to achieve high temporal resolution. Two spatial frequencies (0 and 0.26 mm−1) were acquired with three phase projection images, as described in Section 12.2.2, at each of four wavelengths (680, 730, 780, and 830 nm). The resulting 12 images took in total 6 s, permitting a repetition rate of 10 measurements per minute. The animals were followed for a period of 10 min for rats 1 and 2, and a period of 30 min for rat 3.
All images in this study were smoothed by 2D convolution with a Gaussian filter function (FWHM = 3 pixels), and baseline repetitions were averaged prior to data processing. Additionally, time-series data were post-processed by smoothing slightly in time (Gaussian FWHM of 2 timepoints = 12 s).
12.2.6.3. Spatial Frequency Sensitivity Analysis
Because of the differential absorption sensitivity at low and high frequencies, optimal optical property separation is achieved when a large range of frequencies is used [31]. In Figure 12.2a, we depict this differential sensitivity using diffuse reflectance (MTF) predictions versus frequency, increasing μa by 100% from 0.02 (black line) to 0.04mm−1 (gray line). This is done for two values of μs′, 0.6 (solid lines) to 1.2mm−1 (dashed lines), simulating a 100% change in scattering. Notice that the low frequencies have a significant reflectance change due to absorption, while high frequency reflectance remains nearly unchanged. Conversely, reflectance changes due to scattering are observed at all spatial frequencies. In Figure 12.2b, we further visualize this by plotting the reflectance sensitivity to 1% changes in absorption and scattering. Whereas DC reflectance is equivalently sensitive to a fractional change in either absorption or scattering, at high spatial frequencies absorption contrast is lost while scattering contrast is retained. For instance, notice that at our maximum measurement frequency of 0.26 mm−1 the reflectance is roughly 24 times more sensitive to scattering compared to absorption (ΔRd = 0.56 μs′ versus 0.024 * 10−3 for μa). This plays an important role in Section 12.3.2 during our discussion of dynamic scattering measurement.
FIGURE 12.2
(a) Reflectance contrast in absorption and scattering covering a typical range of brain optical properties. (b) The frequency-dependent sensitivity to absorption (black line) and scattering (gray line), respectively. Reflectance at fx = 0.26 mm−1 (more...)
In realistic heterogeneous tissues, a tradeoff exists between maximizing the frequency range for optical property accuracy and obtaining similar sampling volumes. As tissue is a low-pass spatial filter, high frequencies are attenuated quickly with depth. Using diffusion-based forward modeling, we have estimated mean sampling depths at 650 nm using measured average background optical properties of brain tissue. This was done by predicting the depth sensitivity to contrast from a planar perturbation in absorption, given a background fluence profile from spatial frequencies 0 and 0.26 mm−1. Based on these results, we observe qualitatively similar depth sampling, with mean depth sampling ranging between 2.5 mm and 1.2 mm (for fx = 0 and 0.26 mm−1, respectively). In all cases maximal sensitivity was found in the first 1–2 mm, where cortical hemodynamic changes occur.
12.3. RESULTS AND DISCUSSION
12.3.1. Baseline MI Spectroscopy
In Figure 12.3a we show a grayscale planar reflectance image of the cortical region of rat 1 at 650 nm. A dotted-line box denotes the region-of-interest (ROI) used for analysis, selected for its uniform illumination and the absence of cerebral bruising. The Monte Carlo-model fitting of spatial frequency data allows calculation of the absorption and reduced scattering coefficients. In Figure 12.3b we show the spatially averaged diffuse reflectance at 650 nm and the corresponding multi-frequency fit. Excellent agreement is observed between measurement data and the model-based fit, with derived μa and μs′ coefficients of 0.033 and 0.70 mm−1, respectively.
FIGURE 12.3
(a) Reflectance map for rat 1, showing the 3.8 × 5.9 mm region chosen for quantitative analysis. (b) Sample MTF reflectance data (squares) and fit (solid line) at 650 nm. (c) Recovered optical property maps (above) and corresponding image histogram (more...)
Analysis of multifrequency reflectance data separately at each pixel results in spatial maps of absorption and reduced scattering contrast. In Figure 12.3c, we plot the μa and μs′ maps recovered at 650 nm for rat 1. Note the strong absorption in the vein region, due to a large absorption by HbR at this wavelength. Below the images, we show histogram distributions of the corresponding quantitative maps above, indicating the degree of spatial variation in recovered optical properties. The mean and standard deviation for the pixel-wise μa and μs ′ were 0.030 ± 0.007 mm−1 and 0.63 ± 0.13 mm−1, respectively. These statistical results are in good agreement with the spatially averaged reflectance fit from Figure 12.3b, suggesting that our simple pixel-wise fitting approach yields optical properties similar to that calculated using a global analysis.
By mapping the absorption coefficient at multiple wavelengths, we can perform quantitative spectral imaging of tissue. In Figure 12.4, we summarize the baseline spectroscopy results for all three animals. In Figure 12.4a we show the μa (left) and μs′ (right) coefficients versus wavelength (circles) recovered from spatially averaged fitting. Data for rat 1 is shown in black (rat 2 in dark gray; rat 3 in light gray). Note the distinct spectral features in absorption, resulting from oxy- and deoxy-hemoglobin (HbO2, HbR), and water (H2O) absorption. The calculated scattering coefficient generally decays with increasing wavelength, and the results from a power law (μs ′ = A·λ(nm) −b, solid lines) fit are shown. A small residual coupling is observed between measured scattering and absorption spectral features. In particular, the scattering at the shortest and longest wavelengths appears to be underestimated by 5–10%, occurring where the corresponding absorption is highest (due to HbR and H2O, absorption features, respectively). Based on our experiments in layered tissue phantoms [38], we believe this effect is primarily due to frequency-dependent probing volumes in the presence of depth-heterogeneous structures.

FIGURE 12.4
(a) Average μa (left) and μs′(right) spectra over entire ROI (circles). HbO 2, HbR, and H2O concentrations are determined by subsequent least-squares fitting (solid lines) of molar extinction coefficients to the absorption. Data (more...)
Simultaneous linear fitting of the absorption to known extinction coefficients yields measures of chromophore concentration. Shown in Figure 12.4a, multispectral fitting (solid line) for rat 1 yields HbO2, HbR, H2O, HbT and StO2 values of 56.3 μM, 33.2 μM, 63.9%, 89.6 μM, and 56.3%, respectively. Tabulated results of chromophore values for all three animals are shown in Figure 12.4b. Lipid absorption near 930 nm was not apparent in the μa spectrum, and when included in the spectral analysis was not found to significantly affect the results. The small absorption “bump” at 900–910 nm is an artifact of imperfect phantom calibration due to the presence of a sharp, strong silicone absorption peak that is present in the phantom.
We note that the solution for chromophore concentration is well-determined when the number of wavelengths is at least equal to the number of chromophores. Therefore, as few as two wavelengths can be used to separate HbO2 and HbR (if a constant value of H2O is assumed). Repeating the above analysis with 780 and 830 nm only (assuming H2O = 65%) yields results for HbO2 and HbR within 10% of those from full spectral fitting. Repeating the above analyses using a simple diffusion-based model provided qualitatively similar results for absorption and scattering spectra, but in general was found to overestimate the absorption coefficient by 10–25%.
Absorption spectra at each pixel can be separately analyzed to yield spatial maps of local HbO2, HbR, and H2O distribution, shown in Figure 12.5. Notice the high concentration of HbR over the large superficial draining vessel (venous) regions, also reflected in the StO2 image, highlighting the effect of tissue oxygen extraction. Conversely, notice that the high albedo regions with less structural detail are highly oxygenated, with StO2 levels between 60 and 70%. Lastly, the H2O map reveals a relatively homogeneous distribution of water.
FIGURE 12.5
Chromophore fits to absorption spectra at each pixel yield maps of local HbO2, HbR, and H2O concentration (left). Total hemoglobin (HbT) and oxygen saturation (StO2) maps can then be calculated from HbO2 and HbR.
12.3.2. Dynamic MI Spectroscopy of CSD
We performed measurements of CSD in each of the three rats, as described in Section 12.2.3. The results are presented as follows. We first present data for a single animal, choosing rat 3 for its long observation period of 30 minutes. Three ROIs are selected for analysis, and baseline MI spectroscopy results are reported for each of these regions. Next, the observed dynamic time courses of diffuse reflectance, optical properties, and chromophore concentrations are shown for each ROI. We then present the full spatio-temporal dynamic contrast data for rat 3 (2D + time) in the form of “snapshot” images.
Figure 12.6 summarizes the baseline spectroscopy measurements for rat 3. In Figure 12.6a, we show three regions of interest superimposed on the DC reflectance map, chosen to highlight three different characteristic temporal profiles observed within the field of view. In Figure 12.6b we show the baseline spectral fits for each of these regions, and in Figure 12.6c we tabulate the resulting calculated chromophore concentrations. In general, Region A (black) is a high albedo region lacking any large blood vessels, whereas Regions B (dark gray) and C (light gray) include high-absorption blood vessels and mild cerebral bruising from surgery. These differences are apparent in their recovered absorption spectra and fits, with on average 27% higher HbT, and 32% lower saturation in the vascular regions. Also, 7% higher H2O is found in Regions B and C, which may indicate increased edema due to bruising.
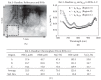
FIGURE 12.6
Regionwise spectral analysis of rat 1 baseline data including the respective (A) ROIs, (B) spectral absorption data (circles) and fit (lines), and (C) tabulated recovered chromophore data for each region
In Figures 12.7–12.9 (for regions A–C, respectively), we present the temporal dynamics of CSD in each ROI of rat 3 as measured by MI. In part (a) of each figure, we plot the multispectral diffuse reflectance changes at fx = 0 mm−1 (DC, top) and fx = 0.26 mm−1 (AC, bottom). In part (b), we plot the recovered Δμa (top) and Δμs′ (bottom) optical properties at each wavelength. While absolute values of diffuse reflectance and optical properties are measured separately at each time point, for visualization purposes all data are displayed as a change from that prior to KCl administration. Absolute optical property values at t = 0 (not shown) demonstrate excellent agreement (~5–10%) with full multifrequency baseline data.
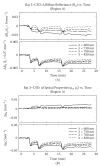
FIGURE 12.7
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx =.26 mm−1, bottom) for Region A of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) coefficients. (more...)
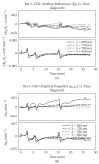
FIGURE 12.8
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx = 0.26 mm−1, bottom) for Region B of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) (more...)
FIGURE 12.9
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx = 0.26 mm−1, bottom) for Region C of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) (more...)
Looking first at the reflectance time courses of Figure 12.7a (Region A), we see in general a series of three CSD events over the 30 minutes, with each transient event occurring for approximately 4.3 minutes. The first event occurs at minute 2.9 after KCl application, indicating an initial latency between the insult and the first resulting spreading depression wave. Reflectance contrast is present in both DC and AC frequency components, but with markedly different signatures. Generally, the DC time course shows a slow, gradual decay, punctuated by sharp, wavelength-dependent spikes/dips (for short/long wavelengths, respectively). Alternatively, the AC signature contains three sets of transient dips consistent across all wavelengths, with final values leveling off progressively lower than baseline. Discussed in detail in the following paragraph, we believe these AC changes are due primarily a result of optical scattering and may be related to neuronal depolarization. The corresponding derived optical properties in Figure 12.7b reflects this, with μs′ trends tracking directly with the measured AC reflectance. As expected, μa trends reveal similar wavelength-dependence of the DC reflectance (with opposite polarity), reflecting changes in HbO2 and HbR.
In Section 12.2.3.3 we noted that the diffuse reflectance at fx = 0.26 mm−1 is 23 times more sensitive to scattering changes compared to absorption. In this context, we propose that the observed magnitude of the CSD-induced AC reflectance changes can only be explained by changes in optical scattering. To concretely illustrate this point, we pick as an example the observed 780 nm AC diffuse reflectance dip in Figure 12.7a at t = 3.7 min of -0.003. Here, the corresponding change in reduced scattering in Figure 12.7b, Δμs′, is calculated to be −0.03 mm−1. In order for this change to instead be due to an absorption-only event, μa would need to increase by 121% from baseline (from 0.038 to 0.084 mm−1). This increase would also need to be accompanied by a drop in Rd (fx = 0 mm−1) of 0.12 (33%), whereas the actual observed DC reflectance only drops by 0.008 (<1%) and thus cannot explain the change. Secondly, we note that the three sets of AC reflectance dips occur consistently across all four wavelengths. While an approximate 120% increase in HbT could induce this decrease at high frequency, it would also require a large broad-wavelength decrease in the DC reflectance. We instead observe during these events that the DC increases at short wavelengths while the DC decreases at long wavelengths, suggesting primarily an exchange between HbO2 and HbR volume fractions, as opposed to a dramatic HbT change.
Regions A–C (Figures 12.7–12.9) were chosen to highlight three different time signatures observed in the field of view during the CSD dynamics. The most contrasting feature between all three regions is the measured AC reflectance and the derived scattering coefficient. In Region B (Figure 12.8), each CSD event appears to cause a biphasic scattering change, with a sharp increase and then decrease, whereas a monophasic dip was observed in Region A (Figure 12.7). Region C (Figure 12.9) appears even more complex with a triphasic rise-dip-rise temporal profile. We observe that Regions A to C are located with increasing proximity to the CSD induction point (3 mm above the imaging field).
Because fractional changes in scattering and absorption have an equal (and opposite) effect on DC reflectance (see Section 12.2.3.3), any scattering (i.e., pathlength) changes measured here could be misinterpreted as absorption events with traditional ISOI analyses (i.e., DC reflectance only). In our observations, the measured scattering change of up to −0.05 mm−1 would be interpreted as an increase in absorption of up to +0.005 mm−1, more than the maximum measured absorption change for wavelengths 730, 780, or 830 nm in any of the three regions. In order to account for differential pathlength changes, Kohl et al. proposed a multispectral model [39], which they used to differentiate dynamic scattering and absorption changes using ISOI. This approach improves ISOI accuracy, and has been generally adopted as the method of choice for quantitative functional imaging. For dynamic measurements, we see MI as an improvement over this approach as it alternatively uses frequency domain measurements at a single wavelength to derive absolute scattering and absorption coefficients. This potentially provides a simplified single-wavelength measurement apparatus for detection of scattering, and also avoids potential mis-estimation of background optical properties.
Light scattering changes induced by spreading depression have been reported previously, and a comprehensive review is provided by Somjen. With in vivo spatially resolved reflectance measurements, Kohl et al. [11] separated absorption from scattering and observed a biphasic scattering response similar to that of Region A. With simultaneous laser scattering and electrophysiological measurements, both Jarvis et al. and Tao et al. found a strong correlation between electrical and optical scattering changes [12,13,40]. Tao et al. noted spatial heterogeneity in the dynamic spreading depression (SD) waveform related to the proximity to the SD induction site, similar to our results.
Using linear spectral analysis of absorption at all four wavelengths, we calculated the time-dependent chromophore concentration for Regions A, B, and C, presented in Figure 12.10A,B,C, respectively. In each region, the calculated baseline concentrations of H2O were assumed to be constant. All three regions exhibit remarkably similar trends in HbR, HbO2, HbT, and StO2. This similarity is not clear in the DC traces of Figures 12.7–12.9, further highlighting the benefit of accurate separation of μa and μs′. Focusing on the first CSD event, there is a very consistent signature of: (1) a 2-minute latency post-KCl administration, (2) a 30-second period of decreasing StO2 (3) a dramatic spike in both StO2 (3–10%) and HbT (2–4 μM) with rise and decay times of approximately 1 minute each. For each region, the final StO2 is approximately 5–10% lower than baseline, while the HbT restores to baseline values. This process repeats again twice more, except that the phase (2) desaturation appears to be absent. Additionally, in the “vessel” Region 3, we observe a gradual increase in HbT over the 30 minutes, indicating chronic blood pooling.
FIGURE 12.10
Recovered HbR, HbO2, HbT, and STO2, for ROIs A, B, and C (top, middle, and bottom), recovered by analysis of the multispectral absorption coefficients from Figures 12.7–12.9b (top).
We show in Figure 12.11 the spatio-temporal evolution of both chromophore concentration and scattering changes from the first SD wave in rat 3. These are depicted in the form of a time derivative, i.e., (C(tn + 1) − C(tn))/(tn + 1 − tn), where C represents concentration/saturation/scattering values and tn represents time of acquisition for data point n. This visualization is appealing as it highlights the changes with high contrast [18]. From left to right, we show HbO2, HbR, HbT, StO2, and μs′. Notice the wave in scattering which propagates from top right to bottom left, at a rate of approximately 3 mm/min. An increase, or “spike” in scattering is observed initially in the top right hand corner, in close proximity to the location of KCl administration. Note the large spikes in HbT and StO2 due to vascular activity from depression wave propagation through the measurement field. We observe a transient increase in saturation and blood volume. Over the longer time periods, however, we observe a slow, sustained trend toward hypoxia in the vein regions.

FIGURE 12.11
Spatio-temporal evolution of the hemodynamic and neural scattering response during a single spontaneous CSD event in rat 3. For visualization, a time derivative of the image sequence is displayed to highlight changes.
The spatio-temporal evolution of the scattering coefficient in Figure 12.11 reveals a spatially defined scattering wave (reduction in μs′) that precedes hemodynamic changes. The scattering drop is presumed to be a consequence of neuronal depolarization accompanying CSD. This observed wave pattern has been shown previously with reflectance ISOI and attributed to blood volume changes [18]. Interestingly, the scattering depolarization wave is clearly followed in space and time by the increase in deoxyhemoglobin (HbR), decrease in saturation (StO2), and drop in oxyhemoglobin (HbO2); changes that are consistent with depolarization-induced neural tissue oxygen consumption.
12.3.3. Dynamic MI Spectroscopy of Stroke
In order to assess the sensitivity of MI to stroke, we conducted preliminary studies in a rat middle cerebral artery occlusion (MCAo) model, the most commonly involved artery in ischemic strokes. The left MCA was surgically cauterized using monopolar cautery or ligated to produce a permanent stroke. Figure 12.12 shows pre-versus post-MCAo results for a representative animal. Data were acquired at 5 wavelengt
Original Source: https://www.ncbi.nlm.nih.gov/books/NBK20233/
An Hour of Light and Sound a Day Might Keep Alzheimer’s at Bay
Angus Chen - Scientific American March 14, 2019 (Publication) 4521This is an summary article about the work of Shannon Macauley, a neuroscientist at Wake Forest School of Medicine who found that light and sound has a siginicant impact on the Alzheimer's
There is no cure for Alzheimer’s disease. Although a few drugs manage temporarily certain cognitive symptoms of the illness, none can stop or meaningfully slow its progression. “We really don’t have much to offer people,” says Shannon Macauley, a neuroscientist at Wake Forest School of Medicine. Virtually all new treatments have failed in clinical trials. But new research is looking beyond drugs to see what relief might come from a simple LED light and a speaker. Bathing patients in flashing light and pulsing sounds both tuned to a frequency of 40 hertz might reverse key signs of Alzheimer’s in the brain, according to a paper published in Cell on Thursday. “I think it’s an absolutely fascinating paper to be honest,” says Macauley, who was not involved in this work. “It’s a very provocative idea. It’s noninvasive and easy and low cost, potentially, so if it were to come to fruition in humans—that’s fabulous.” Still, all this is a big if, Macauley acknowledges. The work was done in mice with genetic alterations that doomed them to develop key symptoms and pathology of Alzheimer’s disease. One batch of mice formed neurofibrillary tangles inside their neurons—dysfunctional knots of a protein called tau that can lead to the cell’s death. Another batch of the mice developed amyloid beta plaques—sticky heaps of protein that dam the flow of communication between neurons. All the mice also had a third hallmark of the disease—irregular brain activity in the gamma range of brain waves that oscillate between 30 and 100 times a second. In 2015 neuroscientist Li-Huei Tsai, director at The Picower Institute for Learning and Memory at Massachusetts Institute of Technology, was working on an experiment to manipulate that brain activity by flashing a white light at these mice. Like light strobes, our brains flicker. Brain waves are generated when large groups of neurons oscillate on and off together. Neurons encode our thoughts and actions and senses in this rhythmic electrical flutter. So when Tsai tuned her light to flash 40 times a second, or 40 hertz, and flickered it at the mice, their brains flickered back—generating gamma waves at a corresponding 40 hertz. Then, something unexpected happened. When Tsai dissected the mice brains afterward, the amount of amyloid plaques and tau tangles in the mice that saw the light had plummeted. “It was the most remarkable thing,” Tsai says. “The light flicker stimulation triggers a tremendous microglia response. These are the brain’s immune cells that clear cell debris and toxic waste including amyloid. They’re impaired in Alzheimer’s disease, but [the light] seems to restore their abilities.” When Tsai dissected the mice brains afterward, the amount of amyloid plaques and tau tangles in the mice that saw the light had plummeted. “It was the most remarkable thing,” Tsai says. “The light flicker stimulation triggers a tremendous microglia response. These are the brain’s immune cells that clear cell debris and toxic waste including amyloid. They’re impaired in Alzheimer’s disease, but [the light] seems to restore their abilities.” This clearing-out process only happened in the visual cortex where the brain processes light information. To get these effects to penetrate deeper into the brain, she added a clicking sound like a dolphin’s chirrup that also had a 40-hertz frequency. When the mice sat in a room with both the flashing light and the droning sound for an hour day, seven days in a row, amyloid plaques and tau tangles began falling in not just the audio and visual cortices but the prefrontal cortex and the hippocampus as well. “This was one of the big jumps in the new paper,” Macauley says. “These are the learning and memory centers of the brain. And there was about a 40 or 50 percent decrease in amyloid and tau levels. It’s an absolutely impressive feat.” That showed when Tsai put the mice through a set of cognitive tests. In one, where the mice were given a familiar and an unfamiliar object to explore, mice that didn’t get the treatment acted as though they’d never seen the familiar object. “That shows some memory problems,” Tsai says. Mice that saw the light and heard the sound spent about two thirds of the time that untreated mice did examining the familiar object. “It was unbelievable,” Tsai says. “This is the first time we’ve seen that this noninvasive stimulation can improve cognitive function. It’s not a drug or an antibody or anything, it’s just light and sound.” One possible explanation for this is brains with Alzheimer’s have irregular, often hyperactive, neurons, says Jorge Palop, a neurologist at the University of California, San Francisco, who did not work on the study. By providing the brains with a steady and regular beat, the repeating light and sound might work as a kind of metronome for brain activity. “This could be like resetting the mice every day and correcting some of this abnormal activity that they have,” he says. “Then downstream of that are all these beneficial effects.” All of this is still at the level of speculation. Researchers simply do not know why these brain waves, specifically ones rising from light and sound stimulation at 40 hertz and no other frequencies, can lead to a reversal of Alzheimer’s disease symptoms. “That’s a mystery,” says Terrence Town, a neuroscientist, at the University of Southern California who was not involved with the work. It’s also not clear if these beneficial effects would appear or if 40 hertz is the “magic” frequency in humans, he says. Tsai is already working on answering those questions. In human studies underway at Cognito Therapeutics, a start-up she founded with her colleague Ed Boyden, she says light and sound seem to increase gamma waves in healthy participants without negative side effects. “Nobody gets sick or even complains about it,” Tsai says. “But to see a [therapeutic] effect in humans, you’ll have to wait a long time. If this approach has an impact, the experiment could easily take five years to have some conclusive answer.”
Original Source: https://www.scientificamerican.com/article/an-hour-of-light-and-sound-a-day-might-keep-alzheimers-at-bay/
Review of transcranial photobiomodulation for major depressive disorder: targeting brain metabolism, inflammation, oxidative stress, and neurogenesis
Paolo Cassano; Samuel R. Petrie; Michael R. Hamblin; Theodore A. Henderson; Dan V. Iosifescu; - Neurophotonics, 3(3), 031404 (2016). doi:10.1117/1.NPh.3.3.031404 March 4, 2016 (Publication) 4471This study shows some of the most detailed parameters (power, wavelenght, dosage) for working with the brain and seems to be unbiased because of the diverse background of authors..
1.
Introduction
Infrared (IR) light is ubiquitously present to most life on the earth. Of the total amount of solar energy reaching the human skin, 54% is IR and 30% is IR type A—near-infrared—(NIR; with a wavelength range of 760 to 1440 nm),1 which penetrates through the human skin and reaches deeply into tissue, depending on wavelength and energy.2
NIR is used to treat a variety of conditions such as muscle pain,3 wounds,4 neuropathic pain,5 and headache.6 NIR is also used for wellness and lifestyle purposes such as for cosmetic improvement in peri-orbital wrinkles.7,8 The clinical use of NIR light applied in NIR-spectroscopy dates from the mid-1980s, when it was used for monitoring of the brain in the neonate and the fetus.9
The use of transcranial phototherapy for treating brain disorders started with its application to acute stroke. Numerous preclinical animal studies1011.–12 suggested that the application of NIR laser (810 nm) to the head at various times (hours) after induction of an acute stroke had beneficial effects on subsequent neurological performance and reduced lesion size. Evidence was obtained for the anti-inflammatory, anti-apoptotic, and proneurogenesis effects in the brain stimulated by this approach.13,14 These promising animal studies led to the conduction of a series of clinical trials called NeuroThera Effectiveness and Safety Trials (NEST). All together there were three large studies conducted in 1410 stroke patients [NEST-1 (
), NEST-2 (
)] that demonstrated that NIR light delivered transcranially with a class-IV laser is safe, with no significant differences in rates of adverse events with NIR, when compared to sham exposure.1516.–17 Other preclinical studies and clinical trials have suggested that transcranial photobiomodulation (PBM: laser or light emitting diodes—LED) is safe and effective for acute1819.20.21.–22 and chronic2324.–25 traumatic brain injury (TBI) and has beneficial effects on neurodegenerative diseases (Alzheimer’s and Parkinson’s).26,27
For the transcranial treatment of major depressive disorder (MDD), both PBM LEDs and lasers have been experimentally tested, although PBM is not FDA-approved for the treatment of MDD. Certain forms of PBM treatment are also referred to as low-level light therapy (LLLT), since it utilizes light at a low power (0.1 to 0.5 W output at the source) to avoid any heating of tissue. The irradiance of the PBM medical devices (or power density) typically ranges from 1 to 10 times the NIR irradiance from sunlight on the skin (
at the zenith). However, most PBM medical devices only deliver light energy at one or two selected wavelengths, as opposed to the whole spectrum of IR that is contained in sunlight. To our knowledge and to this date, transcranial PBM treatment has not caused any retinal injury—one of the most likely postulated adverse events, although care is taken routinely in such studies to protect the eyes with goggles or eye covers.28
In this review, we will first discuss the mechanisms of action by which NIR and red light (PBM) might improve symptoms of depression, and then present the clinical evidence for their use as a treatment for MDD and other comorbid psychiatric syndromes.
2.
Methods
We found clinical and preclinical studies via PubMed search (December 15, 2015), using the following keywords: “near-infrared radiation,” “NIR,” “low-level light therapy,” “low-level laser therapy,” or “LLLT” plus “depression.” We chose studies that had a clinical focus, and we excluded studies involving NIR spectroscopy. We also located studies using the references from the articles found in the PubMed search. As the searched literature encompassed different conditions and disorders frequently comorbid with depression, a specific section of this review was devoted to the effect of PBM on psychiatric comorbidity. In the latter section, the following conditions were included, based on available literature: TBI, anxiety and post-traumatic stress syndromes, insomnia, and suicidal ideation. The literature search for the use of PBM to treat comorbid conditions was neither systematic nor extensive, but rather a secondary focus of this review. The information is presented in an organized fashion to allow the reader to easily grasp the potential applications of PBM for the treatment of depression and of its comorbid conditions. To attain this goal, the authors have allowed a margin of redundancy, by distributing different information derived from any given publication in separate sections of this review. To avoid an artificial inflation of the extant literature on the chosen topic, we referenced the main authors—and when appropriate their affiliation—when referring to the same articles more than once. The reader will find a table summarizing the six key clinical articles reviewed, also to avoid unintended inflation of the literature. The six clinical reports included in this review where extracted from a pool of 58 articles, that were originally identified with the literature search.
In addition, we used PubMed to find articles that examined the link between PBM and each of the various biological processes including metabolism, inflammation, oxidative stress, and neurogenesis.
3.
Targeting Brain Metabolism
Multiple studies have reported regional and global hypometabolism in MDD, which could be related (either causally or consequentially) to the neurobiology of mood disorders.2930.31.–32 Positron emission tomography studies have shown abnormalities in glucose consumption rates and in blood flow in several brain regions of subjects with major depression.33 Moreover, metabolic abnormalities in the anterior cingulate, the amygdala-hippocampus complex, the dorsolateral prefrontal cortex (DLPFC), and inferior parietal cortex seem to improve after antidepressant treatment or after recovery.3435.–36
With phosphorus magnetic resonance spectroscopy (
), the baseline pool of nucleotide triphosphate (NTP)—a product of the cellular utilization of glucose and a marker of the cellular energy availability—was low in subjects who subsequently responded to antidepressant treatment.32 Iosifescu et al.32 also demonstrated for the first time with
metabolite changes were noted in brain-only voxels of responders, but not in nonresponders to antidepressants.
In experimental and animal models, PBM (NIR and red light) noninvasively delivers energy to the cytochrome c oxidase and by stimulating the mitochondrial respiratory chain leads to increased ATP production (see Fig. 1).3738.–39 A study of the effects of NIR on patients with MDD found that a single session of NIR led to a marginally significant increase in regional cerebral blood flow.40 Whether the observed changes in cerebral blood flow resulted from fundamental changes in neuronal metabolism or changes in vascular tone remain to be clarified. Given the correlation of both hypometabolism and abnormal cerebral blood flow with MDD, the beneficial effect of NIR on brain metabolism is one potential mechanism for its antidepressant effect.
Fig. 1
Cellular targets of NIR radiation mechanisms of transcranial NIR for psychiatric disease. The NIR photons are absorbed by cytochrome c oxidase in the mitochondrial respiratory chain. This mitochondrial stimulation increases production of ATP but also activates signaling pathways by a brief burst of ROS. This signaling activates antioxidant defenses reducing overall oxidative stress. Proinflammatory cytokines and neuroinflammation are reduced. Neurotrophins such as brain-derived neurotrophic factor are upregulated, which in turn activate synaptogenesis (formation of new connections between existing neurons) and neurogenesis (formation of new neurons from neural stem cells).

4.
Targeting Inflammation
Animal and clinical research suggests that the inflammatory arm of the immune system contributes to MDD. Post-mortem gene expression profiling on tissue samples from Brodmann area 10 (BA10—prefrontal cortex) have shown that MDD is characterized by increased inflammation and apoptosis.41 In a case-control study, Simon et al.42 found that antidepressant-naive MDD subjects had significant elevations in the following cytokines and chemokines when compared to healthy controls:
,
. Although IL-10 is an anti-inflammatory cytokine, the results suggested that the elevated levels of this IL-10 were likely induced in response to the overall elevation of proinflammatory cytokine levels. In a review of the research on inflammation in MDD, Raison et al.43 proposed that proinflammatory cytokines might cause brain abnormalities that are characteristic of MDD. Indeed, animal research has shown that IL-1 mediates chronic depression in mice by suppressing hippocampal neurogenesis.44
One proinflammatory cytokine that may be of particular relevance to depression is CSF IL-6 (IL6 measured in cerebrospinal fluid). In a recent report, patients with MDD had significantly higher CSF IL-6 levels compared to healthy controls; CSF IL-6 levels were significantly higher than in the serum, and there was no significant correlation between CSF and serum IL-6 levels.45 These findings are consistent with a prior report showing a positive correlation between CSF IL-6 levels and the severity of depression and suicide attempts, with the strongest correlation found in violent suicide attempters.46 One report in a smaller sample of depressed patients has shown that CSF IL-647 was lower or comparable to healthy controls.
NIR light and red light (600 to 1600 nm) decreased synovial IL-6 gene expression (decreased mRNA levels) in a rat model of rheumatoid arthritis.48 In another study, NIR (810 nm) used as a treatment for pain in patients with rheumatoid arthritis decreased production of the following proinflammatory cytokines:
,
, and IL-8.49 Khuman et al.50 showed that transcranial NIR improved cognitive function and reduced neuroinflammation as measured by Iba1+ activated microglia in brain sections from mice that had suffered a TBI. Finally, NIR (970 nm) has been found to be an effective treatment for inflammatory-type acne.51 In summary, it is reasonable to predict that transcranial NIR treatment would likewise have an anti-inflammatory effect in patients suffering from MDD.
5.
Targeting Oxidative Stress
Research has demonstrated a correlation between MDD and vulnerability to oxidative stress.52 For example, depression-induced rats show a significant decrease in glutathione peroxidase (GSH-Px) activity in the cortex.53 Glutathione (GSH) is the most abundant and one of the important antioxidants in the brain; GSH-Px enzymes protect against oxidative stress via reducing hydroperoxides and scavenging free radicals.54 GSH also appears reduced in the brains of MDD subjects.55 Additionally, a study by Sarandol et al.52 demonstrated that MDD patients have higher levels of malondialdehyde, a toxic molecule and a biomarker of oxidative stress.56 Moreover, depressed patients have more red blood cell (RBC) oxidation compared to healthy controls.52 In the same study, the authors found a significant positive correlation between RBC superoxide dismutase (SOD) activity and depression severity. SOD serves to catalyze the removal of the toxic superoxide radical.57 Thus, elevated SOD activity in depressed patients might indicate higher levels of oxidative stress. Finally, catalase activity and nitric oxide (NO) levels have also been shown to be lower in depressed patients than in healthy controls.58 Catalase is an enzyme that protects cells against damaging reactive oxygen species (ROS) via degradation of hydrogen peroxide to water and oxygen.59 NO has protective effects against cell damage, which are likely due to its pleiotropic functions in regulating antioxidant enzymes and many other aspects of cell metabolism.60,61
Oxidative stress may be an effective target for antidepressant treatments. However, successful treatments for MDD vary in regard to their protective effects against oxidative stress.52,53,62 Animal research suggests that PBM may have beneficial effects on oxidative stress. In a rat model of traumatized muscle, NIR (904 nm) blocked the release of harmful ROS and the activation of the transcription factor, nuclear factor κB (NF-κB), both induced by muscle trauma. Trauma activates NF-κB by destroying a specific protein inhibitor of NF-κB called IκB, and this destruction was inhibited by NIR light. Furthermore, NIR reduced the associated overexpression of the inducible form of nitric oxide synthase (iNOS) and reduced the production of collagen.63 This regulation of iNOS is important because excessive levels of iNOS can lead to formation of large amounts of NO that combine with superoxide radicals to form the damaging species peroxynitrite, and can interfere with the protective benefits of other forms of NO synthase.64 These findings suggest that NIR protects against oxidative stress induced by trauma. Finally, an in vitro study of the effects of red light and NIR (700 to 2000 nm) on human RBCs found that NIR significantly protected RBCs against oxidation.65
Original Source: https://www.spiedigitallibrary.org/journals/Neurophotonics/volume-3/issue-03/031404/Review-of-transcranial-photobiomodulation-for-major-depressive-disorder--targeting/10.1117/1.NPh.3.3.031404.full?SSO=1
Treatments for Traumatic Brain Injury With Emphasis on Transcranial Near-Infrared Laser Phototherapy
Larry D Morries, Paolo Cassano, Theodore A Henderson, - This article was published in Neuropsychiatric Disease and Treatment, 20 August 2015 (Publication) 4406This exceptional research indicated prefered wavelenghts and dosages for treating patients with traumatic brain injuries. The found some surprising results.
Abstract:
Traumatic brain injury (TBI) is a growing health concern affecting civilians and military personnel. In this review, treatments for the chronic TBI patient are discussed, including pharmaceuticals, nutraceuticals, cognitive therapy, and hyperbaric oxygen therapy. All available literature suggests a marginal benefit with prolonged treatment courses. An emerging modality of treatment is near-infrared (NIR) light, which has benefit in animal models of stroke, spinal cord injury, optic nerve injury, and TBI, and in human trials for stroke and TBI. The extant literature is confounded by variable degrees of efficacy and a bewildering array of treatment parameters. Some data indicate that diodes emitting low-level NIR energy often have failed to demonstrate therapeutic efficacy, perhaps due to failing to deliver sufficient radiant energy to the necessary depth. As part of this review, we present a retrospective case series using high-power NIR laser phototherapy with a Class IV laser to treat TBI. We demonstrate greater clinical efficacy with higher fluence, in contrast to the bimodal model of efficacy previously proposed. In ten patients with chronic TBI (average time since injury 9.3 years) given ten treatments over the course of 2 months using a high-power NIR laser (13.2 W/0.89 cm2 at 810 nm or 9 W/0.89 cm2 at 810 nm and 980 nm), symptoms of headache, sleep disturbance, cognition, mood dysregulation, anxiety, and irritability improved. Symptoms were monitored by depression scales and a novel patient diary system specifically designed for this study. NIR light in the power range of 10-15 W at 810 nm and 980 nm can safely and effectively treat chronic symptoms of TBI. The clinical benefit and effects of infrared phototherapy on mitochondrial function and secondary molecular events are discussed in the context of adequate radiant energy penetration. Keywords: infrared, traumatic brain injury, TBI, transcranial infrared light therapy, transcranial laser therapy
INTRODUCTION
Traumatic brain injury (TBI) has recently moved into the limelight due to the recognition of its impact on professional athletes and military personnel. Yet, TBI is neither a new problem nor limited to those two populations. The Centers for Disease Control and Prevention estimated that 1.5 million Americans sustained TBI annually in 2000.1 As of 2006, the estimates had risen to 1.7 million brain injuries annually.2,3 Undoubtedly, these point prevalence proportions will increase as military personnel return home,4 and the problem of repeated mild TBI (mTBI) becomes more recognized in sports.5 Current estimates of the prevalence of TBI among veterans range from 9.6%6 to 20%,7 with an estimated total of more than 300,000 cases of TBI among military personnel since 2000.4 The current estimates of the combined number of sportsrelated concussions and brain injuries in the US are 1.6-3.8 million annually.8-10 TBI results in a wide spectrum of neurological, psychiatric, cognitive, and emotional consequences. In part, the variation is related to the severity of the injury (mild, moderate, severe TBI), which is stratified based on Glasgow Coma score, periods of unconsciousness, and degrees of amnesia. Furthermore, the diversity of sequalae can be related to the areas of the brain that are injured, the severity of the injury (highly variable within the classification of “mild” and “moderate”), and the evolution of the injury over time due to neuroinflammatory processes.11,12 Additional mechanisms thought to underlie the damage of TBI include decreased mitochondrial function, calcium and magnesium dysregulation, excitotoxicity, disruption of neural networks, free radicalinduced damage, excessive nitric oxide, ischemia, and damage to the blood-brain barrier. Together, these can contribute to a progression of the damage over time. Patients with TBI can experience headache, visual disturbances, dizziness, cognitive impairment, loss of executive skills, memory impairment, fatigue, impulsivity, impaired judgment, emotional outbursts, anxiety, and depression.3,13-23 The situation can be further clouded by secondary and/ or comorbid posttraumatic stress disorder (PTSD), depression, and anxiety,17-25 which can have symptoms that overlap with those described above and appear to be increasingly likely with repetitive concussive or subconcussive brain injury.5,24,26
TREATMENTS FOR TBI
Pharmacological treatments Pharmacological treatment largely targets the neuropsychiatric sequalae of TBI, rather than providing any means of healing or repairing injury. In general, pharmacological treatment is focused on the modulation of major neurotransmitter systems – dopaminergic, serotonergic, noradrenergic, acetylcholinergic, and glutaminergic.20 Disruption of the major neurotransmitter pathways may result from direct injury or excitotoxicity and other cytotoxic mechanisms. The treatment of depression secondary to TBI is often approached with serotonin reuptake inhibitors. Several studies have examined the benefit of sertraline in post- TBI depression.27-29 Other serotonin reuptake inhibitors also have been examined. Tricyclic antidepressants appear to have some use in the treatment of post-TBI depression, although cautious dose titration is required. Patients with TBI are at greater vulnerability to sedation and cholinergic side effects of confusion and memory impairment. With serotonergic agents other than sertraline, cognitive effects also have been reported.30 Similarly, lithium may be a less desirable agent in this population due to sedation and cognitive impairment. Patients with TBI may respond at lower doses and lower blood levels than expected. Modulation of the dopaminergic system may improve alertness, attention, and cognitive processing speed. The stimulants are most commonly used for this purpose. Methylphenidate facilitates the release of dopamine and slows its reuptake. Dextroamphetamine strongly inhibits reuptake of dopamine, slows down the breakdown of dopamine by monoamine oxidase, and somewhat increases the release of dopamine. These subtle differences are sometimes imperceptible to the patient, but at other times, a patient will do best on one or the other stimulant. Increasing dopamine in the reticular activating system leads to enhanced arousal. Increasing dopamine within the frontal cortex and the striatum leads to enhanced processing speed and attention. Some evidence suggests that the stimulants may enhance neuronal recovery after injury.31-33 There are numerous potential side effects with stimulants, including abnormal heart rhythms, decreased seizure threshold, and death, but these severe side effects are extremely rare. The most common side effects with stimulants are decreased appetite, stomach upset, and headache. These are most severe at the beginning of treatment and improve over time for most patients. Insomnia is another common side effect, which may be more frequent in those with a TBI. Amantadine and bromocriptine may also increase dopamine. Studies of these agents have shown reduced abulia, anergia, and anhedonia in those with TBI.34,35 Amantadine may cause confusion, hallucinations, and hypotension. Small studies have suggested some benefits of bromocriptine in cognitive function.36,37 Arousal-enhancing agents also have found a use in the treatment of the neurocognitive sequalae of TBI. Modafinil is the oldest form of these medications, and armodafinil is an isomer of modafinil with longer activity and less side effects. These medications help to increase alertness and wakefulness. The precise mechanism of action of odafinil is unclear. It appears to increase histamine in parts of the brain involved in controlling the sleep-wake cycle; however, knock-out mice that lack histamine receptors still show increased wakefulness with modafinil.38,39 The picture is also murky for modafinil’s effect on orexins, which are wakefulness molecules in the hypothalamus.40 Modafinil has been shown to weakly bind to the dopamine transporter – like the stimulants,41 and dopamine transporter knock-out mice show no response to modafinil.42 A number of research studies have examined the benefit of these agents in fatigue associated with multiple sclerosis, TBI, cancer, and other conditions. Cognitive and memory impairments after TBI may reflect disruption of cholinergic function. The impact of anticholinergic agents on cognitive function of those with TBI supports this contention. Donepezil is the safest and most widely used of the cholinesterase inhibitors. Several easonably large studies have shown improved memory and cognitive function.43-45 Donepezil has benefits in memory and cognition even several years after injury.45,46 Anticonvulsants are often prescribed initially after a TBI due to heightened risk for seizures. Post-TBI mania or mood lability may respond well to anticonvulsants, such as carbamazepine or sodium valproate. They are also often used to treat aggression after TBI. The anticonvulsant agent, topiramate, has been shown to adversely affect cognitive function in the TBI patients.47 While insomnia is a significant issue for patients with TBI, affecting between 15% and 84% (mean of 40%),3,13,19,21,23,48,49 little has been published on the treatment of this aspect of TBI. Benzodiazepines may be effective but carry a risk of disinhibition. Kemp et al48 found that commonly used sleep aid, melatonin, was not effective. Antidepressants, including serotonin reuptake inhibitors and tricyclic antidepressants, are not effective in resolving insomnia in this population.49 No single agent has emerged as a good solution for this symptom. Cognitive rehabilitation Cognitive rehabilitation now takes many forms and is often individualized to the particular needs of the patients. Protocols have been devised to remediate cognitive difficulties often encountered in those with TBI, such as impaired concentration, executive dysfunction, inattention, visual disturbances, memory dysfunction, and impaired language function. They range from simple strategies (using a planner to aid memory and organization) to specific protocols targeting particular cognitive functions (eg, short-term memory) that can be monitored with sequential neuropsychological testing. These interventions have been extensively reviewed elsewhere.50,51 Comprehensive programs which include psychotherapy and social skills components have been shown to have greater efficacy.50,52,53 Overall, reports of benefits have been mixed.54,55 Behavioral therapies Behavioral remediation strategies to eliminate problematic behaviors following TBI have met with mixed success, most often in terms of the poor generalization of specific skills to the outside world. Behavioral deficits that create difficulties for those with TBI and their families include poor hygiene, decline in tidying/cleaning habits, social withdrawal, reduced social comprehension, impaired memory, and poor organization. Behavioral excesses that create difficulties for those with TBI and their families include aggression, sleep disruption, and perseverations. These have been reviewed elsewhere.56 Nutritional supplements Nutritional supplements, herbs, and nootropics have been utilized for many years and are increasingly popular among the patient populations. There remains little clinical research on many of these agents, perhaps reflecting a lack of funding more than a lack of efficacy. Acetyl-l-carnitine is an ester of l-carnitine and is thought to protect brain cells after injury when glucose metabolic pathways are compromised. During this period, acetyll- carnitine supports alternative ketogenic pathways for metabolism.57 It is also believed to enhance cholinergic function. While there are several clinical studies on patients with Alzheimer’s disease and preclinical data on animal models of TBI, the clinical literature on TBI remains sparse. Ginkgo biloba is a natural product of the tree by the same name. It has been shown to improve membrane fluidity and increase resistance to free-radical damage. It provides some subtle benefits to cognitive function in clinical studies of stroke, dementia, aging, and hypoxia damage.58 It has not been systematically studied in TBI but is used extensively in clinic, often in combination with meclofenoxate which is an avid scavenger of free radicals.59 S-Adenosylmethionine (SAMe) is a nutritional supplement which improves cell membrane fluidity and promotes the production of glutathione, an antioxidant. The benefit of SAMe has been assessed in a single clinical study of TBI.60 Patients receiving SAMe had a 77% improvement in clinical scores of post-concussive symptoms. Citicholine provides a source of choline which can cross the bloodbrain barrier. It has been used extensively in Europe and Japan as a treatment for TBI, stroke, and dementia. However, two large US studies failed to demonstrate significant benefit.61,62 Piracetam and the related oxiracetam and phenylpiracetam have shown some promise as nootropic agents. In one double-blind, placebo-controlled study, piracetam improved several symptoms of postconcussive syndrome, including headache and vertigo.63 More recent clinical studies have shown marginal benefit.64 Huperzine-A, an extract of Japanese club moss, is a natural acetylcholinesterase inhibitor. It may serve as a natural alternative to donepezil, rivastigmine, or galantamine. Galantamine warrants special mention as it appears to also modulate nicotinic eceptors and appears to have more persistent benefit in the treatment of Alzheimer’s disease. It appears to modulate neuroimmune responses, in addition to its effects on acetylcholinesterase.65 Cerebrolysin is a polypeptide that purportedly mimics the actions of neurotrophic factors.66,67 Studies have shown that it can reduce beta amyloid and phosphorylated tau protein accumulation. It may promote neurogenesis, synapse formation, and functional recovery.66 In animal models of acute TBI, cerebrolysin-treated rats had more surviving neurons in the area of impact and showed greater functional recovery.67 In a clinical trial of acute TBI, patients were recruited within 24 hours of injury and treated for 3 months with daily intravenous infusion of cerebrolysin. At 3 months, those receiving cerebrolysin performed significantly better on the Cognitive Abilities Screening Instrument.68 It remains unclear if cerebrolysin provides long-term nootropic benefit. The elevation of free radicals in TBI suggests that antioxidants should be beneficial. Clinical trials of pharmacological antioxidants over the past 30 years have not yielded a useful agent in acute TBI.69 Agents, such as tirilazad70 and polyethylene glycol- onjugated superoxide dismutase, have failed to show benefit in acute TBI. Omega-3 fatty acids may enhance brain repair and recovery, based on animal and clinical studies.71 Similarly, vitamin D may offer neuroprotective and restorative benefits72 in the acute TBI setting. In chronic TBI, vitamin D and omega-3 fatty acids may work synergistically, as they both may reduce neuroinflammation, apoptosis, and oxidative stress.73 Other nutritional supplements have been recommended, but prolonged therapy is necessary to possibly see benefits in TBI. A 6-month trial of ginkgo, vinpocetine, acetyl-lcarnitine, huperzine, alpha-lipoic acid, n-acetyl-cysteine, multivitamins, and over 5 g of omega-3 fatty acids daily yielded improved performance in cognitive testing and increased perfusion (function) in single-photon emission computed tomography (SPECT) scan.74 Long-term use of dietary flavanols may improve cognition in mTBI.75 Transcranial magnetic stimulation Transcranial magnetic stimulation (TMS) has shown some promise in animal models of TBI.76 However, a Cochrane review of the clinical application of TMS for depression noted no difference between repetitive TMS (rTMS) and sham rTMS using the Beck Depression Inventory (BDI) or the Hamilton Depression Rating Scale, except during the initial 2-week period.77 The application of TMS in the post-TBI patients is limited by the risk of seizure induction.78 Hyperbaric oxygen Hyperbaric oxygen treatment has been explored as a treatment for TBI.79-91 Hyperbaric oxygen therapy is neither a benign treatment, given the concerns of oxygen toxicity,79 nor a clear treatment in that the placebo condition of moderate hyperbaric room air also effectively improves cognitive function.80,81 The most carefully performed study compared a group in a cross-over design with an interval of both null treatment and hyperbaric oxygen at 100% oxygen and 1.5 atm.82 The study described improvement in many of the symptoms associated with persistent TBI including headache, tinnitus, vision disturbance, memory dysfunction, and impaired cognitive function. Cognitive testing also showed improvement in attention, information processing speed, and a battery of cognitive tests. In an uncontrolled case series of 16 subjects, Harch et al83 demonstrated that an abbreviated series of hyperbaric treatments using 100% oxygen at 1.5 atm could mitigate subjective symptoms of TBI (eg, headache, sleep disruption, irritability), improve cognitive testing scores, and improve cortical function based on SPECT imaging.83 A study of a higher dose (2.4 atm) did not reveal any significant benefit of hyperbaric oxygen therapy compared to a sham-control group treated with 1.3 atm,84 and this result has been extended and confirmed by a related group.85 However, this may reflect an inverse dose- esponse curve, rather than an absence of benefit, in that the low-dose sham group demonstrated significant changes in cognitive testing and symptom frequency.86 Hyperbaric oxygen remains a controversial area in both acute TBI86-89 and chronic TBI.82,83,85,86,90,91 Physical exercise High-energy activities and exercise programs completed through a health club facility or comprehensive rehabilitation program should focus on the same parameters of an age-adjusted and diagnosis-specific program for aerobic conditioning – flexibility, stabilization, and strength. Though it appears safe and is an accepted intervention for TBI, there is a need for further welldesigned studies.92 Exercise was a part of a 6-month study of lifestyle changes described above which yielded improved function based on cognitive testing and perfusion SPECT scans.74
A NEW TREATMENT FOR TBI
Unfortunately, little has been found to reverse the damage of TBI or repetitive concussion which is the root cause of residual cognitive and psychological impairment following TBI.20,93 One potential avenue of treatment for TBI is infrared light, which has shown promising data in a number of applications. Near-infrared (NIR) light has been investigated for its ability to modulate intracellular mechanisms related to healing. The application of NIR light by low-power laser or by light-emitting diode (LED) is also known as laser phototherapy94 or near-infrared photobiomodulation.92 NIR irradiation can facilitate wound healing,95,96 promote muscle repair,95 and stimulate angiogenesis.95,96 NIR phototherapy has been studied and applied clinically in a wide array of ailments, including skin ulcers,97 osteoarthritis,98 peripheral nerve injury,95,96 low back pain,99 myocardial infarction,100 and stem cell induction.101 The finding that NIR light passes relatively efficiently through bone has spurred interest in its application to treating disorders of the brain. Over the past decade, transcranial near-infrared light therapy (NILT)102 has been studied in animal models to understand its ability to repair damaged or dysfunctional brain tissue resulting from stroke and TBI. The first published study of NILT for TBI in humans described two cases of chronic mTBI with significant disability.103 Each patient was treated with an LED device delivering low-level low-level light therapy (LLLT) in the red and NIR range for 6-10 minutes per area daily for several months. Both patients had marked neuropsychological improvement after a minimum of 7-9 months of LLLT treatment. The precise mechanisms underlying photobiomodulation and its therapeutic benefits are not fully understood. The purported effects of NIR are illustrated in Figure 1. Light in the wavelength range of 600-1,200 nm has significant photobiomodulation capability.104 Current data most strongly support that absorption of NIR photons by cytochrome c oxidase in the mitochondrial respiratory chain is the key initiating event in photobiomodulation.95,96,104,105 This induces an increase in cytochrome c oxidase activity which in turn increases adenosine triphosphate (ATP) production. Such an increase in ATP in wounded or underperfused cells may be sufficient to activate cells in areas of injury or metabolic derangement.106 Data from numerous tissue culture and animal studies point to the importance of several secondary molecular and cellular events. For example, NIR photonic energy can modulate reactive oxygen species,95,96,102 activate mitochondrial DNA replication,95,96 increase early-response genes,95 increase growth factor expression, induce cell proliferation, and alter nitric oxide levels.95,96,102 These mechanisms are more fully described in the companion paper.105 When examined in the specific model of neural tissue injury, NIR phototherapy can lead to demonstrable neural repair and recovery. For example, LLLT of a power density of 0.9-36 J/cm2 applied at 24 hours poststroke in a rodent model yielded a 32% reduction in neurological deficits, as well as histochemical evidence of neuron proliferation and migration.106-108 LLLT had similar benefits in a rodent model of TBI.96,109-111 Interestingly, these cellular changes evolved over a period of days after light exposure and persisted for considerably longer than the interval of actual NIR exposure. These findings are consistent with a progressive regeneration cascade set in motion by the NIR light exposure. NILT in stroke NILT, predominately in the form of LLLT, has been investigated in laboratory models of stroke. LLLT applied in a single dose to an ischemic stroke model appeared to induce expression of the growth factor transforming growth factor – beta 1 and suppress the production of peroxynitrite.112 In a rat model of middle cerebral artery occlusion, LLLT at a dose of 0.5-7.5 mW/ cm2 using continuous wavelength light at 808 nm was administered at 24 hours after the acute stroke.108,113 This single application was estimated to deliver 1.8 J/cm2 in total to the cortex surface and resulted in demonstrable neurological improvement. Functional changes were not manifested until approximately 2 weeks after the single treatment. While there was no significant change in the size of the stroke lesion, histochemical evidence of neurogenesis and migrating neurons108 indicate that a cascade of secondary processes was initiated by NILT. A rabbit model of stroke utilizing injection of a blood clot embolus also demonstrated benefit from LLLT.102,114,115 Herein, 808 nm light was applied with an LED delivering 7.5 mW/cm2 and an estimated 0.9-2.6 J/cm2 to the cortical surface. Cortical ATP levels were increased, indicative of increased mitochondrial activity.114 Significant behavioral recovery was also noted; however, neither ATP increased nor neurological function changed at doses less than 0.3-0.7 J/cm2.114,115 At higher doses of 0.9-15 J/cm2, neurological improvement was seen.114,115 The clinical trials of NILT in acute stroke, the Neuro- Thera Effectiveness and Safety Trials 1, 2, and 3 (NEST- 1,-2, -3), were conducted between 2006 and 2009. The Phase II clinical trial (NEST-1) involved 120 patients in a double-blind, placebo- ontrolled study of the effects of NILT within 24 hours of ischemic stroke.116,117 Approximately 60% of the patients experienced clinical benefit, and the safety profile was very good. Thus, NEST-2, a Phase III clinical trial, was undertaken in 2007. A total of 660 patients were enrolled.118 Somewhat surprisingly, the study did not demonstrate statistical clinical improvement using a different outcome measure.119 Post hoc analysis revealed that a portion of the patients who were moderately affected and/or had strokes limited to the cerebral cortex did realize clinically and statistically significant improvement.102 The NEST-3 trial was halted midpoint when it failed to demonstrate statistical benefit on futility analysis.120 A key factor in the interpretation of the results of NEST-3 is that, different from NEST-1, all types of stroke were included as opposed to just cortical strokes. Continuous laser light has a limited depth of penetration (#1 cm into brain tissue) which likely prevents an effect on deeper brain matter. Therefore, the lack of significant benefits from NIR phototherapy in NEST-3 could be related to the fact that ischemic penumbra was not reached by the light (Luis DeTaboada, personal communication, January 2015). While pulsed NIR was not used in the NEST-3 study, it is estimated that pulsed NIR could penetrate up to 3 cm in depth from the cortical surface, therefore possibly extending the therapeutic target to deeper strokes (Luis DeTaboada, personal communication, January 2015). Figure 1 Hypothesized mechanism of action of NiR light therapy. Notes: NiR light (600-980 nm) penetrates tissue to variable depths depending on wavelength, the tissue involved, coherence, and time. A fraction of the photonic energy reaches the mitochondria and is absorbed by cytochrome c oxidase. This activates increased ATP production, increases production of ROS and RNS, and possibly increases NO. Downstream events include increased early-response genes (c-fos and c-jun) and activation of NF-?B, which in turn induces increased transcription of gene products leading to synaptogenesis, neurogenesis, and increased production of inflammatory mediators and growth factors. Abbreviations: NiR, near-infrared; ATP, adenosine triphosphate; ROS, reactive oxygen species; RNS, reactive nitrogen species; NO, nitric oxide; NF-?B, nuclear factor kappa B. NILT in TBi Oron et al109 conducted the first animal studies of NILT for TBI. They found that a single application of NIR light at 808 nm from a 200 mW emitter at 4 hours post-injury resulted in a significant reduction in lesion size by 5 days.109 To date, several groups have studied NILT in animal models, and this material has previously been reviewed.95,121-123 Single applications of 800-810 nm NIR light within 4 hours of injury have been shown to improve neurological function significantly.110,124-126 The same dose of NIR light at 6 hours was less effective125 and at 8 hours had no appreciable benefit.125 NIR photonic energy at other wavelengths was less effective. Wu et al110 examined red light (670 nm) at 4 hours and found a similar improvement in neurological function; however, 730 nm and 980 nm had no neurological benefit. Similar data for lesion volume have been reported. A single dose of 800-810 nm NIR light (fluence of 36 J/cm2) yielded an approximate 50% reduction in the volume of the lesion at 3-4 weeks110,111,124-126 and a possible reduction in the initial spread of neurological injury, based on the marked reduction in lesion volume found at 5 days post-injury.109 Repeated NIR phototherapy treatments appear to have some benefit, but the frequency and number of treatments are critical factors. While a single NIR light application had benefit, daily applications for 3 days yielded much greater neurological benefit126,127 with smaller lesion size,126 fewer degenerating neurons,126 more proliferating cells,126 and greater levels of brain-derived neurotrophic factor (BDNF)127 compared to a single treatment in a mouse model. In contrast, daily treatment for 7 days128 or 14 days126 showed no difference from controls. NIR energy densities in the range of 0.9-36 J/cm2 resulted in significant biochemical and behavioral changes.109-111,124-127 Pulsing of NIR light appears to yield a greater neurological response but only within certain parameters. Pulsing at 10 Hz yielded greater neurological improvement and a significant reduction in lesion size compared to either continuous-wave or pulsed NIR at 100 Hz.111 In the mouse model of moderate TBI, NILT (800-810 nm) improved learning and memory (Morris water maze performance),128 as well as behaviors associated with depression and anxiety (immobility during tail suspension).111,124 The finding that NILT brought about a smaller lesion in the rodent model of TBI compared to untreated mice suggests that decreased apoptosis, reduced spreading lesion penumbra, and/or neurogenesis are induced by NILT. Indeed, NILT can decrease BAX expression, a pro-apoptosis gene,129 increase expression of BCL-2, an anti-apoptosis gene,129 increase nerve growth factor,95 increase BDNF,127 decrease inflammatory markers,130 and decrease numbers of degenerating neurons.126 Together, these mechanisms may reduce the enlargement of the initial lesion during the first day following the lesion.109 Moreover, increased BDNF and nerve growth factor may contribute to synaptogenesis as shown by increased levels of synapsin-1,127 and neurogenesis, as shown by increased numbers of proliferating cells.127 In a double-blind study in healthy volunteers, NILT was beneficial – compared to sham – in memory and attention.131 In this study, the authors shed only one application of NIR light to the right forehead, targeting the right frontal pole of the cerebral cortex (Brodmann’s area 9 and 10). The device was a Class IV laser CG-5000 (Cell Gen Therapeutics, Dallas, TX, USA), and the parameters were as follows: wavelength 1,064 nm, irradiance 250 mW/cm2, fluence 60 J/cm2, and time 4 minutes per site (two sites).131 The subjects who received the NIR treatment had better attention after 2 weeks, measured by the psychomotor vigilance test. They also had better delayed visual memory at the Delayed Match-to-Sample test. This is the only published controlled trial assessing the impact of NILT on cognition; however, other reports have shown the therapeutic effects of NILT in small numbers of TBI patients. In a two-case report in TBI patients,103 NILT (870 nm) improved sustained attention, memory, and executive functions. Both patients were treated with an instrument with three separate LED cluster heads. The parameters used for the treatment were the following: NIR wavelength 870 nm and 633 nm (red light), irradiance 2.2-25.8 mW/cm2, fluence 13.3 J/cm2, and time 10 minutes per site.103 The same group reported on a cohort of eleven subjects with persistent cognitive dysfunction and treated with a similar NILT protocol for chronic mTBI.132 The eleven subjects received NILT with a device with three LED cluster heads (Model 1100; MedX Health, Toronto, ON, Canada). The parameters used for the treatment were the following: NIR wavelength 870 nm and 633 nm (red light), irradiance 22.2 mW/cm2, fluence 13 J/cm2, and approximate time 10 minutes per site. The NIR light was applied three times per week for 6 weeks (18 sessions), on eleven sites for 10 minutes per site (the total duration of each session was 20 minutes).132 The sites on the skull were chosen on the midline, and bilaterally on frontal, parietal, and temporal areas. At the follow-up neuropsychological testing, NILT had a powerful effect on attention, inhibition, and inhibition switching in the Stroop task, and similarly improved verbal learning and memory, as well as enhanced long-delay free recall on the California Verbal Learning Test. Eight subjects, from the same cohort, were identified as having mild, moderate, or severe depression based on the BDI-II total score (range: 15-34).132 The three cases, who entered the study with only mild depression, remained the same after NILT treatment. Results for the five cases with moderate-severe depression were as follows: two moderate cases improved to mild/minimal depression 8 weeks after the end of NILT series, and one severe case improved to moderate depression. Two moderate or severe depression cases remained the same after 8 weeks of follow-up from the last NILT session.132 Dose response and photonic penetration A prevailing theory in photobiomodulation postulates that a bimodal response curve exists for the biological effects of NIR light.95 The so-called Arndt-Schulz curve (a fundamental principle in homeopathic medicine) is frequently used to describe this biphasic dose response. Some data indicate that low levels of light have a much better effect on stimulating and repairing tissues than higher levels of light. Laboratory studies of cells in culture have demonstrated a bimodal dose response to light exposure in lymphocytes133 and fibroblasts.134,135 For example, Chen et al135 found that a range of 0.03-0.3 J/cm2 was beneficial in activating transcription factors in culture, while 3-30 J/cm2 inhibited the activation of these factors. In contrast, an order-of-magnitude greater dose (2 J/cm2) was best at activating fibroblasts in a superficial wound model.136 Furthermore, an order-ofmagnitude greater dose (30 J/cm2) proved to be best in a rodent joint inflammation model.137 Thus, a dosedependent effect for many biological responses to NIR light has been demonstrated,95,137-139 but the critical parameter is dose at the level of the target tissue, rather than at the surface.137,140 The amount of energy that reaches a volume of tissue at depth is determined by the attenuation of the photonic energy as it passes through the overlying tissue. For example, only 2.45% of the energy from a 980 nm laser emitter penetrates to the level of the peroneal nerve.140 Nevertheless, the biphasic dose response does not appear to be universally true. In primary microglial cell culture, a dose-dependent response to NIR was demonstrated with no detrimental effects at doses as high as 30 J/cm2.141 So a critical question in the use of NILT is that of radiant energy penetration. In particular, some authors have challenged the efficacy of low-power LEDs used in LLLT.142-144 In laboratory studies, LLLT radiant energy is almost entirely absorbed in the first 1 mm of skin.145,146 In two unrelated studies, LLLT diode devices proved to be ineffective in the treatment of diabetic neuropathy,142,144 in contrast with prior reports.147 Similarly, laboratory studies of NILT using LLLT transcranially have not consistently yielded positive results. For example, in a rat model of TBI, Giacci et al148 found no benefit from daily 30-minute irradiation with either 670 nm or 830 nm 0.5 W LED emitters for a period of 7 days. Doses at the skin surface were 28.4 J/cm2 and 22.6 J/cm2, respectively.148 Similarly, treatment of a rat model of contusive spinal cord injury with LLLT (830 nm at 22.6 J/cm2 or 670 nm at 28.4 J/cm2) for 30 minutes per day for 5 days resulted in no significant functional improvement and no reduction in lesion size, despite delivering 2.6 J/cm2 to the spinal cord.148 Lapchak102 reported that the physical parameters of NILT in the clinical trials for the treatment of stroke utilized in the NEST-1 and NEST-2 trials116-120 may have delivered insufficient energy to cortical tissues to be effective. Therein, NIR light of 808 nm wavelength with infrared energy densities of 0.9 J/cm2 was applied to the human scalp for a total of 40 minutes with applications at multiple sites during that time.116,118 Recall that animal models of both stroke and TBI suggest that NIR energy densities in the range of 0.9-36 J/cm2 resulted in significant biochemical and behavioral changes.96,106-115,125-127 The concern raised from the NEST studies102 is that current clinical trials testing the effectiveness of lowenergy NIR diodes to treat TBI may yield negative or inaccurate efficacy data, not because of a failure of infrared light to invoke a change but due to a dose error. Doses that are effective when directly applied to cells in a Petri dish149,150 or to 3-5 mm thick rodent brains96,109-111,125,126,128 may be insufficient to penetrate 2-4 cm into the human brain. In a companion paper, our own studies of photonic energy penetration are detailed.105 To summarize, the laboratory tissue studies showed that 0.5 W LED emitters did not penetrate the 2 mm thickness of human skin. No detectable energy from 0.5 W LED NIR light emitters could be detected penetrating a similar thickness (1-2 mm) of sheep skin or 3 cm thick section containing sheep skin, skull, and brain. In contrast, 11% of the photonic energy from a 10 W 810/980 nm coherent NIR laser penetrated 2 mm of human skin. Similarly, 17% of the photonic energy from a 15 W 810 nm coherent NIR laser penetrated the same distance.105 Energy from these more powerful NIR emitters could be detected penetrating 3 cm of sheep skin, skull, and brain with 0.4% of the 10 W 810/980 nm NIR laser’s energy reaching the depth of 3 cm and 2.9% of the 15 W 810 nm NIR laser’s energy traversing the same distance.105 Anders also has demonstrated penetration of 808 nm light to 40 mm in the brain using a 5 W laser emitter (JJ Anders, personal communication, January 2015). Prompted by the mixed results in the literature and the observations by Lapchak,102 Franzen-Korzendorfer et al,144 Wan et al151 and Lavery et al142 we have been utilizing relatively high-power (10- 5 W) lasers at the wavelengths of 810 nm and 980 nm in the clinic to treat patients with TBI. Clinically, the patients have shown excellent responses with resolution of many of their long-standing symptoms of TBI or post-concussive syndrome. Below is a retrospective series of such patients to illustrate the extent and character of response to this modality. Methods Patients in the case series were sequentially treated patients at a clinic which is engaged in ongoing NILT for a number of clinical conditions. The risks, benefits, and current state of research on the use of NILT were explained to each patient. Each patient consented to treatment. Institutional Review Board approval was obtained in a post hoc review, noting that the risk-benefit ratio was acceptable. Between March 16, 2011 and February 20, 2013, sequential new referrals for chronic mild-to-moderate TBI were evaluated for treatment and selected for NILT using Class IV lasers, either the LT1000 (LiteCure, Newark, DE, USA), a 10 W adjustable NIR laser emitter with wavelengths of 810/980 nm capable of delivering continuous or pulsed NIR light, or the Diowave 810 (Diowave, Riviera Beach, FL, USA), an adjustable NIR emitter up to 15 W with a wavelength of 810 nm capable of delivering continuous or pulsed NIR energy. Demographics and laser treatment settings are detailed in Table 1. The fluence delivered to the skin of patients ranged from 55 J/cm2 to 81 J/cm2. No other treatment modalities (medications, exercise regimen, supplements) were added, discontinued, or changed while receiving NILT. Symptoms were monitored clinically. A baseline Quick Inventory of Depressive Symptomatology Self-Report (QIDS-SR)152 was completed for all patients, and the BDI153 was administered to seven of the ten patients before and after the course of treatment. In addition, each patient was instructed on how to create and maintain a patient and spousal diary of symptoms and subjective progress. Each of six patients received a single series of ten treatments with the LT1000 Class IV laser. Three additional patients each received a single series of 20 treatments with the LT1000 Class IV laser. One patient was treated with the Diowave 810 nm Class IV laser device in a series of 20 treatments. The patients and treating clinician wore protective eyewear. There were no incidents of burns or thermal discomfort (Figure 2). The impact of high-watt NILT While the patient group represented a diverse mix (Table 1 presents demographics), some notable commonalities of symptoms emerged. Over 90% of the patients had complaints of anxiety, depression, irritability, and insomnia. Other symptoms included headache (60%), suicidal ideation (50%), cognitive difficulties (50%), attention problems (50%), short-term memory problems (40%), loss of libido (30%), substance abuse (20%), fatigue (20%), and panic attacks (20%). Six of the patients were unemployed prior to treatment. Three of the patients were experiencing severe marital difficulties. All carried or had a confirmed diagnosis of TBI, but other comorbid diagnoses included PTSD, major depressive disorder, generalized anxiety disorder, bipolar disorder, and attention deficit/hyperactivity disorder. The patients’ baseline scores on the BDI were 25.3±12.1 (moderate depression range), and baseline scores on the QIDS-SR were 12.9±4.6 (moderate depression range). During NILT treatments, skin temperature increased no more than 3°C with rapid cooling after removal of the NIR light. A continuous sweeping motion was utilized to minimize skin heating and cover a larger area. After a course of ten treatments of NILT (20 treatments in four patients), each patient experienced significant clinical improvement with resolution of many of their symptoms (Table 2). In addition, the BDI scores dropped to 12±6.5 (nondepressed range). This represented a significant decrease (P,0.01, Student’s t-test, one-tailed, Microsoft Excel). The QIDS-SR scores after treatment were 2.2±2.3 (nondepressed range), and the difference from baseline was highly significant (P,0.00001, Student’s t-test, one-tailed). Patients noted improvement in cognitive function, mood, anxiety, and sleep. None of the patients continued to have suicidal thoughts (50% at baseline). Other symptoms, such as anxiety and irritability, were markedly improved. Most notable were the nonquantifiable changes in patients’ lives. Patients reported improved cognitive ability and a desire to return to meaningful work. Five of the six unemployed patients have returned to work. The two patients who were Iraq/ Afghanistan veterans have found new careers in highly skilled trades. The patients with marital difficulties have reconciled and were purchasing homes or otherwise solidifying their marriages. The clinical change can be attributed to NILT because no changes in medications, supplements, or exercise regimen were permitted during the course of NILT treatment. All patients in the case series experienced significant clinical improvement which supports the conjecture that high-power NIR laser delivers sufficient energy to the human brain for photobiomodulation to occur. Insomnia and suicidal ideation, common symptoms in those with TBI or post-concussive syndromes,3,17-20,24,25 resolved in 100% of cases. Headache, another common symptom for patients following a TBI,6,14,15,23 was reduced or resolved in the six patients so afflicted. Symptoms such as anxiety,14,15,21,24 depression,21,24,25,27-29 and irritability resolved or were dramatically reduced in all patients. Cognitive function appeared to improve based on return to work or improved work performance, although cognitive tests were not performed. The quality of life dramatically improved in all cases, based on the observations of the patients, their family members, and the treating clinician. At follow-up intervals of 6-7 months post-treatment, patients have reported continued improvements in symptoms. The precise areas of brain injury were not elucidated in Figure 2 Treatment parameters per individual, based on area of the skull treated. Notes: Dimensions varied per head/skull size and hair line. Treatment was warm and comfortable for each patient. There were no incidences of discomfort. Areas treated were (A) temporal- ilateral, (B) frontal, and in patients 1-3, 5, and 6 (B) frontal only. Table 1: Infrared light treatment parameters for each of the ten patients in the case series Patient Area treated Sex Mechanism of TBI Interval since TBI Wavelength of Dosage per area Duration before treatment NIR-PT dual wave Scanning technique per area pulsed 10 Hz 1 B, bilateral frontal Male Concussive blast 2 years 810 and 980 nm 2,700 J 10 minutes Fluence – 20.45 J/cm2 2 areas Area – 132 cm2 10 visits 2 B, bilateral frontal Female MVA 18 years 810 and 980 nm 2,400 J 9 minutes Fluence – 18 J/cm2 2 areas Area – 133 cm2 10 visits 3 B, bilateral frontal Female MVA 5 years 810 and 980 nm 2,400 J 8 minutes Abuse Fluence – 18.3 J/ cm2 2 areas Area – 131 cm2 10 visits 4 A–B, bilateral frontal, left temporal Female MVA x2 8 years and 13 years 810 and 980 nm 2,400 J 8 minutes Fluence – 18.3 J/cm2 3 areas Area – 131 cm2 10 visits 5 B, bilateral frontal Male Vietnam Veteran 20+ years 810 and 980 nm 3,000 J 10 minutes Concussion Fluence – 28.3 J/cm2 2 areas Child abuse Area – 106 cm2 10 visits 6 B, bilateral frontal Male Concussion 5+ years 810 and 980 nm 2,400 J 12 minutes Fluence – 14.8 J/cm2 2 areas Area – 162 cm2 10 visits 7 B–A, bilateral frontal, left temporal Male Afghanistan, Iraqi Disability 810 and 980 nm 3,000 J 10 minutes Disability due to TBI 2 years Fluence – 22.7 J/cm2 3 areas Area – 132 cm2 20 visits B–A, bilateral frontal, bilateral temporal Female Hypoxic encephalopathy Childbirth-related 810 and 980 nm 2,700 J 9 minutes injury, 8 years Fluence – 27.8 J/cm2 3 areas Area – 97 cm2 20 visits 9 B–A, bilateral frontal, bilateral temporal Male MVA-TBI Numerous episodes 810 and 980 nm 3,000 J 10 minutes Concussions Fluence – 22.72 J/cm2 3 areas Area – 132 cm2 20 visits 10 B–A, bilateral frontal, left temporal Female Bicycle vs car >30 days 810 nm single 2,700 J 9 minutes Concussion, amnesia, LOC wavelength – Fluence – 17.1 J/cm2 3 areas different device Area – 158 cm2 20 visits Note: All safety precautions were followed, including metal protective eyewear (laser eye protection). Abbreviation: LOC, loss of consciousness; MvA, motor vehicle accident; TBi, traumatic brain injury. the majority of these cases, so a correlation of symptoms changes and cortical function changes cannot be made; however, perfusion SPECT imaging in other patients has shown significant increases in perfusion in injured areas of the brain and overall improved cortical function following similar courses of high-watt NILT.154 One concern that has been expressed about high-watt NIR lasers is the risk of tissue heating.155 We explored this issue in our companion paper on NIR penetration.105 Temperature change was 1°C-3°C at the skin surface using continuous-wave NIR lasers in the range of 10-15 W. Using pulsed settings, the high-powered lasers showed no significant temperature change in tissue samples. The temperature change on human skin was 1°C or less in the in vivo penetration studies while maintaining continuous movement of the laser probe head.105 Clinically, patients in this case series reported only slight warming of the skin, but no discomfort, using the continuous motion technique. Laboratory studies have largely focused on treatment of acute brain injury. The processes involved in the benefits of NIR light in chronic TBI as seen in this clinical case series may be quite distinct. Nevertheless, Schiffer et al156 found that a single application of LLLT at 810 nm and 250 mW to the forehead over 8 minutes reduced depression and anxiety symptoms in ten patients for approximately 2 weeks. Similarly, the small case series by Naeser et al103 demonstrated some benefit using NIR light, albeit at very low power levels over a prolonged course of several months with only transient benefit. Together with our clinical data, these findings suggest that at least some of the photobiomodulatory effects of NIR energy likely do occur in chronic neurological conditions. Prior presentations on NILT for the treatment of TBI or stroke in humans have focused on getting photonic energy through the skull to the cortex surface which traverses a distance of about 6-10 mm; however, this model is flawed in that the distance to the areas of damage may be far greater. In other words, the cortex immediately subjacent to a portion of the skull may be 10 mm from the surface, but the NIR light energy may need to penetrate 3-7 cm to reach areas of damage. Much of the cortical surface is actually lining the walls and floors of sulci, rather than immediately subjacent to the skull. Analysis of NIR spectroscopy reveals that light propagation through varying media with irregular boundaries is subject to high levels of scatter.157 In addition, review of the neuroimaging literature on TBI has revealed that the most common areas injured in TBI are the orbitofrontal cortex (at the ventral surface of the frontal lobe) and the anterior and medial temporal lobes.158 It is not anatomically possible to position an NIR light emitter immediately exterior to the skull overlying these areas. Indeed, the orbitofrontal cortex positioned immediately above the eyes can only be reached from the forehead by angling the light emitter. Similarly, the temporal lobes are separated from the surface by epidermis, dermis, subcutaneous fat, subcutaneous blood vessels, accessory head of the temporalis muscle, connective tissue, temporalis muscle, skull, and dura mater.159 Each of these structures has different absorption and refraction properties, and each interface between different materials also creates a barrier to transmission of photonic energy.157 Blood flowing in the subcutaneous vessels is believed to create a unique barrier to transmission.160 In summary, effectively targeting the areas most commonly injured in TBI with sufficient photonic energy to initiate reparative processes represents a significant challenge in NILT. This appears to have been overcome with the high-power laser protocol presented here and in a related paper.154 As yet, the mechanism of action of NILT in treating TBI is not entirely clear. Moreover, the neurological benefits are not immediately apparent. Rather, a delay of 1-4 weeks was noted, consistent with a progressive regeneration cascade set in motion by the NILT.96,103,105 ,107,109,121,122,124,127,135 Similarly, most of the patients in the present case series did not notice benefits immediately or within the first few treatments. Instead, they reported benefits emerging over an interval of weeks, and in some cases, continuing after completion of the course of NILT. In addition, the clinical improvement reported by the patients in the above case series is more profound than that reported by patients treated with LLLT or low-powered lasers.103 In fact, we observed that among seven subjects with documented moderate depression, per BDI scores, four had an antidepressant response (≥50% decrease of depression severity). In contrast, Naeser et al132 reported that out of eight subjects with TBI and comorbid depression, only three had a significant improvement in their depressive symptoms (37.5%). Our results may be due to the greater penetration of more powerful, coherent, and pulsed NIR light from a laser source. A unique outcome measure was developed for this protocol (Morries and Henderson, unpublished data, 2015). A patient diary and separate spousal diary provided a weekly update of patient’s response in his or her home environment. This novel approach to capturing the patient treatment experience provided the patient and family with tangible and pertinent documentation of the clinical response. While time consuming, the experiences recorded in these diaries proved to be valuable clinical tools to the treating clinicians.
CONCLUSION
To date, there has been little progress in developing effective treatments for chronic mild-to-moderate TBI or repetitive concussions. This area of need has become even more pressing with the return of veterans from military conflicts in Iraq and Afghanistan4,6,7,16,17,19,161 and the recognition of the magnitude of sport-related TBI.5,8-10 In addition, the dramatic growth in the geriatric population with attendant proprioceptive dysfunction has resulted in a rising incidence of fall-related TBI.162 NILT has shown promise as a tool for the treatment of TBI. A critical issue is to assure that adequate photonic energy reaches the injured areas of the brain. The use of high-wattage lasers, as we have demonstrated, results in marked clinical improvement in patients with chronic TBI. Moreover, symptoms consistent with PTSD, anxiety, and/or depression also improved considerably or resolved in this group of patients. Further work in the use of highwattage NILT in the treatment of TBI, depression, and other neurological disorders is encouraged.
ACKNOWLEDGMENTS
The authors would like to acknowledge the technical assistance of Mr Charles Vorwaller (Aspen Lasers) and Lite Cure Corporation. The authors also acknowledge the contribution of Ms. Taylor Tuteur in the artistic creation of Figure 1.
DISCLOSURE
Dr. Larry D Morries is the CEO of Neuro-Laser Foundation, a nonprofit foundation. He has a private practice in Lakewood, CO. Theodore A Henderson is the president of The Synaptic Space, a medical consulting firm. He is Table 2 NiLT case series with demographics, symptoms, and treatment response
PRETREATMENT POSTTREATMENT
Patient # Sex Occupation Mechanism of TBI Diagnoses Sleep Symptoms Suicidal BDI Sleep Symptoms Suicidal BDI 1 M Veteran, Blast – 5 years; TBI, PTSD, MDD Primary and H, S, I, D, X, L, A, M, + – Resolved None, back No – unemployed Iraqi middle C, SL with spouse, insomnia working 2 F Nurse, MVA – 8 years TBI, PTSD Middle and H, F, I, X, C, A, STM, L, + 18 Resolved A and HA – No 15 unemployed terminal HA, SL but mild, insomnia return to work 3 F Unemployed Assault and TBI, PTSD, MDD, Primary and D, X, P, M, L, HA, S, + 23 Resolved HA – mild, No – MVA, 5 years GAD, ADHD middle insomnia, SA, C, N, STM back with Prior nightmares spouse, no SA, working 4 F Unemployed MVA – 3 years, TBI, PTSD, MDD Primary and D, X, HA, I, M, SA, S, N + 23 Resolved None, marriage No 17 assault middle insomnia, improved, numerous violent nightmares no SA, working 5 M Veteran, Blast – 20+ years TBI, MDD, GAD Primary and D, X, I, S, SL + 18 Resolved None No 1 unemployed 1960s; Vietnam middle insomnia 6 M executive Trauma – TBI, GAD, MDD Primary D, X, I, P, HA, A, S – – Resolved HA, X, and P – No – chronic insomnia but improved 7 M Veteran, Multiple blasts TBI, MDD, GAD Primary and S, D, I, X, C, A, S, STM, – 22 Resolved HA and C – No 16 disability (>12); Afghan middle HA mild, new and Iraqi wars insomnia career 8 F Student Childbirth TBI, learning Primary D, I, X, C, A, SL, F, STM – 16 Resolved, STM improved, No 7 disorder insomnia no bads reading .20% dream more animated 9 F Sales MVA and TBI, LOC Primary and HA, SL, N, D, I, X, H, A – 29 Resolved Mild HA, No 9 sports TBI middle insomnia, job nightmares promotion 10 F Physicist Recent car– TBI, LOC, amnesia Primary and D, I, X, neck, knee pain – 51 Resolved No loss No 19 bicycle middle of skills, accident insomnia maintain intellectual job Notes: Demographics for each of the ten patients in this case study is presented. Also presented is their history of mechanism of injury, diagnosis, and related symptoms. Changes in anxiety levels, sleep patterns, depression, and suicidal ideation were important symptoms and outcomes to track. Patients were instructed for no medication changes, with their primary treatment provider’s approval. Cognitive difficulties, attention problems, and short-term memory difficulties were by patient interpretation of their symptomatic improvement and patient diary changes. Symptom occurrence % was as follows: Anxiety – 100%, Depression – 90%, Irritability – 90%, Primary And Middle Insomnia – 90%, Headache – 60%, Sadness – 60%, Suicidal Ideation – 50%, Cognitive Difficulties – 50%, Attention Problems – 50%, Short-Term Memory Problems – 40%, Marital Difficulties – 30%, Loss Of Libido – 30%, Substance Abuse – 20%, Fatigue – 20%, Panic Attacks – 20%. Abbreviations: NILT: Near-Infrared Light Therapy, TBI: Traumatic Brain Injury, PTSD: Post-traumatic Stress Disorder, MDD: Major Depressive Disorder, GAD: General Anxiety Disorder, ADHD: Attention Deficit/Hyperactivity Disorder, H: Hyperarousal, S: Sadness, I: Irritability, D: Depression, X: Anxiety, L: Loss Of Libido, A: Attention Problems, M: Marital Difficulties, C: Cognitive Problems, SL: Sleep Issues, F: Fatigue, STM: Short- erm Memory Problems, HA: Headache, P: Panic Attacks, SA: Substance Abuse, N: Nightmares, BDI: Beck Depression Inventory, LOC: Loss of Consciousness, MVA: Motor Vehicle Accident. the president of Dr. Theodore Henderson, Inc., a clinical service firm. He is the co-owner of Neuro-Luminance, a clinical service organization. He is the president of the International Society of Applied Neuroimaging. He is the CFO of the Neuro-Laser Foundation, a nonprofit foundation. Dr. Paolo Cassano received funding from the Brain and Behavior Research Foundation; Photothera Inc and from the Dupont Warren Fellowship (Harvard Medical School) to conduct research on NIR light for the treatment of major depressive disorder.
ABOUT THE AUTHORS:
Larry D. Morries, DC brings a distinguished 30-year career studying and treating the brain and body through his private practice based in Lakewood, Colorado. As Neuro-Laser Foundation’s co-founder, his chiropractic expertise is complemented with extensive study of near infrared-light therapy applications, clinical radiology, clinical neurology and sports injury and rehabilitation. In practice since 1973, Dr. Morries has contributed extensively to both chiropractic and medical professions throughout his career. He is a recognized expert often called upon for review services, treatment utilizations, and documentation presentations. In recent years, he has guided the Colorado State of Colorado Workers Compensation Board with a review of treatment guidelines for Chronic Pain, and Complex Regional Pain Syndrome, Shoulder Pain, Low Back Pain, Traumatic Brain Injury, and was asked to present in 2016 on Thoracic Outlet Syndrome.
Other professional involvement include:
• Colorado Chiropractic Association, Board member, President in 1982, Chairman in 1984
• Colorado Chiropractic Society, Vice President and Secretary in 1995-2004
• Colorado Chiropractic Journal Club, Chairman,since 2008
Dr. Morries has continued his study of the human body and brain with postgraduate work in Neurodiagnostic testing at the American Academy of Neurology, and Harvard Medical School-Massachusetts General Hospital. He is also educated on Spinal Mechanics at Chicago Rehabilitation Institute. He earned his Doctorate in Chiropractic from Logan Chiropractic College, with recognition as Student Clinical Director, Teaching Assistant in Radiology. Dr. Morries is most proud of his research papers and awards, in America Academy of Pain Medicine, Sciatic and Suprascapular Nerve Blocks with Dr. Steve Gulevich, MD. He was asked to share two Poster presentations at the North American Laser Foundation in 2011on Low Back Pain, plus Polyneuropathy treatment with Laser (NIR) therapy. His Podium Presentation and publication on Hip dysplasia, in American Board of Chiropractic Sports Physicians®. Additionally, he has given presentations abroad at State of Chiropractic Research, Foundation of Chiropractic Education and Research, in Bournemouth England and Vancouver, BC, Canada. Dr. Theodore Henderson has extensive training and experience to the practice of Psychiatry. He trained in Psychiatry at the prestigious Barnes/Jewish Hospitals at Washington University in St. Louis. Dr. Henderson completed a fellowship in Child & Adolescent Psychiatry at the University of Colorado. He also has training in Radiology, Nuclear Medicine, and the genetics of psychiatry. He established his private practice in Centennial Colorado in July of 2000. Dr. Henderson brings a unique blend of expertise in psychopharmacology, neurobiology, and an understanding of human nature to the practice of psychiatry. Dr. Henderson attended medical school at Saint Louis University School of Medicine. While in medical school, he began studying heart pathology under Dr. Vernon Fischer. He earned an American Heart Association Medical Student Research Fellowship. With this fellowship, he spent one year at the University of Washington studying the pathology of atherosclerosis. In 1991, Dr. Henderson founded the Child Abuse Prevention Task Force at Saint Louis University. This program taught children, parents, and teachers about child sexual abuse and how to prevent it. Each year, this program reached over 8,000 children throughout the metro St. Louis area, primarily in the poor inner-city schools. The program was awarded numerous awards, including a Saint Louis University Community Service Award, Commendations from the school districts, and an award from the American Medical Student Association. Dr. Henderson was nominated for a Student Life Leadership Award and earned a Departmental Award from the Department of Community and Family Medicine. He also received a Weis Humanitarian Award recognizing outstanding humanitarian care as a medical student. Dr. Henderson wrote a training manual on this program that was implemented at other medical schools and he cowrote a book chapter in the book, A Parent’s & Teacher’s Handbook on Identifying and Preventing Child Abuse (1998). During graduate school and medical school, Dr. Henderson published numerous research studies. He published 9 articles and 27 abstracts about his research in brain development. He also published a book chapter on brain development in collaboration with his research professor, Dr. Mark Jacquin. His research focused on the role of neural growth factors and impulse activity on the development of brain organization. He collaborated with leading researchers, including Drs. Thomas Woolsey, Eugene Johnson, and Thomas Rhoades. While a medical student, Dr. Henderson wrote two research grants (as part of program project grants). Both were funded. He continued conducting research at Saint Louis University and Washington University throughout his residencies. Dr. Henderson trained for one year in Radiology, focusing on neuroimaging and pediatrics. With this strong base, he then undertook a residency in Psychiatry at Washington University’s program at Barnes/Jewish Hospitals in St. Louis. His residency included extended training in general pediatrics at St. Louis Children’s Hospital. In 1997, He was awarded the National Institute of Mental Health Outstanding Resident Award for his ongoing work in child abuse prevention and his neurobiological research while a resident. Dr. Henderson completed a residency in Adult (or General) Psychiatry and then undertook a fellowship in Child Psychiatry at the University of Colorado. This included additional specialization in Autism and Autism Spectrum Disorders. He compl
Photobiomodulation for Traumatic Brain Injury and Stroke
Michael R Hamblin - J Neurosci Res. Author manuscript; available in PMC 2018 Oct 1. (Publication) 4533This study compare wavelength and pulsing frequencies to find the highest efficacy. It shows how much better 810nm (fig 3)and 10Hz (fig 4) are superior for TBI.
There is a notable lack of therapeutic alternatives for what is fast becoming a global epidemic of traumatic brain injury (TBI). Photobiomodulation (PBM) employs red or near-infrared (NIR) light (600-1100nm) to stimulate healing, protect tissue from dying, increase mitochondrial function, improve blood flow and tissue oxygenation. PBM can also act to reduce swelling, increase antioxidants, decrease inflammation, protect against apoptosis, and modulate microglial activation state. All these mechanisms of action strongly suggest that PBM delivered to the head should be beneficial in cases of both acute and chronic TBI. Most reports have used NIR light either from lasers or from light-emitting diodes (LEDs). Many studies in small animal models of acute TBI have found positive effects on neurological function, learning and memory, and reduced inflammation and cell death, in the brain. There is evidence that PBM can help the brain to repair itself by stimulating neurogenesis, upregulating BDNF synthesis, and encouraging synaptogenesis. In healthy human volunteers (including students and healthy elderly women) PBM has been shown to increase regional cerebral blood flow, tissue oxygenation and improve memory, mood and cognitive function. Clinical studies have been conducted in patients suffering from the chronic effects of TBI. There have been reports of improvements in executive function, working memory, and improved sleep. Functional magnetic resonance imaging has shown modulation of activation in intrinsic brain networks likely to be damaged in TBI (default mode network and salience network).
Keywords: photobiomodulation, low-level laser therapy, traumatic brain injury, stroke, chromophores, animal studies, clinical trials, human studies
1. Introduction
Photobiomodulation (PBM) formerly known as low-level laser (light) therapy (LLLT) is approaching its 50th anniversary, after being discovered by Endre Mester working in Hungary in 1967 (Hamblin et al. 2016). Originally thought to be a property of red lasers (600-700 nm), PBM has broadened to include near-infrared (NIR) wavelengths 760-1200 nm, and even blue and green wavelengths. Moreover the advent of inexpensive and safe light emitting diodes (LEDs) has supplanted the use of expensive lasers in many indications. The better tissue penetration properties of NIR light, together with its good efficacy, has made it the most popular wavelength range overall. The best-known medical applications of PBM have been for indications such as stimulation of wound healing (Hopkins et al. 2004; Kovacs et al. 1974), reduction of pain and inflammation in orthopedic and musculoskeletal conditions (Aimbire et al. 2006; Gam et al. 1993), and mitigation of cancer therapy side-effects (Zecha et al. 2016a; Zecha et al. 2016b). However in recent years there has been growing interest in the use of PBM in various brain disorders (Hamblin 2016b; Hennessy and Hamblin 2016; Naeser and Hamblin 2011; Naeser and Hamblin 2015). The almost complete lack of any adverse side-effects of PBM, coupled with growing disillusion with pharmaceutical drugs that affect brain function, have combined together to suggest an alternative physical therapy approach to improving brain function.
Traumatic brain injury (TBI) is caused by some type of trauma to the head, often resulting from road traffic accidents, assaults, falls, sports injuries, or blast injuries suffered in military conflict. TBI is classified as mild (loss of consciousness 0-30 minutes; altered mental state <24 hours; post-trauma amnesia <1 day); moderate (loss of consciousness 30 minutes to 24 hours; altered mental state >24 hours; post-trauma amnesia >1-7 days), or severe (loss of consciousness >24 hours; altered mental state >24 hours; post-trauma amnesia >7 days) (Blennow et al. 2016). There are three cases of TBI sustained each minute in the US (Faul et al. 2010). Repeated mild episodes of TBI (also known as concussions) even without loss of consciousness, may have devastating cumulative effects (Kamins and Giza 2016). Chronic traumatic encephalopathy is a recently recognized condition resulting from repeated head trauma, found in boxers, football players, and military personnel (McKee et al. 2016; Safinia et al. 2016). There is presently no accepted treatment for TBI, although some investigational approaches are being tested in both the acute (neuroprotection) and chronic (neurorehabilitation) settings (Loane and Faden 2010). One of these novel approaches is PBM or LLLT (Hamblin 2016a; Hamblin 2016b; Huang et al. 2012; Thunshelle and Hamblin 2016).
2. Mechanisms of action
Uncertainties about the mechanism of action of PBM at the molecular and cellular levels, have undoubtedly held back its acceptance in the wider biomedical community. However in recent years substantial progress has been made in this regard (de Freitas and Hamblin 2016). In the following section the state-of-the-art knowledge about the mechanisms of PBM is summarized. Figure 1 shows a graphical representation of the cellular and molecular mechanisms of PBM.
Molecular mechanisms of tPBM
Light passes through the scalp and skull, where depending on the wavelength it is absorbed by two different chromophores. Red and NIR (up to 940nm) is primarily absorbed by cytochrome c oxidase in the mitochondrial respiratory chain of the cortical neurons. Longer wavelength NIR light (980nm, 1064nm) is primarily absorbed by heat and light-sensitive transient receptor potential ion channels. In both cases cell signaling and messenger molecules are upregulated as a result of stimulated mitochondrial activity, including reactive oxygen species (ROS), and adenosine triphosphate (ATP). hv is light, TRPV is transient receptor potential vanilloid (ion channels).
2.1 Chromophores
The first law of photobiology states that a photon must be absorbed by some molecule within the tissue to have any biological effect. The identity of these chromophores has been the subject of much scientific investigation and speculation. Largely due to the efforts of Tiina Karu in Russia, the enzyme cytochrome c oxidase (CCO) has been identified as a major chromophore of red/NIR light (Karu 1999; Karu and Kolyakov 2005; Karu et al. 2004a; Karu et al. 2004b). CCO is unit IV in the mitochondrial respiratory chain and has absorption peaks reaching well into the NIR spectral region (up to 900 nm) as well as in the red and blue regions. The most discussed hypothesis to explain exactly how photon absorption can stimulate the activity of CCO involves the photodissociation of inhibitory nitric oxide (NO) that can bind to the copper and heme centers in the enzyme and prevent oxygen from gaining access to the active sites (Lane 2006). In experimental models (such as isolated mitochondria) oxygen consumption and ATP production are increased, and the mitochondrial membrane potential is raised (Passarella et al. 1984).
A less well-appreciated mechanism involves light and heat-gated ion channels. These cation ion channels are thought to be members of the transient receptor potential (TRP) superfamily consisting of over 28 distinct members organized into six subfamilies, based on their primary amino acid structures (Caterina and Pang 2016). TRPV (vanilloid sub-family) members including TRPV1 (capsaicin receptor) have been shown to be activated by various wavelengths of light including green, red and NIR.
2.2 Cellular mechanisms
After the primary photon absorption event occurs, whether that the photons are absorbed by CCO, or by TRP ion channels a series of secondary events occurs. One of these events is the generation of reactive oxygen species (ROS), which are thought to be produced inside the mitochondria due to an increase in electron transport, and a rise in the mitochondrial membrane potential above the baseline levels (Suski et al. 2012). It should be noted that mitochondrial ROS can be produced when MMP is raised above normal, and also when ROS is reduced below normal. It is thought that the ROS produced when MMP is lowered (mitochondrial dysfunction) are more damaging than ROS produced when MMP is raised (mitochondrial stimulation). Nitric oxide is produced after PBM (Hamblin 2008), possibly by photodissociation from CCO where it inhibits oxygen consumption and electron transport (Lane 2006). Cyclic adenosine monophosphate (cAMP) (Gao and Xing 2009) and intracellular calcium are increased (Alexandratou et al. 2002). Many of these secondary mediators in the signaling pathways triggered by PBM, can induce activation of transcription factors, that go on to upregulate or downregulate expression levels of a large number of genes. One of the best-known transcription factors is NF-kB that can regulate expression of over one hundred genes including proteins with antioxidant, anti-apoptotic, pro-proliferation, and pro-migration functions. PBM (810 nm 3J/cm2) was shown to activate NF-kB in mouse embryonic fibroblasts via ROS production (Chen et al. 2011a). Since NF-kB is known to be a pro-inflammatory transcription factor, it might be thought that PBM would be pro-inflammatory. However it was shown that NF-KB was decreased in already activated (treated with Toll-like receptor ligands) inflammatory dendritic cells by PBM (810 nm 3J/cm2) (Chen et al. 2011b).
2.3 Tissue mechanisms
The changes in expression levels of proteins involved in antioxidant and redox-regulation, anti-apoptotic and pro-survival, cellular proliferation, etc mean that distinct changes in tissue homeostasis, healing and regeneration can be expected after PBM. For instance, structural proteins such as collagen are newly synthesized in order to repair tissue damage (Tatmatsu-Rocha et al. 2016). Cells at risk of dying in tissue that has been subjected to ischemic or other insults are protected (Sussai et al. 2010). Stem cells are activated to leave their niche, proliferate and differentiate (Oron and Oron 2016; Zhang et al. 2016). Pain and inflammation are reduced (Chow et al. 2009). Blood flow is increased (Samoilova et al. 2008) (possibly as a result of the release of NO (Mitchell and Mack 2013)), which also stimulates lymphatic drainage thereby reducing edema (Dirican et al. 2011).
2.4 Brain specific mechanisms
In addition to the foregoing, there are some PBM tissue mechanisms that are specific to the brain. One of the most important is an increase in cerebral blood flow often reported after transcranial photobiomodualtion (tPBM) (Salgado et al. 2015), leading to increased tissue oxygenation, and more oxidized CCO as measured by NIR spectroscopy (Rojas and Gonzalez-Lima 2013). tPBM has been shown to reduce activated microglia in the brains of TBI mice as measured by IBA1 (ionized calcium-binding adapter molecule-1) expression thus demonstrating reduced neuroinflammation (Khuman et al. 2012). tPBM has been shown to increase neurogenesis (formation of new brain cells derived from neuroprogenitor cells) (Xuan et al. 2014), and synaptogenesis (formation of new connections between existing brain cells) (Xuan et al. 2015) both in TBI mice. Figure 2 shows a graphical representation of a variety of these brain-specific tissue mechanisms.
Brain-specific mechanisms of tPBM
The gene transcription process described in Figure 1 can lead to decreases in neuronal apoptosis and excitotoxicity and lessening of inflammation and reduction of edema due to increased lymphatic flow, which together with protective factors such as antioxidants, will all help to reduce progressive brain damage. Increases in angiogenesis, expression of neurotrophins leading to activation of neural progenitor cells and more cell migration, and increased synaptogenesis may all contribute to the brain repairing itself from damage sustained in the trauma. AUC is area under the curve.
3. Transcranial photobiomodulation
Transcranial PBM is a growing approach to many different brain disorders that may be classified as sudden onset (stroke, TBI, global ischemia), neurodegenerative (Alzheimer's, Parkinson's, dementia), or psychiatric (depression, anxiety, posttraumatic stress disorder)(Hamblin 2016b; Hennessy and Hamblin 2016; Thunshelle and Hamblin 2016). In the following section some issues concerning where the light should be delivered, and the effects of PBM on uninjured mice and humans are addressed.
3.1 Light penetration
Several laboratories working in the field of tissue optics, have investigated the penetration of light of different wavelengths though the scalp and the skull, and to what depths into the parenchyma of the brain this light can penetrate. Answering the question “can light shone on the head sufficiently penetrate to reach the brain?” is difficult. The main reason is that at present it is unclear exactly what threshold of power density is necessary (expressed in mW/cm2) at some depth inside the brain to have a biological effect. There clearly must be a minimum value below which, the light can be delivered for an infinite time without having any effect, but whether this threshold is in the region of μW/cm2 or mW/cm2 is unknown at present.
Haeussinger et al. estimated that the mean penetration depth (5% remaining intensity) of NIR light through the scalp and skull was 23.6 + 0:7 mm (Haeussinger et al. 2011). Other studies have found comparable results with some variations depending on the precise location on the head and the precise wavelength studied (Okada and Delpy 2003; Strangman et al. 2014).
Jagdeo et al. (Jagdeo et al. 2012) used human cadaver heads (skull with intact soft tissue) to measure penetration of 830 nm light, and found that penetration depended on the anatomical region of the skull (0.9% at the temporal region, 2.1% at the frontal region, and 11.7% at the occipital region). Tedord et al. (Tedford et al. 2015) also used human cadaver heads to compare penetration of 660 nm, 808 nm, and 940 nm light. They found that 808 nm light penetrated best, and could reach a depth in the brain of 40–50 mm. Lapchak et al. compared the transmission of 810 nm light through the skulls (no soft tissue) of four different species, and found the mouse skull transmitted 40%, while for rat it was 21%, for rabbit it was 11.3 and for the human skull it was only 4.2% (Lapchak et al. 2015). Pitzschke and colleagues compared penetration of 670 nm and 810 nm light into the brain when delivered by a transcranial or a transphenoidal approach, and found that the best combination was 810 nm delivered transphenoidally (Pitzschke et al. 2015). Yaroslavsky et al. examined light penetration of different wavelengths through different parts of the brain tissue (white brain matter, gray brain matter, cerebellum, and brainstem tissues, pons, thalamus). Best penetration was found with wavelengths between 1000 and 1100 nm (Yaroslavsky et al. 2002). Henderson and Morries found that between 0.45% and 2.90% of 810 nm or 980 nm light penetrated through 3 cm of scalp, skull and brain tissue in ex vivo lamb heads (Henderson and Morries 2015a).
3.2 Local vs systemic effects of light
It is possible that the beneficial effects of PBM on the brain cannot be entirely explained by penetration of light through the scalp and skull into the brain itself, at a sufficient intensity to have an effect on the brain cells. The surface power density that can be safely applied to the head, is limited by heating of the skin. Perceptible heating of the skin starts to be felt when the power density is over about 500 mW/cm2, and can become severe at 1 W/cm2.
There has been one study that explicitly addressed whether direct transcranial PBM or indirect PBM is best for the brain. In a study of PBM for Parkinson's disease in a mouse model, Mitrofanis and colleagues compared the direct delivery of light to the mouse head, and they also covered up the head with aluminum foil so that the light was delivered to the remainder of the mouse body. They found that there was a highly beneficial effect on brain histology with light delivered to the head, but nevertheless there was also a statistically significant although less pronounced benefit (referred to as an “abscopal effect”) when the head was shielded from light. Moreover Oron and co-workers (Farfara et al. 2015) have shown that delivering NIR light to the mouse tibia (using either surface illumination or a fiber optic) resulted in improvements in memory and spatial learning in a transgenic mouse model of Alzheimer's disease. They proposed the mechanism involved PBM stimulating c-kit-positive mesenchymal stem cells (MSCs) that were normally resident in autologous bone marrow. These MSCs were proposed to be able to infiltrate the brain, and clear β-amyloid plaques (Oron and Oron 2016). It should be noted in general that the calvarial bone marrow of the skull contains substantial numbers of stem cells (Iwashita et al. 2003).
3.3 PBM for brain in uninjured animals
Several laboratories have reported that shining light onto the head of uninjured healthy mice or rats can improve various cognitive and emotional parameters. The first study reported that exposure of the middle aged (12 months) CD1 female mice to 1072 nm LED arrays (Michalikova et al. 2008) produced improved performance in a 3D maze compared to sham treated age-matched controls. Gonzalez-Lima and coworkers (Gonzalez-Lima and Barrett 2014) showed that transcranial PBM (9 mW/cm2 with a 660 nm LED array) delivered to rats induced dose-dependent increases in oxygen consumption (5% after 1 J/cm2 and 16% after 5 J/cm2) [113]. They also found that tPBM reduced fear renewal and prevented the reemergence of the extinguished conditioned fear-responses (Rojas et al. 2012).
3.4 PBM for enhancement of brain function in uninjured human volunteers
Gonzalez-Lima et al delivered transcranial PBM (1064 nm laser, 60 J/cm2 at 250 mW/cm2) to the forehead in uninjured human volunteers in a placebo-controlled, randomized study. The goal was to improve performance of cognitive tasks related to the prefrontal cortex, including a psychomotor vigilance task (PVT), a delayed match-to-sample (DMS) memory task, and improved mood as measured by the positive and negative affect schedule (PANAS-X) (Barrett and Gonzalez-Lima 2013). Subsequent studies in uninjured humans showed that tPBM with 1064 nm laser could improve performance in the Wisconsin Card Sorting Task (considered the gold standard test for executive function) (Blanco et al. 2015). They also showed that tPBM to the right forehead (but not the left forehead) could improve attention bias modification (ABM) in humans with depression (Disner et al. 2016).
Salgado et al. applied transcranial LED to enhance cerebral blood flow in healthy elderly women, as measured by transcranial Doppler ultrasound (TCD) of the right and left middle cerebral artery and basilar artery. Twenty-five non-institutionalized elderly women (mean age 72 years), with cognitive status > 24, were assessed using TCD before and after transcranial LED therapy. tPBM (627 nm, 70 mW/cm2, 10 J/cm2) was performed at four points of the frontal and parietal region for 30 s each twice a week for 4 weeks. There was a significant increase in the systolic and diastolic velocity of the left middle cerebral artery (25 and 30%, respectively) and the basilar artery (up to 17 and 25%), as well as a decrease in the pulsatility index and resistance index values of the three cerebral arteries analyzed (Salgado et al. 2015).
3.5 PBM for acute stroke
Transcranial PBM delivered to the head, has been investigated as a possible treatment for acute stroke (Lapchak 2010). Animal models such as rats and rabbits, were first used as laboratory models, and these animals had experimental strokes induced by a variety of methods and were then treated with light (usually 810 nm laser) within 24 h of stroke onset (Lampl 2007). In these studies intervention by tLLLT within 24 h had meaningful beneficial effects.
Treatment of acute stroke in human patients was then addressed in a series of three clinical trials called “Neurothera Effectiveness and Safety Trials” (NEST-1 (Lampl et al. 2007), NEST-2 (Huisa et al. 2013), and NEST-3 (Zivin et al. 2014)). The protocol used an 810 nm laser applied to the shaved head (20 separate points in the 10/20 EEG system) within 24 h of patients suffering an ischemic stroke. The first study, NEST-1, enrolled 120 patients between the ages of 40 to 85 years of age and found a significantly improved outcome (p < 0.05 real vs sham, NIH Stroke Severity Scale) 5 days after a single laser treatment had been administered (Lampl et al. 2007). This significantly improved status was still present 90 days post-stroke in 70% of the PBM patients (but only 51% of the sham patients). The second clinical trial, NEST-2, enrolled 660 patients, aged 40 to 90, who were randomly assigned to one of two groups (331 to PBM, 327 to sham) (Zivin et al. 2009). Significant improvements (p < 0.04) were found in the moderate and moderate-severe (but not for the severe) stroke patients. The last clinical trial, NEST-3, was planned for 1000 patients enrolled, but the study was prematurely terminated by the DSMB for futility (an expected lack of statistical significance) (Lapchak and Boitano 2016). Many commentators have asked how tPBM could work so well in the first trial, yet fail in the third trial. Insufficient light penetration, too long an interval between stroke onset and PBM, inappropriate stroke severity measurement scale, use of only one single tPBM treatment, and failure to illuminate different specific areas of the brain for individual patients, have all been suggested as contributory reasons (Hamblin 2016b). It is undoubtedly the case that the failure of NEST-3 has cast a cloud over the whole application of PBMT for TBI as well as for stroke. Many commentators have asked “Why are you testing PBMT for TBI, if it has been shown not to work for stroke?” The failure of the investigators not to take into account the anatomical location of the stroke (and also whether it was deep or superficial) was also likely to have played a role in the failure of NEST-3. It is logical that light should be applied to the same side of the head where the lesion was located, not both sides of the head (Naeser et al. 2012). In my opinion the use of a single application of PBMT also bore some of the responsibility. Although a single application of PBM to the head works very well for experimental animals (mice, rats, rabbits) who have suffered a stroke or a TBI, the same may not apply to humans.
4. Animal studies of PBM in acute TBI models
4.1 Studies from the Oron laboratory
Oron's group was the first (Oron et al. 2007) to demonstrate that a single exposure of the head of a mouse a few hours after creation of a TBI lesion using a NIR laser (808 nm) could improve neurological performance and reduce the size of the brain lesion. A weight-drop device was used to induce a closed-head TBI in the mice. An 808 nm diode laser with two energy densities calculated at the surface of the brain (1.2-2.4 J/cm2 delivered by 2 minutes of irradiation with 200mW laser power to the scalp) was delivered to the head 4 hours after TBI was induced. Neurobehavioral function was assessed by the neurological severity score (NSS). There was no significant difference between the control and laser-treated group in NSS between the power densities (10 vs 20 mW/cm2), and no significant difference at early time points (24 and 48 hours) post TBI. However, there was a significant improvement (27% lower NSS score) in the PBM group at times between 5 days and 4 weeks. The laser treated group also showed a smaller loss of cortical tissue than the sham group (Oron et al.). In another study (Oron et al. 2012) they varied the pulse parameters (CW, 100Hz, or 600Hz) and tested whether the tPBM was equally effective when delivered at 4, 6, or 8 hours post-TBI. They first established that a calculated dose to the cortical surface of 1.2 J/cm2 of 808nm laser at 200mW applied to the head, was more effective when delivered at 6 hours post TBI than at 8 hours. They then selected an even shorter time post-TBI (4 hours) and compared CW with 100Hz and 600Hz. At 56 days, more mice in the 100Hz group (compared to the CW and 600 Hz groups) had fully recovered. The 600Hz group had lower NSS scores than the CW and 100Hz groups up to 20 days. Magnetic resonance imaging (MRI) analysis demonstrated significantly smaller lesion volumes in PBM-treated mice compared to controls.
4.2 Studies from the Hamblin laboratory
Wu et al. (Wu et al. 2012) first explored the effect of varying the laser wavelengths of PBM had on closed-head TBI in mice. Mice were randomly assigned to a PBM treatment group with a particular wavelength, or to a sham treatment group as a control. Closed-head injury (CHI) was induced via a weight- drop apparatus. To analyze the severity of the TBI, the neurological severity score (NSS) was measured and recorded. The injured mice were then treated with varying wavelengths of laser light (665, 730, 810 or 980 nm) at an energy density of 36 J/cm2 directed onto the scalp at 4 hours post-TBI. The 665 nm and 810 nm laser groups showed significant improvement in NSS when compared to the control group between days 5 to 28. By contrast, the 730 nm and 980 nm laser groups did not show any significant improvement in NSS (Wu et al. 2012) (Figure 3). The tissue chromophore cytochrome c oxidase (CCO) is proposed to be responsible for the underlying photon absorption process that underlies many PBM effects. CCO has absorption bands around 665 nm and 810 nm while it has a low absorption region at the wavelength of 730 nm (Karu et al.). It should be noted that this particular study (Wu et al. 2012) found that the 980 nm did not produce the same positive effects as the 665 nm and 810 nm wavelengths did; nevertheless previous studies did find that the 980 nm wavelength was an active one for PBM (Anders et al. 2014). Wu et al. suggested that these dissimilar results may be due to differences in the energy density, irradiance etc. between the other studies and the Wu study (Wu et al. 2012). In particular a much lower dose of 980 nm might have been effective had it been tested (Wang et al. 2016). Ando et al. (Ando et al. 2011) next used the 810 nm wavelength produced by a Ga-Al-As diode laser delivered at parameters used in the Wu study, and varied the pulse modes of the laser. These modes consisted of either pulsed wave at 10 Hz or at 100 Hz (50% duty cycle) or continuous wave laser. They used a different mouse model of TBI induced with a controlled cortical impact device directly inflicting a lesion on the cortex via an open craniotomy. A single treatment with a power density of 50 mW/m2 and an energy density of 36 J/cm2 (duration of 12 minutes) was given via tLLLT to the closed head in mice at 4 hours post CCI. At 48 hours to 28 days post TBI, all laser treated groups had significant decreases in the measured neurological severity score (NSS) when compared to the controls. Although all laser treated groups had similar NSS improvement rates up to day 7, the PW 10 Hz group began to show even greater improvement beyond this point as seen in Figure 4. At day 28, the forced swim test for depression and anxiety was used and showed a significant decrease in the immobility time for the PW 10 Hz group. In the tail suspension test, which measures depression and anxiety, there was also a significant decrease in the immobility time at day 28, and also at day 1, in the PW 10 Hz group.
Effect of different laser wavelengths of tPBM in closed-head TBI in mice
(A) Sham-treated control versus 665 nm laser. (B) Sham-treated control versus 730 nm laser. (C) Sham-treated control versus 810 nm laser. (D) Sham-treated control versus 980 nm laser. Points are means of 8–12 mice and bars are SD. *P < 0.05; **P < 0.01; ***P < 0.001 (one-way ANOVA). Reprinted with permission from (Wu et al. 2012)
Effects of pulsing in tPBM for CCI-TBI in mice
(A) Time course of neurological severity score (NSS) of mice with TBI receiving either control (no laser-treatment), or 810 nm laser (36 J/cm2 delivered at 50 mW/cm2 with a spot size of 0.78 cm2 in either CW, PW 10 Hz or PW 100 Hz modes. Results are expressed as mean +/- S.E.M ***P < 0.001 vs. the other conditions. (B) Mean areas under the NSS-time curves in the two-dimensional coordinate system over the 28-day study for the 4 groups of mice. Results are means +/- SD (n = 10). Reprinted from (Ando et al. 2011) (open access).
Studies using immunofluorescence staining of sections cut from mouse brains showed that tPBM increased neuroprogenitor cells (incorporating BrdU) in the dentate gyrus (DG) of the hippocampus and the subventricular zone (SVZ) at 7 days after the treatment (Xuan et al. 2014). The neurotrophin known as brain derived neurotrophic factor (BDNF) was also increased in the DG and SVZ at 7 days, while the protein marker (synapsin-1) for synaptogenesis and neuroplasticity was increased in the cortex at 28 days but not in the DG, SVZ or in any location at 7 days (Xuan et al. 2015). Learning and memory as measured by the Morris water maze was also improved by tPBM (Xuan et al. 2014).
4.3 Studies from the Wu laboratory
Zhang et al. (Zhang et al. 2014) first showed that secondary brain injury occurred to a worse degree in mice that had been genetically engineered to lack “Immediate Early Response” gene X-1 (IEX-1). When these mice were exposed to a gentle head impact (thought to closely resemble mild TBI in humans) they had a worse NSS than uninjured mice with the same TBI. Exposure of IEX-1 knockout mice to PBM (150 mW/cm2, 4 min, and 36 J/cm2) delivered at 4 hours post injury, restored the NSS to almost baseline levels, suppressed proinflammatory cytokine expression of interleukin (IL)-Iβ and IL-6, but upregulated TNF-α. The original lack of IEX-1 decreased ATP production, but exposing the injured brain to LLLT elevated ATP production back to near normal levels.
Dong et al. (Dong et al. 2015) asked whether the beneficial effects of PBM on TBI in mice could be enhanced by combining PBM with administration of metabolic substrates such as pyruvate and/or lactate. The goal was to even further improve mitochondrial function in the brain. This combinatorial treatment was able to reverse memory and learning deficits in TBI injured mice back to normal levels as well as leaving the hippocampal region completely protected from tissue loss; a stark contrast to control TBI mice that exhibited severe tissue loss from secondary brain injury.
4.4 Studies from the Whalen laboratory
Khuman et al (Khuman et al. 2012) delivered PBM (800nm) either directly to the injured brain tissue (through the craniotomy) or transcranially in mice beginning 60-80 min after CCI TBI. At a dose of 60J/cm2 (500mW/cm2) the mice showed increased performance in the Morris water maze (latency to the hidden platform, p<0.05, and probe trial, p<0.01) compared to non-treated controls. When PBM was delivered via open craniotomy there was reduced microgliosis at 48h (IbA-1+ cells, p<0.05). Little or no effect of tPBM on post-injury cognitive function was observed using lower or higher doses, a 4-h administration time point or 60J/cm2 at 7-days post-TBI.
4.5 Studies from the Whelan laboratory
Quirk et al (Quirk et al. 2012) studied Sprague-Dawley rats who had received a severe CCI TBI and were divided into three groups: real TBI, sham surgery, and anesthetization only. Each group received either real or sham PBM consisting of 670nm LED treatments of 15J/cm2, 50mW/cm2, 5min, given two times per day for 3 days (chemical analysis) or 10 days (behavioral analysis using a TruScan nose-poke device). Significant differences in task entries, repeat entries, and task errors were seen in the TBI rats treated with PBM vs untreated TBI mice, and in sham surgery mice treated with PBM vs untreated sham surgery mice. A statistically significant decrease was found in the pro-apoptotic marker Bax, and increases in the anti-apoptotic marker Bcl-2 and reduced glutathione (GSH) levels in tPBM TBI mice.
4.6 Studies from the Marques laboratory
Moreira et al used a different model of TBI (Moreira et al. 2009). Wistar rats received a craniotomy and a copper probe cooled in liquid nitrogen was applied to the surface of the brain to create a standardized cryogenic injury. They treated the rats with either a 780nm or 660nm laser at one of two different doses (3J/cm2 or 5J/cm2) twice (once immediately after the injury and again 3 hours later). Rats were sacrificed 6h and 24h after the injury. The 780nm laser was better at reducing levels of pro-inflammatory cytokines (TNFα, IL1β, IL6) particularly at early timepoints (Moreira et al. 2009). In a follow-up study using 3 J/cm2 (Moreira et al. 2011) these workers reported on the healing of the injuries in these rats at timepoints 6h, 1, 7 and 14 days after the last irradiation. Cryogenic injury created focal lesions in the cortex characterized by necrosis, edema, hemorrhage and inflammatory infiltrate. The most striking findings were: PBM-treated lesions showed less tissue loss than control lesions at 6h. During the first 24h the amount of viable neurons was significantly higher in the PBM groups. PBM reduced the amount of GFAP (glial fibrillary acidic protein, a marker of astrogliosis) and the numbers of leukocytes and lymphocytes, thus demonstrating its anti-inflammatory effect.
5. Patients with chronic TBI
The majority of studies of PBM for TBI in laboratory animals have been conducted in the acute setting, while the majority of human studies of PBM for TBI have been conducted in patients who have suffered head injuries at various times in the past (sometimes quite a long time ago).
5.1 Naeser case reports
In 2011 Naeser, Saltmarche et al., published the first report describing two chronic, TBI cases treated with tPBM (Naeser et al. 2011). A 500 mW CW LED source (mixture of 660 nm red and 870 nm NIR LEDs) with a power density of 22.2 mW/cm2 (area of 22.48 cm2), was applied all over the head, for 10 minutes at each placement location (13.3 J/cm2). In the first case study the patient reported that she could concentrate on tasks for a longer period of time (the time able to work at a computer increased from 20 minutes to 3 hours). She had a better ability to remember what she read, decreased sensitivity when receiving haircuts in the spots where PBM was applied, and improved mathematical skills after undergoing PBM. The second patient had statistically significant improvements compared to prior neuropsychological tests after 9 months of treatment. The patient had a 2 standard deviation (SD) increase on tests of inhibition and inhibition accuracy (9th percentile to 63rd percentile on the Stroop test for executive function and a 1 SD increase on the Wechsler Memory scale test for the logical memory test (83rd percentile to 99th percentile) (Naeser et al. 2011).
5.2 Naeser case series
Naeser et al then went on to report a case series containing a further eleven patients (Naeser et al. 2014). This was an open protocol study that examined whether scalp application of red and NIR LED could improve cognition in patients with chronic, mild TBI (mTBI). This study enrolled 11 participants ranging in age from 26 to 62 years (6 males, 5 females) who suffered from persistent cognitive dysfunction after mTBI. The injuries in the participants had been caused by motor vehicle accidents, sports related events and for one participant, an improvised explosive device (IED) blast. tPBM consisted of 18 sessions (Monday, Wednesday, and Friday for 6 weeks) and was started anywhere from 10 months to 8 years post-TBI. A total of 11 LED cluster heads (5.25 cm in diameter, 500 mW, 22.2 mW/cm2, 13 J/cm2) were applied for 10 minutes per set (5 or 6 LED placements per set, Set A and then Set B, in each session). Neuropsychological testing was performed pre-LED application and 1 week, 1 month and 2 months after the final treatment. They found that there was a significant positive linear trend for the Stroop Test for executive function, in trial 3 inhibition (p = 0.004); Stroop, trial 4 inhibition switching (p = 0.003); California Verbal Learning Test (CVLT)-II, total trials 1-5 (p = 0.003); CVLT-II, long delay free recall (p = 0.006). Improved sleep and fewer post-traumatic stress disorder (PTSD) symptoms, if present beforehand, were observed after treatment. Participants and family members also reported better social function and a better ability to perform interpersonal and occupational activities. Although these results were significant, the authors suggested that further placebo-controlled studies would be needed to ensure the reliability of this approach (Naeser et al. 2014).
Naeser has proposed (Naeser et al. 2016; Naeser et al. 2014) that specific scalp placements of the LED cluster heads may affect specific cortical nodes in the intrinsic networks of the brain, such as the default mode network (DMN), the salience network (SN), and the central executive network (CEN). These intrinsic networks are often dysregulated after TBI (Sharp et al. 2014). Naeser proposed that the specific areas of the head to receive light, to target cortical nodes in these networks were as follows:
-
For the DMN, placement of the LED cluster head on the midline of face, centered on the upper forehead and the front hairline, targeted the left and right mesial prefrontal cortex; and on a midline, scalp location half-way between the occipital protuberance and the vertex of the head, targeted the precuneus; and on left and right LED placements superior to the tip of each ear and posterior to each ear, targeted the inferior parietal cortex/angular gyrus areas.
-
For the SN, placement of LED cluster heads on the left and right temple areas, to target the anterior insula (but due to depth of insula, unknown if the photons reached the target); midline of the vertex of the head, to target the left and right presupplementary motor areas; and the LED cluster head placed on the midline of face, centered on the upper forehead and the front hairline, also targeted the left and right dorsal anterior cingulate cortex.
-
For the CEN, left and right scalp LED placements immediately posterior to the front hairline (on a line directly superior from the pupils of the eyes), targeted the dorso-lateral prefrontal cortex areas; and the left and right LED placements superior to the tip of each ear and posterior to each ear, also targeted the posterolateral inferior parietal cortex/angular gyrus areas (also treated as part of the DMN).
Further studies from Naeser and colleagues (Naeser et al. 2016) tested an intranasal LED (iLED) device. Two small iLEDs (one red and the other NIR) were clipped into each nostril and used at the same time for 25 min. The parameters were as follows: red, 633nm, 8mW CW, 1 cm2, energy density 12 J/cm2 (25 min); NIR 810nm, 14.2mW, pulsed 10Hz, 1cm2, 21.3J/cm2. The first mTBI participant (24-year old female) who had sustained four sports-related concussions (two during snowboarding and two during field hockey), received iLED PBM three times per week for 6 weeks. Significant improvements were observed in tasks measuring executive function and verbal memory as well as attention and verbal fluency. At 1 week after the 18th iLED treatment, the average total time asleep had increased by 61 min per night and her sleep efficiency (total sleep time divided by total time in bed) had increased by 11%. At 12 weeks after the last iLED treatment, she was able to discontinue all sleep medications that she had previously been using. The second, mTBI participant who received the intranasal only, LED treatment series is a 49 Yr. M (non-Veteran) who sustained mTBI in a MVA, 30 years prior to receiving the intranasal LED treatment series. He showed significant improvement on the Controlled Oral Word Association-FAS Test post- the iLED treatment series, improving by +1.3 SD and +1.5 SD at 1 and 2 months post- the 18th iLED treatment. His sleep data indicated he was already a good sleeper, at entry.
5.3 Bogdanova and Naeser studies
Bogdanova reported (Bogdanova et al. 2014) a case report of two patients (1 female) with moderate TBI (medical records and clinical evaluation) and persistent cognitive dysfunction (as measured by neuropsychological tests of executive function and memory). Patients received 18 sessions of transcranial LED therapy (3×/week for 6 weeks) using the mixed red/NIR cluster described above (Naeser et al. 2011).
Standardized neuropsychological tests for executive function, memory, depression, PTSD and sleep measures (PSQI, actigraphy) were administered to participants pre-(T1), mid-(T2), and one week (T3) post-PBM treatment. Both PBM treated cases (P1 and P2) showed marked improvement in sleep (actigraphy total sleep) 1 week post-LED treatment (T3), as compared to pre-treatment (T1). P1 also improved in executive function, verbal memory, and sleep efficiency; while P2 significantly improved on measures of PTSD (PCL-M) and depression. No adverse events were reported.
5.4 Studies from Henderson and Morries
Henderson and Morries (Henderson and Morries 2015b) used a high-power NIR laser (10-15 W at 810 and 980 nm) and applied it to the head to treat a patient with moderate TBI. The patient received 20 NIR applications over a 2-month period. They carried out anatomical magnetic resonance imaging (MRI) and perfusion single-photon emission computed tomography (SPECT). The patient showed decreased depression, anxiety, headache, and insomnia, whereas cognition and quality of life improved, accompanied by changes in the SPECT imaging.
They next reported (Morries et al. 2015) a series of ten patients with chronic TBI (average time since injury 9.3 years) where each patient received ten treatments over the course of 2 months using a high-power NIR laser (13.2 W/0.89 cm2 equivalent to 14.6 W/cm2 at 810nm; or 9 W/0.89 cm2 equivalent to 10.11 W/cm2 at 980nm). A continuous sweeping motion over the forehead was utilized to minimize skin heating and cover a larger area. Skin temperature increased no more than 3°C. Overall symptoms of headache, sleep disturbance, cognition, mood dysregulation, anxiety, and irritability improved. Symptoms were monitored by depression scales and a novel patient diary system specifically designed for this study. These authors have proposed that high power lasers are preferable for tPBM treatments because the photons can better reach the brain (Henderson and Morries 2015a).
5.5 Case study from Nawashiro
Nawashiro et al (Nawashiro et al. 2012) treated a single patient who had suffered a severe TBI. The patient survived but was left in a persistent vegetative state for 8 months after the accident. He showed no spontaneous movement of limbs and a CT scan of the head 8 months after the accident showed a focal low-density area in the right frontal lobe. The device had 23 individual 850nm LEDs (13mW each; total power 299mW, total area 57cm2). A treatment time of 30 min per session delivered 20.5 J/cm2 over the left and right forehead areas repeated twice daily (6h apart), for 73 days. Five days after beginning the PBM (after 10 treatments), the patient began to spontaneously move his left arm and hand, which had not occurred during the previous 8 months. Single-photon emission computed tomography with N-isopropyl-[123I]p-iodoamphetamine (IMP-SPECT) was performed twice. The IMP-SPECT scans showed a focal increase (20% higher) in cerebral blood flow in the uninjured left anterior frontal lobe 30 min after the last (146th) PBM treatment, compared to before PBM began.
6. Conclusion and future prospects
As was mentioned above, one of the most important questions to be answered when contemplating clinical treatment of TBI patients with tPBM, is what is the best time to administer the treatment? All the available reports of studies using PBM in laboratory animal models of TBI and stroke, and also in patients treated for stroke, have been in the acute phase where the overall goal of the intervention can be best described as neuroprotection. Not only that but there are several reports (Lapchak et al. 2007; Oron et al. 2012) that PBM for both TBI and stroke is most effective when it is delivered as soon as possible after the actual event (head impact or ischemic stroke). The protocols for the series of NEST clinical trials specified that patients should be treated with PBM within 24 hours of the stroke occurring. By contrast, all the clinical trials of PBM for patients with TBI, that have so far been carried out, have been with chronic TBI, after varying periods of time having elapsed after the original head injury, sometimes as long as 8 years. Although it would be generally supposed that tPBM would be effective when delivered to acute TBI patients, this has not yet been actually tested. If tPBM were to be used for acute TBI patients, then presumably the PBM should be delivered perhaps beginning at 4 to 6 hours post-TBI, for a limited number of times after the injury; perhaps once a day for 7 days?
The dosimetry and optimum delivery apparatus of tPBM is still uncertain. Although there is some consensus that wavelengths in the region of 800-900nm will penetrate the scalp and skull, other workers have used longer NIR wavelengths, 980nm, 1064nm, or 1072nm. Pulsing or CW is another unresolved question. The exact locations on the head that should receive the light are still unknown. Naeser has proposed (Naeser et al. 2016) some interesting considerations regarding the scalp placements of the tLEDS, and their effect on various intrinsic cortical networks of the brain. Targeted LED placements could promote better neuromodulation (activation/deactivation) in specific cortical nodes. It is possible that communication between nodes within one single network, and/or across networks could be improved. Moreover preliminary data indicate that intranasal, red plus near-infrared LEDs can also benefit TBI patients, although the degree to which light incident on the nasal mucosa, and possibly delivered transsphenoidally (Pitzschke et al. 2015) can penetrate directly into the brain, remains to be determined.
An advantage of intranasal and/or transcranial LED PBM therapy is that it can be performed in the home, for long-term use (Naeser et al. 2011). Also, 5 chronic, mild to moderately-severe dementia cases recently showed significant improvement on the Mini-Mental State Examination (p<0.003), and on the Alzheimer's Disease Assessment Scale-Cognitive subscale (p<0.023) after 12 weeks of daily, at-home, intranasal, near-infrared LED PBM treatments (810nm, pulsed at 10 Hz), and once-a-week in-office, tLED treatments applied to the cortical nodes of the Default Mode Network (Saltmarche et al. 2017). Anecdotally, there was also improved sleep, fewer angry outbursts, and less wandering. When all LED treatments were withdrawn after 12 weeks of active LED PBM treatment, there was precipitous decline in cognition and behavior. Thus, at-home, long-term use of iLED plus tLED PBM offers a potential therapy to mitigate the sequelae of Alzheimer's disease and possibly other neurodegenerative disorders, as well as TBI and stroke.
One highly distressing aspect of TBI symptomatology that has not so far been addressed by PBM, is that of post-traumatic epilepsy (PTE). TBI is the most significant cause of symptomatic epilepsy in people from 15 to 24 years of age. The frontal and temporal lobes are the most frequently affected regions, but imaging (MRI) often fails to show the precise cause. During PTE seizures there is an abnormal electrical discharge in the brain, with staring and unresponsiveness, stiffening or shaking of the body, legs, arms or head; strange sounds, tastes, visual images, feelings or smells; inability to speak or understand, etc (Cotter et al. 2017). Epilepsy has traditionally been considered to be a contra-indication for PBMT (Navratil and Kymplova 2002). However the knowledge that has recently been gained concerning the beneficial effects of PBMT on the damaged brain, suggests that this view may need to be critically revisited.
Moreover there is also potential of tPBM to treat a wide range of brain disorders only loosely associated with TBI, including Parkinson's disease (Purushothuman et al. 2013), depression, anxiety, post-traumatic stress disorder, autism spectrum disorder and so on (Hamblin 2016b).
The ongoing and accelerating clinical research efforts in testing PBM for TBI, are expected to lead to the answering of many of these questions in the coming years.
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5803455/
Shining light on the head: Photobiomodulation for brain disorders
Michael R. Hamblin - 10.1016/j.bbacli.2016.09.002 (Publication) 4466This is 27 pages of independent analysis of how photobiomodulation effects the brain. Covers wavelengths, dosage, pulsing, depths and underlying reactions. Amazing.
Photobiomodulation (PBM) describes the use of red or near-infrared light to stimulate, heal, regenerate, and protect tissue that has either been injured, is degenerating, or else is at risk of dying. One of the organ systems of the human body that is most necessary to life, and whose optimum functioning is most worried about by humankind in general, is the brain. The brain suffers from many different disorders that can be classified into three broad groupings: traumatic events (stroke, traumatic brain injury, and global ischemia), degenerative diseases (dementia, Alzheimer's and Parkinson's), and psychiatric disorders (depression, anxiety, post traumatic stress disorder). There is some evidence that all these seemingly diverse conditions can be beneficially affected by applying light to the head. There is even the possibility that PBM could be used for cognitive enhancement in normal healthy people. In this transcranial PBM (tPBM) application, near-infrared (NIR) light is often applied to the forehead because of the better penetration (no hair, longer wavelength). Some workers have used lasers, but recently the introduction of inexpensive light emitting diode (LED) arrays has allowed the development of light emitting helmets or “brain caps”. This review will cover the mechanisms of action of photobiomodulation to the brain, and summarize some of the key pre-clinical studies and clinical trials that have been undertaken for diverse brain disorders.
Keywords: Photobiomodulation, Low level laser (light) therapy, Ischemic stroke, Traumatic brain injury, Alzheimer's disease, Parkinson's disease, Major depression, Cognitive enhancement
Graphical abstract

1. Introduction
Photobiomodulation (PBM) as it is known today (the beneficial health benefits of light therapy had been known for some time before), was accidently discovered in 1967, when Endre Mester from Hungary attempted to repeat an experiment recently published by McGuff in Boston, USA [1]. McGuff had used a beam from the recently discovered ruby laser [2], to destroy a cancerous tumor that had been experimentally implanted into a laboratory rat. However (unbeknownst to Mester) the ruby laser that had been built for him, was only a tiny fraction of the power of the laser that had previously been used by McGuff. However, instead of curing the experimental tumors with his low-powered laser, Mester succeeded in stimulating hair regrowth and wound healing in the rats, in the sites where the tumors had been implanted [3], [4]. This discovery led to a series of papers describing what Mester called “laser biostimulation”, and soon became known as “low level laser therapy” (LLLT) [5], [6], [7].
LLLT was initially primarily studied for stimulation of wound healing, and reduction of pain and inflammation in various orthopedic conditions such as tendonitis, neck pain, and carpal tunnel syndrome [8]. The advent of light emitting diodes (LED) led to LLLT being renamed as “low level light therapy”, as it became more accepted that the use of coherent lasers was not absolutely necessary, and a second renaming occurred recently [9] when the term PBM was adopted due to uncertainties in the exact meaning of “low level”.
2. Mechanisms of action of photobiomodulation
2.1. Mitochondria and cytochrome c oxidase
The most well studied mechanism of action of PBM centers around cytochrome c oxidase (CCO), which is unit four of the mitochondrial respiratory chain, responsible for the final reduction of oxygen to water using the electrons generated from glucose metabolism [10]. The theory is that CCO enzyme activity may be inhibited by nitric oxide (NO) (especially in hypoxic or damaged cells). This inhibitory NO can be dissociated by photons of light that are absorbed by CCO (which contains two heme and two copper centers with different absorption spectra) [11]. These absorption peaks are mainly in the red (600–700 nm) and near-infrared (760–940 nm) spectral regions. When NO is dissociated, the mitochondrial membrane potential is increased, more oxygen is consumed, more glucose is metabolized and more ATP is produced by the mitochondria.
2.2. Reactive oxygen species, nitric oxide, blood flow
It has been shown that there is a brief increase in reactive oxygen species (ROS) produced in the mitochondria when they absorb the photons delivered during PBM. The idea is that this burst of ROS may trigger some mitochondrial signaling pathways leading to cytoprotective, anti-oxidant and anti-apoptotic effects in the cells [12]. The NO that is released by photodissociation acts as a vasodilator as well as a dilator of lymphatic flow. Moreover NO is also a potent signaling molecule and can activate a number of beneficial cellular pathways [13]. Fig. 2 illustrates these mechanisms.
Tissue specific processes that occur after PBM and benefit a range of brain disorders. BDNF, brain-derived neurotrophic factor; LLLT, low level light therapy; NGF, nerve growth factor; NT-3, neurotrophin 3; PBM, photobiomodulation; SOD, superoxide dismutase. ...
2.3. Light sensitive ion channels and calcium
It is quite clear that there must be some other type of photoacceptor, in addition to CCO, as is clearly demonstrated by the fact that wavelengths substantially longer than the red/NIR wavelengths discussed above, can also produce beneficial effects is some biological scenarios. Wavelengths such as 980 nm [14], [15], 1064 nm laser [16], and 1072 nm LED [17], and even broad band IR light [18] have all been reported to carry out PBM type effects. Although the photoacceptor for these wavelengths has by no means been conclusively identified, the leading hypothesis is that it is primarily water (perhaps nanostructured water) located in heat or light sensitive ion channels. Clear changes in intracellular calcium can be observed, that could be explained by light-mediated opening of calcium ion channels, such as members of the transient receptor potential (TRP) super-family [19]. TRP describes a large family of ion channels typified by TRPV1, recently identified as the biological receptor for capsaicin (the active ingredient in hot chili peppers) [20]. The biological roles of TRP channels are multifarious, but many TRP channels are involved in heat sensing and thermoregulation [21].
2.4. Signaling mediators and activation of transcription factors
Most authors suggest that the beneficial effects of tPBM on the brain can be explained by increases in cerebral blood flow, greater oxygen availability and oxygen consumption, improved ATP production and mitochondrial activity [22], [23], [24]. However there are many reports that a brief exposure to light (especially in the case of experimental animals that have suffered some kind of acute injury or traumatic insult) can have effects lasting days, weeks or even months [25]. This long-lasting effect of light can only be explained by activation of signaling pathways and transcription factors that cause changes in protein expression that last for some considerable time. The effects of PBM on stimulating mitochondrial activity and blood flow is of itself, unlikely to explain long-lasting effects. A recent review listed no less than fourteen different transcription factors and signaling mediators, that have been reported to be activated after light exposure [10].
Fig. 1 illustrates two of the most important molecular photoreceptors or chromophores (cytochrome c oxidase and heat-gated ion channels) inside neuronal cells that absorb photons that penetrate into the brain. The signaling pathways and activation of transcription factors lead to the eventual effects of PBM in the brain.
Molecular and intracellular mechanisms of transcranial low level laser (light) or photobiomodulation. AP1, activator protein 1; ATP, adenosine triphosphate; Ca2 +, calcium ions; cAMP, cyclic adenosine monophosphate; NF-kB, nuclear factor kappa ...
Fig. 2 illustrates some more tissue specific mechanisms that lead on from the initial photon absorption effects explained in Fig. 1. A wide variety of processes can occur that can benefit a correspondingly wide range of brain disorders. These processes can be divided into short-term stimulation (ATP, blood flow, lymphatic flow, cerebral oxygenation, less edema). Another group of processes center around neuroprotection (upregulation of anti-apoptotic proteins, less excitotoxity, more antioxidants, less inflammation). Finally a group of processes that can be grouped under “help the brain to repair itself” (neurotrophins, neurogenesis and synaptogenesis).
2.5. Biphasic dose response and effect of coherence
The biphasic dose response (otherwise known as hormesis, and reviewed extensively by Calabrese et al. [26]) is a fundamental biological law describing how different biological systems can be activated or stimulated by low doses of any physical insult or chemical substance, no matter how toxic or damaging this insult may be in large doses. The most well studied example of hormesis is that of ionizing radiation, where protective mechanisms are induced by very low exposures, that can not only protect against subsequent large doses of ionizing radiation, but can even have beneficial effects against diseases such as cancer using whole body irradiation [27].
There are many reports of PBM following a biphasic dose response (sometimes called obeying the Arndt-Schulz curve [28], [29]. A low dose of light is beneficial, but raising the dose produces progressively less benefit until eventually a damaging effect can be produced at very high light [30]. It is often said in this context that “more does not mean more”.
Another question that arises in the field of PBM is whether the coherent monochromatic lasers that were used in the original discovery of the effect, and whose use continued for many years, are superior to the rather recent introduction of LEDs, that are non-coherent and have a wider band-spread (generally 30 nm full-width half-maximum). Although there are one or two authors who continue to believe that coherent lasers are superior [31], most commentators feel that other parameters such as wavelength, power density, energy density and total energy are the most important determinants of efficacy [8].
3. Tissue optics, direct versus systemic effects, light sources
3.1. Light penetration into the brain
Due to the growing interest in PBM of the brain, several tissue optics laboratories have investigated the penetration of light of different wavelengths through the scalp and the skull, and to what depths into the brain this light can penetrate. This is an intriguing question to consider, because at present it is unclear exactly what threshold of power density in mW/cm2 is required in the b5rain to have a biological effect. There clearly must be a minimum value below which the light can be delivered for an infinite time without doing anything, but whether this is in the region of μW/cm2 or mW/cm2 is unknown at present.
Functional near-infrared spectroscopy (fNIRS) using 700–900 nm light has been established as a brain imaging technique that can be compared to functional magnetic resonance imaging (fMRI) [32]. Haeussinger et al. estimated that the mean penetration depth (5% remaining intensity) of NIR light through the scalp and skull was 23:6 + 0:7 mm [33]. Other studies have found comparable results with variations depending on the precise location on the head and wavelength [34], [35].
Jagdeo et al. [36] used human cadaver heads (skull with intact soft tissue) to measure penetration of 830 nm light, and found that penetration depended on the anatomical region of the skull (0.9% at the temporal region, 2.1% at the frontal region, and 11.7% at the occipital region). Red light (633 nm) hardly penetrated at all. Tedord et al. [37] also used human cadaver heads to compare penetration of 660 nm, 808 nm, and 940 nm light. They found that 808 nm light was best and could reach a depth in the brain of 40–50 mm. Lapchak et al. compared the transmission of 810 nm light through the skulls of four different species, and found mouse transmitted 40%, while for rat it was 21%, rabbit it was 11.3 and for human skulls it was only 4.2% [38]. Pitzschke and colleagues compared penetration of 670 nm and 810 nm light into the brain when delivered by a transcranial or a transphenoidal approach, and found that the best combination was 810 nm delivered transphenoidally [39]. In a subsequent study these authors compared the effects of storage and processing (frozen or formalin-fixed) on the tissue optical properties of rabbit heads [40]. Yaroslavsky et al. examined light penetration of different wavelengths through different parts of the brain tissue (white brain matter, gray brain matter, cerebellum, and brainstem tissues, pons, thalamus). Best penetration was found with wavelengths between 1000 and 1100 nm [41].
Henderson and Morries found that between 0.45% and 2.90% of 810 nm or 980 nm light penetrated through 3 cm of scalp, skull and brain tissue in ex vivo lamb heads [42].
3.2. Systemic effects
It is in fact very likely that the beneficial effects of PBM on the brain cannot be entirely explained by penetration of photons through the scalp and skull into the brain itself. There have been some studies that have explicitly addressed this exact issue. In a study of PBM for Parkinson's disease in a mouse model [43]. Mitrofanis and colleagues compared delivering light to the mouse head, and also covered up the head with aluminum foil so that they delivered light to the remainder of the mouse body. They found that there was a highly beneficial effect on neurocognitive behavior with irradiation to the head, but nevertheless there was also a statistically significant (although less pronounced benefit, referred to by these authors as an ‘abscopal effect”) when the head was shielded from light [44]. Moreover Oron and co-workers [45] have shown that delivering NIR light to the mouse tibia (using either surface illumination or a fiber optic) resulted in improvement in a transgenic mouse model of Alzheimer's disease (AD). Light was delivered weekly for 2 months, starting at 4 months of age (progressive stage of AD). They showed improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. They proposed that the mechanism of this effect was to stimulate c-kit-positive mesenchymal stem cells (MSCs) in autologous bone marrow (BM) to enhance the capacity of MSCs to infiltrate the brain, and clear β-amyloid plaques [46]. It should be noted that the calvarial bone marrow of the skull contains substantial numbers of stem cells [47].
3.3. Laser acupuncture
Laser acupuncture is often used as an alternative or as an addition to traditional Chinese acupuncture using needles [48]. Many of the applications of laser acupuncture have been for conditions that affect the brain [49] such as Alzheimer's disease [50] and autism [51] that have all been investigated in animal models. Moreover laser acupuncture has been tested clinically [52].
3.4. Light sources
A wide array of different light sources (lasers and LEDs) have been employed for tPBM. One of the most controversial questions which remains to be conclusively settled, is whether a coherent monochromatic laser is superior to non-coherent LEDs typically having a 30 nm band-pass (full width half maximum). Although wavelengths in the NIR region (800–1100 nm) have been the most often used, red wavelengths have sometimes been used either alone, or in combination with NIR. Power levels have also varied markedly from Class IV lasers with total power outputs in the region of 10 W [53], to lasers with more modest power levels (circa 1 W). LEDs can also have widely varying total power levels depending on the size of the array and the number and power of the individual diodes. Power densities can also vary quite substantially from the Photothera laser [54] and other class IV lasers , which required active cooling (~ 700 mW/cm2) to LEDs in the region of 10–30 mW/cm2.
3.5. Usefulness of animal models when testing tPBM for brain disorders
One question that is always asked in biomedical research, is how closely do the laboratory models of disease (which are usually mice or rats) mimic the human disease for which new treatments are being sought? This is no less critical a question when the areas being studied include brain disorders and neurology. There now exist a plethora of transgenic mouse models of neurological disease [55], [56]. However in the present case, where the proposed treatment is almost completely free of any safety concerns, or any reported adverse side effects, it can be validly questioned as to why the use of laboratory animal models should be encouraged. Animal models undoubtedly have disadvantages such as failure to replicate all the biological pathways found in human disease, difficulty in accurately measuring varied forms of cognitive performance, small size of mice and rats compared to humans, short lifespan affecting the development of age related diseases, and lack of lifestyle factors that adversely affect human diseases. Nevertheless, small animal models are less expensive, and require much less time and effort to obtain results than human clinical trials, so it is likely they will continue to be used to test tPBM for the foreseeable future.
4. PBM for stroke
4.1. Animal models
Perhaps the most well-investigated application of PBM to the brain, lies in its possible use as a treatment for acute stroke [57]. Animal models such as rats and rabbits, were first used as laboratory models, and these animals had experimental strokes induced by a variety of methods and were then treated with light (usually 810 nm laser) within 24 h of stroke onset [58]. In these studies intervention by tLLLT within 24 h had meaningful beneficial effects. For the rat models, stroke was induced by middle cerebral artery occlusion (MCAO) via an insertion of a filament into the carotid artery or via craniotomy [59], [60]. Stroke induction in the “rabbit small clot embolic model” (RSCEM) was by injection of a preparation of small blood clots (made from blood taken from a second donor rabbit) into a catheter placed in the right internal carotid artery [61]. These studies and the treatments and results are listed in Table 1.
Reports of transcranial LLLT used for stroke in animal models.
CW, continuous wave; LLLT, low level light therapy; MCAO, middle cerebral artery occlusion; NOS, nitric oxide synthase; RSCEM, rabbit small clot embolic model; TGFβ1, transforming growth factor β1.
4.2. Clinical trials for acute stroke
Treatment of acute stroke was addressed in a series of three clinical trials called “Neurothera Effectiveness and Safety Trials” (NEST-1 [65], NEST-2 [66], and NEST-3 [67]) using an 810 nm laser applied to the shaved head within 24 h of patients suffering an ischemic stroke. The first study, NEST-1, enrolled 120 patients between the ages of 40 to 85 years of age with a diagnosis of ischemic stroke involving a neurological deficit that could be measured. The purpose of this first clinical trial was to demonstrate the safety and effectiveness of laser therapy for stroke within 24 h [65]. tPBM significantly improved outcome in human stroke patients, when applied at ~ 18 h post-stroke, over the entire surface of the head (20 points in the 10/20 EEG system) regardless of stroke [65]. Only one laser treatment was administered, and 5 days later, there was significantly greater improvement in the Real- but not in the Sham-treated group (p < 0.05, NIH Stroke Severity Scale). This significantly greater improvement was still present at 90 days post-stroke, where 70% of the patients treated with Real-LLLT had a successful outcome, while only 51% of Sham-controls did. The second clinical trial, NEST-2, enrolled 660 patients, aged 40 to 90, who were randomly assigned to one of two groups (331 to LLLT, 327 to sham) [68]. Beneficial results (p < 0.04) were found for the moderate and moderate-severe (but not for the severe) stroke patients, who received the Real laser protocol [68]. These results suggested that the overall severity of the individual stroke should be taken into consideration in future studies, and very severe patients are unlikely to recover with any kind of treatment. The last clinical trial, NEST-3, was planned for 1000 patients enrolled. Patients in this study were not to receive tissue plasminogen activator, but the study was prematurely terminated by the DSMB for futility (an expected lack of statistical significance) [67]. NEST-1 was considered successful, even though as a phase 1 trial, it was not designed to show efficacy. NEST-2 was partially successful when the patients were stratified, to exclude very severe strokes or strokes deep within the brain [66]. There has been considerable discussion in the scientific literature on precisely why the NEST-3 trial failed [69]. Many commentators have wondered how could tPBM work so well in the first trial, in a sub-group in the second trial, and fail in the third trial. Lapchak's opinion is that the much thicker skull of humans compared to that of the other animals discussed above (mouse, rat and rabbit), meant that therapeutically effective amounts of light were unlikely to reach the brain [69]. Moreover the time between the occurrence of a stroke and initiation of the PBMT may be an important factor. There are reports in the literature that neuroprotection must be administered as soon as possible after a stroke [70], [71]. Furthermore, stroke trials in particular should adhere to the RIGOR (rigorous research) guidelines and STAIR (stroke therapy academic industry roundtable) criteria [72]. Other contributory causes to the failure of NEST-3 may have been included the decision to use only one single tPBM treatment, instead of a series of treatments. Moreover, the optimum brain areas to be treated in acute stroke remain to be determined. It is possible that certain areas of the brain that have sustained ischemic damage should be preferentially illuminated and not others.
4.3. Chronic stroke
Somewhat surprisingly, there have not as yet been many trials of PBM for rehabilitation of stroke patients with only the occasional report to date. Naeser reported in an abstract the use of tPBM to treat chronic aphasia in post-stroke patients [73]. Boonswang et al. [74] reported a single patient case in which PBM was used in conjunction with physical therapy to rehabilitate chronic stroke damage. However the findings that PBM can stimulate synaptogenesis in mice with TBI, does suggest that tPBM may have particular benefits in rehabilitation of stroke patients. Norman Doidge, in Toronto, Canada has described the use of PBM as a component of a neuroplasticity approach to rehabilitate chronic stroke patients [75].
5. PBM for traumatic brain injury (TBI)
5.1. Mouse and rat models
There have been a number of studies looking at the effects of PBM in animal models of TBI. Oron's group was the first [76] to demonstrate that a single exposure of the mouse head to a NIR laser (808 nm) a few hours after creation of a TBI lesion could improve neurological performance and reduce the size of the brain lesion. A weight-drop device was used to induce a closed-head injury in the mice. An 808 nm diode laser with two energy densities (1.2–2.4 J/cm2 over 2 min of irradiation with 10 and 20 mW/cm2) was delivered to the head 4 h after TBI was induced. Neurobehavioral function was assessed by the neurological severity score (NSS). There were no significant difference in NSS between the power densities (10 vs 20 mW/cm2) or significant differentiation between the control and laser treated group at early time points (24 and 48 h) post TBI. However, there was a significant improvement (27% lower NSS score) in the PBM group at times of 5 days to 4 weeks. The laser treated group also showed a smaller loss of cortical tissue than the sham group [76].
Hamblin's laboratory then went on (in a series of papers [76]) to show that 810 nm laser (and 660 nm laser) could benefit experimental TBI both in a closed head weight drop model [77], and also in controlled cortical impact model in mice [25]. Wu et al. [77] explored the effect that varying the laser wavelengths of LLLT had on closed-head TBI in mice. Mice were randomly assigned to LLLT treated group or to sham group as a control. Closed-head injury (CHI) was induced via a weight drop apparatus. To analyze the severity of the TBI, the neurological severity score (NSS) was measured and recorded. The injured mice were then treated with varying wavelengths of laser (665, 730, 810 or 980 nm) at an energy level of 36 J/cm2 at 4 h directed onto the scalp. The 665 nm and 810 nm groups showed significant improvement in NSS when compared to the control group at day 5 to day 28. Results are shown in Fig. 3. Conversely, the 730 and 980 nm groups did not show a significant improvement in NSS and these wavelengths did not produce similar beneficial effects as in the 665 nm and 810 nm LLLT groups [77]. The tissue chromophore cytochrome c oxidase (CCO) is proposed to be responsible for the underlying mechanism that produces the many PBM effects that are the byproduct of LLLT. COO has absorption bands around 665 nm and 810 nm while it has low absorption bands at the wavelength of 730 nm [78]. It should be noted that this particular study found that the 980 nm did not produce the same positive effects as the 665 nm and 810 nm wavelengths did; nevertheless previous studies did find that the 980 nm wavelength was an active one for LLLT. Wu et al. proposed that these dissimilar results may be due to the variance in the energy level, irradiance, etc. between the other studies and this particular study [77].
tPBM for TBI in a mouse model. Mice received a closed head injury and 4 hours later a single exposure of the head to one of four different lasers (36 J/cm2 delivered at 150 mW/cm2 over 4 min with spot size 1-cm diameter) ...
Ando et al. [25] used the 810 nm wavelength laser parameters from the previous study and varied the pulse modes of the laser in a mouse model of TBI. These modes consisted of either pulsed wave at 10 Hz or at 100 Hz (50% duty cycle) or continuous wave laser. For the mice, TBI was induced with a controlled cortical impact device via open craniotomy. A single treatment with an 810 nm Ga-Al-As diode laser with a power density of 50 mW/m2 and an energy density of 36 J/cm2 was given via tLLLT to the closed head in mice for a duration of 12 min at 4 h post CCI. At 48 h to 28 days post TBI, all laser treated groups had significant decreases in the measured neurological severity score (NSS) when compared to the control (Fig. 4A). Although all laser treated groups had similar NSS improvement rates up to day 7, the PW 10 Hz group began to show greater improvement beyond this point as seen in Fig. 4. At day 28, the forced swim test for depression and anxiety was used and showed a significant decrease in the immobility time for the PW 10 Hz group. In the tail suspension test which measures depression and anxiety, there was also a significant decrease in the immobility time at day 28, and this time also at day 1, in the PW 10 Hz group.
tPBM for controlled cortical impact TBI in a mouse model. (A) Mice received a single exposure (810 nm laser, 36 J/cm2 delivered at 50 mW/cm2 over 12 min) [121]. (B) Mice received 3 daily exposures starting 4 h post-TBI ...
Studies using immunofluorescence of mouse brains showed that tPBM increased neuroprogenitor cells in the dentate gyrus (DG) and subventricular zone at 7 days after the treatment [79]. The neurotrophin called brain derived neurotrophic factor (BDNF) was also increased in the DG and SVZ at 7 days , while the marker (synapsin-1) for synaptogenesis and neuroplasticity was increased in the cortex at 28 days but not in the DG, SVZ or at 7 days [80] (Fig. 4B). Learning and memory as measured by the Morris water maze was also improved by tPBM [81]. Whalen's laboratory [82] and Whelan's laboratory [83] also successfully demonstrated therapeutic benefits of tPBM for TBI in mice and rats respectively.
Zhang et al. [84] showed that secondary brain injury occurred to a worse degree in mice that had been genetically engineered to lack “Immediate Early Response” gene X-1 (IEX-1) when exposed to a gentle head impact (this injury is thought to closely resemble mild TBI in humans). Exposing IEX-1 knockout mice to LLLT 4 h post injury, suppressed proinflammatory cytokine expression of interleukin (IL)-Iβ and IL-6, but upregulated TNF-α. The lack of IEX-1 decreased ATP production, but exposing the injured brain to LLLT elevated ATP production back to near normal levels.
Dong et al. [85] even further improved the beneficial effects of PBM on TBI in mice, by combining the treatment with metabolic substrates such as pyruvate and/or lactate. The goal was to even further improve mitochondrial function. This combinatorial treatment was able to reverse memory and learning deficits in TBI mice back to normal levels, as well as leaving the hippocampal region completely protected from tissue loss; a stark contrast to that found in control TBI mice that exhibited severe tissue loss from secondary brain injury.
5.2. TBI in humans
Margaret Naeser and collaborators have tested PBM in human subjects who had suffered TBI in the past [86]. Many sufferers from severe or even moderate TBI, have very long lasting and even life-changing sequelae (headaches, cognitive impairment, and difficulty sleeping) that prevent them working or living any kind or normal life. These individuals may have been high achievers before the accident that caused damage to their brain [87]. Initially Naeser published a report [88] describing two cases she treated with PBM applied to the forehead twice a week. A 500 mW continuous wave LED source (mixture of 660 nm red and 830 nm NIR LEDs) with a power density of 22.2 mW/cm2 (area of 22.48 cm2), was applied to the forehead for a typical duration of 10 min (13.3 J/cm2). In the first case study the patient reported that she could concentrate on tasks for a longer period of time (the time able to work at a computer increased from 30 min to 3 h). She had a better ability to remember what she read, decreased sensitivity when receiving haircuts in the spots where LLLT was applied, and improved mathematical skills after undergoing LLLT. The second patient had statistically significant improvements compared to prior neuropsychological tests after 9 months of treatment. The patient had a 2 standard deviation (SD) increase on tests of inhibition and inhibition accuracy (9th percentile to 63rd percentile on the Stroop test for executive function and a 1 SD increase on the Wechsler Memory scale test for the logical memory test (83rd percentile to 99th percentile) [89].
Naeser et al. then went on to report a case series of a further eleven patients [90]. This was an open protocol study that examined whether scalp application of red and near infrared (NIR) light could improve cognition in patients with chronic, mild traumatic brain injury (mTBI). This study had 11 participants ranging in age from 26 to 62 (6 males, 5 females) who suffered from persistent cognitive dysfunction after mTBI. The participants' injuries were caused by motor vehicle accidents, sports related events and for one participant, an improvised explosive device (IED) blast. tLLLT consisted of 18 sessions (Monday, Wednesday, and Friday for 6 weeks) and commenced anywhere from 10 months to 8 years post-TBI. A total of 11 LED clusters (5.25 cm in diameter, 500 mW, 22.2 mW/cm2, 13 J/cm2) were applied for about 10 min per session (5 or 6 LED placements per set, Set A and then Set B, in each session). Neuropsychological testing was performed pre-LED application and 1 week, 1 month and 2 months after the final treatment. Naeser and colleagues found that there was a significant positive linear trend observed for the Stroop Test for executive function, in trial 2 inhibition (p = 0.004); Stroop, trial 4 inhibition switching (p = 0.003); California Verbal Learning Test (CVLT)-II, total trials 1–5 (p = 0.003); CVLT-II, long delay free recall (p = 0.006). Improved sleep and fewer post-traumatic stress disorder (PTSD) symptoms, if present beforehand, were observed after treatment. Participants and family members also reported better social function and a better ability to perform interpersonal and occupational activities. Although these results were significant, further placebo-controlled studies will be needed to ensure the reliability of this these data [90].
Henderson and Morries [91] used a high-power NIR laser (10–15 W at 810 and 980 nm) applied to the head to treat a patient with moderate TBI. The patient received 20 NIR applications over a 2-month period. They carried out anatomical magnetic resonance imaging (MRI) and perfusion single-photon emission computed tomography (SPECT). The patient showed decreased depression, anxiety, headache, and insomnia, whereas cognition and quality of life improved, accompanied by changes in the SPECT imaging.
6. PBM for Alzheimer's disease (AD)
6.1. Animal models
There was a convincing study [92] carried out in an AβPP transgenic mouse of AD. tPBM (810 nm laser) was administered at different doses 3 times/week for 6 months starting at 3 months of age. The numbers of Aβ plaques were significantly reduced in the brain with administration of tPBM in a dose-dependent fashion. tPBM mitigated the behavioral effects seen with advanced amyloid deposition and reduced the expression of inflammatory markers in the transgenic mice. In addition, TLT showed an increase in ATP levels, mitochondrial function, and c-fos expression suggesting that there was an overall improvement in neurological function.
6.2. Humans
There has been a group of investigators in Northern England who have used a helmet built with 1072 nm LEDs to treat AD, but somewhat surprisingly no peer-reviewed publications have described this approach [93]. However a small pilot study (19 patients) that took the form of a randomized placebo-controlled trial investigated the effect of the Vielight Neuro system (see Fig. 5A) (a combination of tPBM and intranasal PBM) on patients with dementia and mild cognitive impairment [94]. This was a controlled single blind pilot study in humans to investigate the effects of PBM on memory and cognition. 19 participants with impaired memory/cognition were randomized into active and sham treatments over 12 weeks with a 4-week no-treatment follow-up period. They were assessed with MMSE and ADAS-cog scales. The protocol involved in-clinic use of a combined transcranial-intranasal PBM device; and at-home use of an intranasal-only PBM device and participants/ caregivers noted daily experiences in a journal. Active participants with moderate to severe impairment (MMSE scores 5–24) showed significant improvements (5-points MMSE score) after 12 weeks. There was also a significant improvement in ADAS-cog scores (see Fig. 5B). They also reported better sleep, fewer angry outbursts and decreased anxiety and wandering. Declines were noted during the 4-week no-treatment follow-up period. Participants with mild impairment to normal (MMSE scores of 25 to 30) in both the active and sham sub-groups showed improvements. No related adverse events were reported.
tPBM for Alzheimer's disease. (A) Nineteen patients were randomized to receive real or sham tPBM (810 nm LED, 24.6 J/cm2 at 41 mW/cm2). (B) Significant decline in ADAS-cog (improved cognitive performance) in real but not sham (unpublished ...
An interesting paper from Russia [95] described the use of intravascular PBM to treat 89 patients with AD who received PBM (46 patients) or standard treatment with memantine and rivastigmine (43 patients). The PBM consisted of threading a fiber-optic through a cathéter in the fémoral artery and advancing it to the distal site of the anterior and middle cerebral arteries and delivering 20 mW of red laser for 20–40 min. The PBM group had improvement in cerebral microcirculation leading to permanent (from 1 to 7 years) reduction in dementia and cognitive recovery.
7. Parkinson's disease
The majority of studies on PBM for Parkinson's disease have been in animal models and have come from the laboratory of John Mitrofanis in Australia [96]. Two basic models of Parkinson's disease were used. The first employed administration of the small molecule (MPTP or 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) to mice [97]. MPTP was discovered as an impurity in an illegal recreational drug to cause Parkinson's like symptoms (loss of substantia nigra cells) in young people who had taken this drug [98]. Mice were treated with tPBM (670-nm LED, 40 mW/cm2, 3.6 J/cm2) 15 min after each MPTP injection repeated 4 times over 30 h. There were significantly more (35%–45%) dopaminergic cells in the brains of the tPBM treated mice [97]. A subsequent study showed similar results in a chronic mouse model of MPTP-induced Parkinson's disease [99]. They repeated their studies in another mouse model of Parkinson's disease, the tau transgenic mouse strain (K3) that has a progressive degeneration of dopaminergic cells in the substantia nigra pars compacta (SNc) [100]. They went on to test a surgically implanted intracranial fiber designed to deliver either 670 nm LED (0.16 mW) or 670 nm laser (67 mW) into the lateral ventricle of the brain in MPTP-treated mice [101]. Both low power LED and high power laser were effective in preserving SNc cells, but the laser was considered to be unsuitable for long-term use (6 days) due to excessive heat production. As mentioned above, these authors also reported a protective effect of abscopal light exposure (head shielded) in this mouse model [43]. Recently this group has tested their implanted fiber approach in a model of Parkinson's disease in adult Macaque monkeys treated with MPTP [102]. Clinical evaluation of Parkinson's symptoms (posture, general activity, bradykinesia, and facial expression) in the monkeys were improved at low doses of light (24 J or 35 J) compared to high doses (125 J) [103].
The only clinical report of PBM for Parkinson's disease in humans was an abstract presented in 2010 [104]
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5066074/
Photobiomodulation of the Brain
Michael R. Hamblin and Yng-Ying Huang - 2019 (Publication) 4512This is Hamblin and Huang's best summary of PBM for treating the brain.
Original Source: https://books.google.com/books/about/Photobiomodulation_in_the_Brain.html?id=P0qiDwAAQBAJ&source=kp_book_description
“Quantum Leap” in Photobiomodulation Therapy Ushers in a New Generation of Light-Based Treatments for Cancer and Other Complex Diseases: Perspective and Mini-Review
Luis Santana-Blank, MD, Elizabeth Rodríguez-Santana, MD, Karin E. Santana-Rodríguez, BS, and Heberto Reyes, MD - Photomedicine and Laser Surgery (Publication) 4490A broad article, but follow the references located at the original document to learn more about more applicable topics.
Abstract
Objective: Set within the context of the 2015 International Year of Light and Light-Based Technologies,and of a growing and aging world population with ever-rising healthcare needs, this perspective and mini-review focuses on photobiomodulation (PBM) therapy as an emerging, cost-effective, treatment option for cancer (i.e., solid tumors) and other complex diseases, particularly, of the eye (e.g., age-related macular degeneration, diabetic retinopathy, glaucoma, retinitis pigmentosa) and the central nervous system (e.g., Alzheimer's and Parkinson's disease). Background data: Over the last decades, primary and secondary mechanisms of PBM have been revealed. These include oxygen-dependent and oxygen-independent structural and functional action pathways. Signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a given set of parameters. Methods: This study was a perspective and nonsystematic literature mini-review. Results: Studies support what we describe as a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photo-cceptors to restore physiologic function. Conclusions: Based on existing evidence, it is argued that PBM therapy can raise the standard of care and improve the quality of life of patients for a fraction of the cost of many current approaches. PBM therapy can, therefore,benefit large, vulnerable population groups, including the elderly and the poor, whilehaving a major impact on medical practice and public finances.
Introduction
The United Nations declared 2015 to be the International Year of Light and Light-Based Technologies (IYL 2015) in recognition of the vital role of light-based systems in our daily lives, and their growing importance to meeting the world's challenges in areas as diverse as energy, education, telecommunication, agriculture, and health.1 Although our perception of light is often limited to the visible band of the electromagnetic (EM) spectrum,2 both lower and shorter wavelengths are increasingly used in new medical technologies3 including soft, injectable, and bioresorbable electronics.4 Described as an imperative cross-cutting discipline of in the twenty-first century, light science has already revolutionized the physical sciences and industry. The control of light at the nanoscale has unveiled a plethora of phenomena, leading to powerful new applications and setting high expectations for years to come.5 In particular, light's ability to control materials and transport coded signals forms the bases for many new photonic devices and systems, wherein photons act as tailor-made EM energy packets that can perform various functions.
Here, we describe a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photoacceptors to control biologic function in medicine through photobiomodulation (PBM) therapy. We propose that progress will lead to the imminent inception of PBM therapy as a mainstream treatment for multiple complex diseases, including solid tumors, as well as neurodegenerative diseases (NDs) of the eye and central nervous system (CNS)6–10 (Fig. 1). PBM therapy can raise the standard of care and improve the quality of life of patients at a fraction of the cost of many current approaches. Thus, a “quantum leap” in PBM therapy will benefit large and vulnerable population groups, including the elderly and the poor, while having a major impact on medical practice and public finances.11 This is particularly important because the high price of drug therapies, which can reach hundreds of thousands of dollars per year,12 as well as a growing and aging world population, are putting a severe strain on family and public finances around the world.13
Flow chart illustrating fields of light-based technologies, highlighting photobiomodulation (PBM) therapy applied to complex diseases as a quantum leap in medical therapeutics.
Origin, Trajectory and Myriad Relationships in PBM's “Quantum Leap” in Medicine
Concurrent with progress in PBM therapy, a long history of discoveries has put medicine at the brink of a revolution in the use of light–water interactions for the treatment of complex diseases.7,8,10,14 Long ago, Albert Szent-Gyorgyi postulated that water was at the core of energy transfer in biological systems (i.e., quantum biology), and that that explained how energy from biomolecules could be translated into free energy for cells.15–17 Ling further elaborated on the physical state of water in living cells,18 and proposed on theoretical grounds that ordered layers of water could extend infinitely under ideal conditions.19,20Later, Huber proposed a structural basis of light energy and electron transfer in biology.21 More recently, Zewail and others showed that, with rapid laser techniques, it is possible to “see” how atoms in a molecule move during a chemical reaction.22 Light science has now reached microscales at the limit of recordable physical observation (e.g., resonant intermolecular transfer of vibrational energy in water at −100 fs)23,24showing, for example, the memory of persistent correlations in water structures within 50 fs, which is important in stabilizing biological systems.25 These and other tremendous achievements have changed our view of water, from a merely passive medium to an integral active player in the physiology of life, and have opened the gates to both direct measurement and control of physiological processes via light–water interaction.
State of the Art in PBM
In 2016, PBM therapy will be added to the MeSH database as an entry term for records spanning five decades of research.26 As argued by Anders et al., this is a key step, as it distinguishes PBM therapy from light-based devices used for heating of tissues, such as near infrared (NIR) lamps or other applications that rely on thermal effects for all or part of their mechanisms of action.26 In contrast, PBM therapy employs low-level monochromatic or quasimonochromatic light, currently from visible blue (∼400 nm) to far-infrared (FIR ∼3200 nm), to induce nonthermal (≤0.01°C) photochemical and photophysical effects. Nonlinear processes through which PBM therapy can stimulate or inhibit; that is, modulate, physiological activity depend upon signal-to-noise rate and target cell/tissue parameters.27–29 Thus, signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a narrow set of parameters.13
Over the last decades, primary and secondary mechanisms of PBM at the tissue, cellular, and molecular levels have been revealed. These include two major structural and functional action pathways. The first, or classic, action pathway relates to oxygen-dependent mechanisms operated by oxidation-reduction enzymes of the respiratory chain, particularly cytochrome c oxidase (CcO), which is partly responsible for light energy absorption and transfer to cells and tissues.30 This pathway is associated to cofactors, pigments, metals, and proteins that act as key redox centers within the body's bioenergetic rack mechanism described by Huber.21 Nitric oxide (NO), as a first-level player, also has an activation and modulation role in the oxygen-dependent pathway.31–33
The second, or oxygen-independent, action pathway centers on the vital role of water not only as the prevalent medium of life but as an active molecule, capable of absorbing radiant energy (e.g., IR light) and transporting/transducing it along extended biological surfaces, from bulk water to confined water in nanoscopic tissue and cell spaces. Light–water dynamics precede/coexist with the classic oxygen-dependent action pathway and complement and facilitate energy transfer for increased adenosine triphosphate (ATP) production.29,34,35 As a point of comparison, correlated internal electron- and proton-transfer reactions have been tracked in real time into the oxidized enzyme (CcO), revealing an overall real time of 3.46 ms.36 This relay is slower by several orders of magnitude than total energy transport through water dynamics from bulk liquid water to confined spaces.34
Oxygen-independent light–water interactions may further power and modulate molecular signaling pathways and gene transcription factors via multiple nonmetabolic pathways.10,35 For examle, the energy of the drive force wave of an infrared pulsed laser device (IPLD) used in our group's previous studies (NIR 0.27 eV) is within the range of the strength of hydrogen bonds,29,37 and the IPLD carrier wave oscillates at a frequency (3x 10e6 Hz) that enters in vibrational resonance with the rate of electron transfer through the DNA double helix.29,37 Theoretical evidence suggests that these wave properties promote the activation of open state dynamics,38,39 allowing the activation of complex chaotic dynamics as well as the regulation of DNA replication and transcription, because the existence of open states in one place of the chain can influence the dynamics of other distant open states.29,34,35 Resulting effects match reported reductions in the frequency of chromosome aberrations induced by that low-energy laser irradiation,40 as well as theoretical,38,39 experimental,27,28 and clinical studies.41–48 These and other oxygen-independent PBM effects are channeled through metabolic control levels to regulate the energy-dependent path from the genotype to the phenotype.49,50
Light–Water Interactions and the Quantum Leap in PBM
We propose that the key to understanding and controlling the biophysics and biochemistry of higher-order organisms stems from their dual aqueous and energy-dependent nature. Water represents ∼70% by mass of an adult human body, or nearly 99% of total molecules by number, given water's low molecular weight. In addition, high-order organisms, including humans, can be represented as complex electrochemical (semiconducting) systems that comprise a vast array of energy-sensitive materials and machinery, such as ion pumps (e.g., chemically driven electron pumping through molecular wires, such as the D pathway in CcO),34 molecular motors (e.g., ATP synthase and Brownian biomotors), transistors-capacitors (e.g., cell membrane), liquid crystals (e.g., membrane structure), and rechargeable electrolytic biological batteries (e.g., hydrophilic interface in cells/tissues). Life system's double nature, whose two main structural and functional pillars are energy and water joined to biomolecules, has, therefore, tremendous consequences for life and health.
Water's permittivity, calculated considering the system as a plane capacitor, is generally high. Therefore, radiant energy can penetrate and be absorbed by tissues to provide powerful tools in medicine.51 One example is the exclusion zone (EZ) described by Pollack.52 High-energy EZ water forms along hydrophilic surfaces (e.g., tissue interfaces) in response to radiant energy.53 Remarkably, EZ water can separate and store electrical charges, and can release up to 70% of such charges when it is perturbed, such as by injury-induced redox potentials.54 We have argued that supplied energy can power and modulate cellular work and signaling pathways, even when the metabolic energy pathway has been compromised, steering cells toward or away from programmed cell death.34 EZ water may, thus, act as an electrolytic bio-battery,35 which can efficiently and selectively transfer energy to sites expressing redox injury potentials, as found in cancer and other complex diseases, triggering reparative and regenerative mechanisms that can lead to restoring homeostasis/homeokinesis and, ultimately, health.29,34,35
Experimentally, IR energy absorption by water has been recently modeled in a porcine model, confirming that absorption depends upon fluence and wavelength. Further, the higher the concentration of water in tissues, the higher IR energy absorption will be.55 This is consistent with controlled clinical studies in solid tumors and complex ophthalmic and neurologic diseases,9,46,56 as well as molecular, biochemical, biophysical, and metabolic mechanistic support for a quantum leap in medical therapeutics based on the simple, but powerful, idea that properly tailored light can power and modulate physiologically reparative mechanisms.30,57–62
Cancer and Tumor Microenvironments
The bases of our understanding of cancer are constantly being questioned and revised, leading to new treatment goals. In a paradigm-changing editorial, Prendergast recently argued that “disorders in microenvironment and peripheral systems that control cancer might increasingly be viewed as primary rather than secondary factors in the root nature of cancer as a clinical disease.” This constitutes “a crucial and radical distinction from prevailing thought, since it implies that cancer may be a symptom of an underlying clinical disorder, rather than the root problem itself that needs to be addressed.” 6,63
Prendergast further suggested that “effective treatment of cancer may not necessarily entail understanding or addressing this complexity, but mastering the use of tissue or systemic systems that have the inherent ability to do so.” Hence, a common thread linking emerging perspectives in oncology and PBM therapy may well be the restitution of tissue homeostasis-homeokinesis via light-energy supplementation, a microenvironment effect that comprises and extends the Warburg effect previously discussed by our group.57,64–67
Photobiomodulation and Cancer
As far back as 1964–1966, McGuff et al. showed 64,65 that “laser energy has a selective effect on certain malignant tumors, resulting in their progressive regression and ultimate dissolution.” Following years of controversy,66,67 editorials by Karu68 and Lanzafame11,69 now stress evidence supporting the potential anticancer effects of PBM.11,68,69 New data confirm that PBM under certain parameters is safe for use in cancer patients.60 This is in accord with clinical results from our group using the abovementioned proof of concept IPLD.4,44
A phase I trial in patients with advanced neoplasias demonstrated that the IPLD studied was safe for clinical use and improved performance status and quality of life.41 Antitumor activity was observed in 88.23% of patients with 10 years of follow-up.41
In that series, T2-weighted MRI data showed increased water content of tumor heterogeneities42,44 preceding tumor-volume reduction and a therapeutic anticancer effect.42,44 Structural, kinetic, and thermodynamic implications of these changes in water dynamics have been analyzed at the tissue, cell, and interstitial levels.27 In conjunction, selective activation of programmed cellular death [i.e., apoptosis, necrosis, and anoikis (cell death by loss of cell adhesion)] and cytomorphologic modification (e.g., reduced size, increased roundness, increased vacuoles) were documented in neoplastic cells, but not in peripheral tissues.8,42 Modulation of cluster of differentiation (CD)4 CD45RA+, CD25 activated, tumor necrosis factor alpha (TNF-α), and soluble interleukin (IL)-2 receptor (sIL-2R) was further documented.43These hallmark results, supported by independent data,70–72 demonstrate that PBM therapy can modulate antitumor effects,6,8 in sharp contrast with long-held views.45,73,74 This evidence is also consistent with growing experimental and clinical reports from multiple other authors.60,75–82
PBM and Ophthalmic and Neurodegenerative Disorders
Recent evidence underscores common mechanisms between cancer and NDs of the eye and CNS. Research suggests that oxidative proteome damage may be the most likely cause of aging and age-related maladies such as cancer and other complex diseases, including NDs.83 Findings also show “common mechanisms of onset,” with a focus on genes such as DJ-1 and Myc-Modulator 1 (MM-1) and signaling pathways that contribute to the onset and pathogenesis of cancer and NDs such as retinitis pigmentosa (RP), Parkinson disease (PD), and cerebellar atrophy.”84 Finally, both disease groups are profoundly energetic in nature, featuring prominent deterioration of metabolic energy pathways.10
External light energy supplementation has been shown to generate neuroprotective, vasoprotective, baroprotective, immunomodulatory, and regenerative effects (Fig. 2). 47 We have documented that such effects may be activated and modulated locally and/or remotely via oxygen-dependent and oxygen-independent pathways that can encompass extended biologic surfaces and may even reach avascular eye tissues (i.e., cornea, lens, aqueous humor, and vitreous) noninvasively. Although a full elucidation of involved mechanisms escapes the scope of this perspective and mini-review, a very brief discussion of results from multiple authors is given subsequently.
Electromagnetic (light) energy supplementation based on water–light interactions. Upper left side shows classic oxygen (O2) dependent pathways by which light energy generates adenosine triphosphate (ATP)/ guanosine-5′-triphosphate (GTP) and other high-energy molecules. Upper right side shows O2 independent pathways by which photoinduced, nonlinear, oscillations in water provide energy for cellular work, signaling, and gene transcription. Top center shows interfacial exclusion zone (EZ) water, which acts as a selective rechargeable electrolytic bio-battery. Together, these pathways activate and modulate physiologically reparative mechanisms which, at appropriate irradiation parameters, can generate neuroprotective, vasoprotective, baroprotective, immunomodulator, and regenerative effects locally and remotely, promoting homeostasis/homeokinesis through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by the second law of thermodynamics. Arrows point to the sequence and direction of events. (Updated from reference 47. Authors retained copyright.)
PBM has shown promise in the treatment of diabetic retinopathy (DR),85,86 age-related macular degeneration (AMD),46 glaucoma,47 RP,87 Stargardt disease,88 Leber's hereditary optic neuropathy,89 Alzheimer's disease (AD), and PD, 90,91 among other conditions.89 Strikingly, although each of these NDs has different etiologies and pathogeneses, “they frequently induce a set of cell signals that lead to well-established and similar morphological and functional changes, including programmed cell death. Furthermore, oxidative stress, activation of apoptotic pathways and inflammatory response, are common features in all these diseases.”92
Remarkably, PBM can modulate apoptosis as well as necrosis.42,45,47 PBM can also be both pro-oxidant in the short term, but antioxidant in the long term,93 thus modulating reactive oxygen species (ROS) generation. We also found clinical evidence of immune regulatory effects over inflammation during treatment of solid tumors with the IPLD, a NIR diode laser pulsed at a frequency of 3 MHz.43 These results are in agreement with the regulating role of the vagal reflex on the inflammatory reflex reported by Tracey, using an electronic device that stimulated nerves to treat inflammation.71,72
In addition, PBM has been shown to protect against retinal dysfunction and photoreceptor cell death in rodent models of retinal injury and retinal degeneration.94 PBM has been further reported to attenuate oxidative stress and inflammation in primary astrocytes induced by amyloid β peptide (Aβ),95 and to reduce Aβ-induced apoptosis,96 which is thought to play a major role in AD. Nevertheless, it has been argued that red to NIR light cannot be transmitted through the scalp to the brain more than a few centimeters,97 which makes it nearly impossible to noninvasively treat AD with PBM 98 using conventional (direct) delivery systems/methods. Similarly, although an absence of adverse effects from 670 and 830 nm PBM applied to the retina in Sprague Dawley albino rats has been reported,94 extreme care must be taken to avoid photodamage of the eye99 from direct PBM procedures.
Conversely, we published an interventional case report of a patient with bilateral geographic atrophic AMD (gaAMD) and associated neurologic disease treated noninvasively, indirectly, and at a distance (i.e., remotely) from ocular structures and the CNS with the above-referenced IPLD/photo-infrared pulsed bio-modulation (PIPBM).46 Results showed neurologic improvement, transitory color vision, enhanced visual acuity, full-field electroretinogram (ERG) modifications toward a normal rhythm, drusen mobilization, decreased lens opacity, and lower intraocular pressure (IOP), in accord with a retrospective noncomparative data analysis from the phase I trial of patients with advanced cancer treated with the IPLD,41 which showed statistically significant evidence of a therapeutic hypotensor effect over IOP,47 and they are consistent with the positive neurological evolution of two trial patients.
Moreover, although trial participants did not develop media opacity, one pre-existing incipient cataract in the right eye of a patient (transitional meningioma) became denser and slightly smaller 3 months post-treatment, and remained unchanged 1 year post-treatment. The left eye lens of the same patient was unaffected. Although the finding could be part of the natural history of the cataract, we stressed that possible deterministic effects related to the initial metabolic or biochemical state of lens opacities should be studied.41
In accordance with the what was described, a robust body of evidence suggests that protein misfolding, insolubility, and aggregation are at the root of both cataracts and other diseases including AD, PD, and Huntington's disease,100 and that external EM energy (light) supplementation can have reparative effect on protein misfolding, activating and modulating metabolic control levels of protein folding/unfolding.10,34In addition, PBM effects on targets such as heat shock proteins (α crystalline), enzymes of the antioxidative system, Na+-K+-ATPase, Ca +2-ATPase, aquaporins (AQPs), and ion pumps have been referred to as part of mechanisms that could have influenced the response observed in the lens on the cases studied.46 We further proposed that, among other effects, PBM can stimulate and/or substitute ATP production via water dynamics, which is vital for the activation and inactivation kinetics in phototransduction.46 PBM can also affect the synthesis of molecules in a liquid crystalline (LC) state (e.g., self-assembly of lipids, water, and other biomolecules such as proteins and sterols, which are sensitive to temperature and/or electric fields) If confirmed, the latter may have multidisciplinary applications in medicine and biology in areas such as photovision, in which LCs are essential functional components.28
A first rapid communication referring to the retina and optic nerve additionally showed first evidence of EZ water as a selective rechargeable bio-battery applicable to PBM, suggesting a new understanding of the eye's energetic environment, which may have deep implications in ocular physiology as well as in the pathophysiology, diagnosis, and treatment of blinding diseases using light-based therapies.48 Therefore, as a promising alternative to drug therapies,101 or in combination with other treatments, PBM therapy may be developed into a viable therapeutic approach with multidisciplinary applications in ophthalmology and neuroscience,46 inducing and modulating physiologically reparative and regenerative effects that can favor homeostasis/homeokinesis27–29 through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by thermodynamics.
Treatment Costs and Availability
At the 2015 American Society of Clinical Oncology (ASCO) annual meeting, Dr. Leonard Saltz, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center, discussed the high cost of cancer drugs. He argued that “the unsustainably high prices of cancer drugs is a big problem, and it's our problem,” citing as examples the cost of nivolumab ($28.78/mg) and ipilimumab ($157.46/mg), which is “approximately 4000 times the cost of gold.”102 Previously, >100 oncologists had protested the high price of cancer drugs, also calling them economically “unsustainable.” They noted that, of 12 cancer drugs approved in 2012, 11 were priced > $100,000 per year,103 with multiple drugs often being required for extended periods. Such high prices and their impact on families, governments, and society at large are leading some to propose that cost should be considered a “financial toxicity” to be assessed with other toxicities when treatments are considered by doctors and patients.104 In contrast, although it has been estimated the cost of developing new drug therapies can run up to USD $1.3–$1.7 billion,105,106 the development cost of new photonics devices can be substantially lower, which can lower therapy costs and increase treatment availability. For the same reasons, PBM can also offer a noninvasive and cost-effective therapeutic option for patients with NDs of the retina, brain, and beyond.56, 85
Conclusions
The celebration of the IYL 2015 by the United Nations1 is a fitting time to announce what we describe here as a “quantum leap” in PBM therapy. It is also a good opportunity to ensure that policy makers and the medical community become aware of and embrace the immense potential of light-based medical technologies, especially PBM therapy, as an emerging treatment option for cancer and other complex diseases.107 Although not all tissues respond to PBM therapy,69 in vitro and in vivo xenografts and evidence from clinical studies does suggest that it is time to begin considering PBM therapy as a potential drug equivalent.11,108 In addition, PBM therapy may have minimal or no adverse effects, improve quality of life and functional status and raise the current standard of care for many cancer patients when used alone or in combination with other therapies.9 PBM therapy further represents a novel hope for the treatment of numerous eye and neurologic diseases. And as stated, PBM may be developed at a lower cost than many current treatments,8,10 which can help meet the healthcare needs of an increasing and aging world population. As such, this perspective and mini-review focuses on the large potential tangible contributions of light-based therapies for large demographic segments of the population, such as aging “baby boomers” who are expected to face a higher incidence of diseases such as cancer, AMD, DR, glaucoma, RP, AD, and PD, as well as other neurologic diseases in the next 15 years. In light of the growing costs of drugs and their impact on developed and developing countries, we propose that PBM therapy may offer a novel, safe, and effective therapy choice that would be more accessible to large vulnerable groups, such as the poor and the elderly.
Concurrently with the United Nations' declaration of 2015 as the year of light and light-based technologies, PBM therapy stands at the brink of delivering a new generation of treatments for complex diseases. New PBM therapies will preserve quality of life and raise standard of care in an efficient and cost-efficient manner. This will particularly benefit the most vulnerable demographic sectors, such as the elderly and the poor, and reduce the strain of growing healthcare costs in both industrialized and developing countries. We propose that such developments and their imminent impact represent a paradigm shift or “quantum leap” in PBM therapy and medicine at large.
Acknowledgments
We thank Jesús Alberto Santana-Rodríguez for reviewing and editing this article, and Luis Rafael Santana-Rodríguez for design and technical support. This study was supported by Fundalas, Foundation for Interdisciplinary Research and Development.
Author Disclosure Statement
No competing financial interests exist.
References
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4782038/
This book covers an astonishing amount of information in its near thousand pages, everthing from basic laser physics to dental, and veteranary useage. Here are some of its contents:

- Basic Laser Physics
- physics
- energy
- radiation
- wavelength and frequency
- photon energy
- the elecromagnetic spectrum
- the optical reigon
- radiation risks
- can electromagnetic radiation cause cancer
- protective mechanisms
- light
- the optical spectrum
- light sources
- various sources of radiation
- natural sources of radiation
- man-made light sources
- the light emmiting diode (LED)
- flash lamps
- the laser
- laser design
- practical lasers
- the properties of laser light coherence
- interference
- laser beam characteristics
- polarisation
- output power
- continuous and pulsed lasers
- the peak power value
- average power output
- power density
- light distribution
- beam divergence
- collimation
- risk of eye injury
- decisive factors in the risk of eye injury
- the laser instrument
- properties of some laser types
- description of common surgical laser types
- the CO2 laser (carbon dioxide laser)
- carbon dioxide lasers in surgery
- carbon dioxide lasers in dental applications
- the Nd:YAG laser
- Nd:YAG lasers in surgery
- Nd:YAG lasers in dentistry
- erbium lasers in dentistry
- "strong" diode lasers in dentistry
- the KTP laser
- Q-switching
- Theraputic Lasers
- the first generation 1975-85
- the second generation 1985-95
- the third generation 1995-2005
- the fourth generation 2005 and onwards
- what is a good laser therapy instrument
- the basic instrument
- sales tricks
- high power-low power
- laser or LED
- high or low price
- penetration of light into tissue
- "a story of a young scientist"
- the wavelength
- how deep does light penetrate into tissue?
- Biostimulation
- history
- a few words on mechanisms
- photoreceptors
- what parameters to use
- laser parameters
- whitch wavelength?
- output power
- average output power
- power density
- energy density
- the dose
- treatment dose
- calculation of doses
- dose ranges
- calculation of treatment time for a desired dose
- "reay reckoner"
- dose per point
- pulsed or continuous light
- pulse repetition rate (PRP)
- patient parameters
- treatment area
- treatment intervals
- pre- or postoperative treatment
- treatment method parameters
- local treatment
- shallow problems
- deeper problems
- treating inside the body
- systemic treatments
- acccupuncture
- trigger points
- spinal processes
- dermatome
- blood irradiation
- irradiation of lymph nodes
- irradiation of ganglions
- combo treatment
- interaction with medication
- other considerations
- what about collimation?
- depth of penetration, greatest active depth
- factors that reduce penetration
- tissue compression
- how deep does the light penetrate?
- laser light irradiation through clothes
- the importance of tissue and cell condition
- the importance of ambient light
- in vitro/ in vivo
- laser therapy with high output lasers
- laser therapy with carbon dioxide lasers
- laser therapy with Nd:YAG lasers
- laser therapy with ruby lasers
- laser therapy with Er:YAG lasers
- laser therapy with surgical diode lasers
- risks and side effects
- the importance of correct diagnose
- cancer
- cytogentic effects?
- a false picture of health
- tiredness
- pain reaction
- do high doses of laser therapy damage tissue?
- is it only an effect of temperature?
- protection against radiation injury
- how to measure effects of laser therapy
- thermography
- magnetic resonance imaging
- high resolution digitized ultrasound B-scan
- tensile strength
- other objective methods
- does it have to be a laser?
- FDA (Food and Drug Administration)
- how well documented?
- confused?
- the funding research
- as time goes by
- Medical indications
- who and what can be treated?
- acne
- allergy
- antibiotic resistance
- arteriosclerosis
- arthritis
- asthma
- blood preservation
- blood pressure
- bone regeneration
- burning mouth syndrome
- cancer
- cardiac conditions
- carpal tunnel syndrome
- cerebral palsy
- crural and venous ulcers
- delayed onset muscular soreness (DOMS)
- depression, psychosomatic problems
- diabetes
- duodenal/gastric ulcer
- epicondylitis
- erythema multiform major
- fibrositis/fribomyalgia
- headache/migraine
- heamorrhoids
- herpes simplex
- immune system modulation
- inflammation
- inner ear conditions
- laryngitis
- lichen
- low back pain
- mastitis
- microcirculation
- morbus sluder
- mucositis
- muscle regeneration
- mycosis
- nerve conduction
- nerve regeneration and function
- oedema
- ophthalmic problems
- pain
- periostitis
- plantar fasciitis
- salivary glands
- sinuitis
- spinal cord injuries
- snake bites
- sports injuries
- stem cells
- stroke, irradiation of the brain
- tendinopathies
- tinnitus, vertigo, meniere's disease
- tonsillitis
- trigeminal neuralgia
- thrombophlebitis
- tuberculosis
- urology
- warts
- wiplash-assosiated dissorders
- vitiligo
- womens' health
- wound healing
- zoster
- idications in the pipeline
- alzheimer's disease
- botox failures
- cellulites
- cholesterol reduction
- complex reigonal pain syndrom (CRPS)
- eczema
- erectile dysfunction
- familiar amyotrophic lateral sclerosis (FALS)
- glomerulonephritis
- obesity
- orofacial granulomatosis
- Parkinson's disease
- post-mestrual stress
- pemphigus vulgaris
- sleeping disorders
- withdrawal periods
- wrinkles
- consumer lasers
- Dental LPT
- the dental laser literature
- on which patients can LPT be used?
- dental indications
- alveolitis
- anaesthetics
- aphthae
- bleeding
- bisphosphonate related osteonecrosis of the jaw
- caries
- dentitio dificilis (pericoronitis)
- endodontics
- extraction
- gingivitus
- herpes zoster
- hypersensitive dentine
- implantology
- leukoplakia
- lingua geographica (glossitis)
- lip wounds
- nausea
- nerve injury
- orthodontics
- mild dental pain
- paediatric dental treatment
- periodontics
- prosthetics
- root fractures
- secondary dentine formations
- temperature caveats
- toemporo-mandibular disorders (TMD)
- TMD and endodontics
- other dental laser applications
- dental pohoto dynamic therapy
- composite curing
- deminerallisation
- tooth bleaching
- caries detection
- lasers as a diagnostic tool
- case reports
- Non Coherent Light Sources
- Veterinary Use
- case reports
- Contra Idications
- pacemakers
- pregnancy
- epilepsy
- thyroid gland
- children
- cancer
- haemophilia
- irradiation of the brain
- radiation therapy patients
- diabetes
- tatoos
- light sensitivity
- Coherence
- the role of coherence in laser phototherapy
- itroduction
- summary
- Dose and Intensity
- basics about energy
- output power
- power density
- the laser beam
- the laser probe
- pulsed lasers
- energy density
- treatment dose
- the dose does not demend on the intensity
- dose per point
- more about treatment technique
- The Mechanisms
- are biostimulative effects laser specific?
- is it possible to prove that laser therapy doesn't work?
- comparisons between coherent and non-coherent light
- what is the importance of the length of coherence
- hode's hamburger
- hode's big burger
- abrahamson's apple
- moonlight
- how deep does light penetrate tissue?
- bright light phototherapy
- similarities and differences
- possible primary mechanisms
- polarisation effects
- what characterises the light in a laser speckle
- porphyrins and polarised light
- cell cultures and tissue have different optical properties
- tthe effect of heat development in the tissue
- macroscopic heating
- the microscopic heat effect
- mechanical forces
- excitation effects
- primary reactions due to excitation
- secondary reactions due to cell signaling
- flourescence-luminescence
- multi-photon effects
- llasting effects in tissue
- non-linear optical effects
- opto-acoustic waves
- secondary mechanisms
- effects on pain
- effects on blood circulation
- stimulatory and regulatory mechanisms
- effects on the immune system
- other interesting possibilities
- summary of mechanisms
- diagnostics with therapeutic lasers
- photodynamic therapy - PDT
- other medical uses of lasers
- A Guide for Scientific Work
- methodology of a trial
- parameters
- technical parameters
- treatment parameters
- medical parameters
- closer description of the technical parameters
- name of instrument (producer)
- laser type and wavelength
- laser beam characteristics
- number of sources
- beam delivery system
- output power
- power density at probe aperture
- calibration of the instrument
- closer description of the treatment parameters
- treatment area
- dose: energy density
- dose per treatment and total dose
- intensity: power density
- treatment method
- treatment distance (spot size), type of movement, scanning
- sites of treatment
- number of treatment sessions
- frequency of treatment sessions
- closer description of the medical parameters
- description of the problem to be treated
- patients (number, age, sex)
- exclusion criteria
- inclusion criteria
- condition of patient
- pre-, parallel-, or post-medication
- treated with other methods before
- drop-out rates
- follow up
- outcome measures
- statistical analysis
- economy
- gallium-alluminium and all that
- recommendations of WALT - the world assosiation for laser therapy
- The Laser Phototherapy Literature
- the importance of reporting all laser parameters - even in the abstract
- diclofenac, dexamethasone or laser phototherapy?
- another pithole in LPT research
- database of abstracts of reviews of effects (DARE)
- the wound healing contradiction
- wikipedia
- poor documentation - compared to what?
- LPT equipment and the future
- english language books od LPT:
- books in other languages, with ISBN
- laser phototherapy journals
- information for your patient
Original Source: http://www.coldlasers.org/lllt-books/

The Biological Basics of Low Level Laser Light Therapy
- summary
- introduction
- Alexander Gurwitsch: cells emit light
- non-linear dynamics
- introducing quantum physics
- itroduction to quantum biology
- quantum coherence in biology
- biological coherence and the sensitivity of living systems
- Fritz Albert Popp: biophotons
- Guenther Albreecht-Buehler: cells respont to light
- Mae-Wan Ho: visualizing coherence
- conclusions
Therapeutic Laser Applications
- how does low level laser therapy work?
- what are the advantages over other modes of therapy?
- cliniclal use of low level laser therapy
- abstract submitted to laser and surgury medicine
- background and objective
- methods
- results
- conclusion
- safety considerations
- eye considerations
- pace makers and other implanted devices
- pregnancy
- excessive toxicity
- preface to treatment section
Nerver Roots
- flexion and extension
- lateral flexion
- rotation
- MRT (muscle response testing) through ROM of cervical spine
- shoulder
- neurological level
- C5
- C6
- C7
- C8
- T1
- S1
- L5
- L4
- L3
- L3-L5
- L2-L4
- L1-L3
- low back
Top Ten Laser Protocols
- organ / glands / tissue
- acute injury (shock)
- pain
- lymphatic protocol
- detox protocol
- immune protocol
- hormone protocol
- basic cranial nerve
- tissue memory
- trauma preparation protocol
A-Z Laser Protocols
- abdominal cramping
- abdominal inflammation/pain
- abrasions
- abscess
- achilles tear / strain (partial only; not rupture)
- acidosis (hyperacidity
- acid reflux
- acne
- acute injury
- adenoids
- (ADD) atention deficit disorder and hyperactivity disorder (ADHD)
- Addiction
- addison's disease
- adhesions
- adhesive capsulitis
- adrenal
- aids
- allergies
- alopecia
- alpha waves
- alzheimer's
- amenorrhea
- amoebas
- amyotrophic lateral sclerosis / lou gehrig's disease / motor neuron
- amnesia
- anemia
- anger
- angina
- anosmia (loss of smell)
- anxiety appendicitis
- arrhythmias
- arteries / arteriosclerosis
- arthritis
- asthma
- ataxia
- athlete's foot
- atrophy
- backache / back pain
- bacteria
- bed sores
- bedwetting
- bell's palsy
- beta waves
- bites
- bladder
- bleeding gums
- bloating
- blood pressure (high)
- blood pressure (low)
- blood sugar balance
- boils
- bone
- bowel
- bradycardia
- brain
- breast augmentation
- bronchitis
- bruises
- buerger's disease
- bunions
- burns
- burns (second degree)
- bursitis
- calcium deposits or formations
- candida
- canker sores
- capsulitis
- carpal tunnel syndrome
- cartilage
- cataracts
- chemical peels / resurfacing
- chest pain
- chicken pox (herpes zoster / varicella)
- cholecystitis
- cholelithiasis
- chronic fatigue
- chronic pain
- circulation
- cirrhosis
- cold sores (herpes simplex 1)
- colds and flu
- colitis
- concussion
- confusion
- congestion
- congestive heart falure (CHF)
- conjunctivitis (pink eye)
- costipation
- cramps (muscle)
- cranial nerves (general)
- cranial nerves VIII
- crepitus
- crohn's disease
- cuts
- cushing's syndrome
- cytomegalovirus (herpes syndrome V)
- deer tick
- delta waves
- depression
- dermatitis
- detoxification
- diabetes
- diabetic neuropathy
- diabetic ulcers
- digestion
- dim vision
- disc herniation
- dizziness
- dupuytren's contracture
- dyslexia
- ear ache
- ear infection
- eczema
- edema
- emotional stress
- emphysema
- emulsification of fat
- endometriosis
- epistaxis
- epstein - barr virus
- esophagitis
- exercise recovery
- eye conditions
- facet syndrome
- facial paralysis
- fever
- fever blisters
- fibromyalgia
- flu
- food intolerance
- food poisoning
- foot fungus
- fracture
- fungus
- gait
- gallbladder (general)
- gallbladder (stones)
- ganglion cyst
- general musculoskeletal
- gerd
- gingivitis
- glaucoma
- goiter
- gout
- gums
- headache
- heart
- heartburn
- hearing difficulty
- hemorrhoids
- hepatitis A
- hepatitis B
- hepatitis C
- hernia
- herpes simplex
- herpes zoster (chickenpox / varicella)
- HIV
- hives
- hoarseness
- hormone balance
- hot flashes
- human papilloma virus (HPV)
- hyperactivity
- hyper/hypo-tension
- hyper/hypo-thyroid
- hyper/hypo-gycemia
- impotence
- immune enhancement
- incontinence
- indigestion
- infection
- inflammatory bowel disease
- inflammation
- influenza
- injuries
- insect bites
- irritable bowel syndrome
- ischemia
- jaundice
- joints
- keloid
- kidney
- kidey stones
- large intestine
- laryngitis
- ligament
- liposuction
- liver (balace and support)
- loss of smell (anosmia)
- loss of taste
- low back pain
- lungs
- lyme disease
- lymphadentis
- lymphatic
- macular degeneration
- memory problems
- meniere's disease
- meniscus sprain (grade 1)
- menopause
- mensturation
- mental fatigue
- meridian balance 15
- migraine
- motion sickness
- multiple sclerosis
- muscle
- muscle spasm
- myocardial inrarction
- nerve root
- neurogenic inflammation
- neuropathy
- nervousness
- nose bleed
- numbness
- nystagmus
- ocular motility disorders
- ocular nerve
- olfactory nerve
- osgood-schlatter disease
- otitis
- pain
- pain (chronic)
- pain (general)
- injury related pain (localized)
- pain (acute injury)
- pancreas
- parasite
- parasympathetic facilitazation
- paresthesia (numbness)
- periodontal disease
- pink eye (conjunctivitis)
- plantar fasciitis
- pneumonia
- polycystic kidney diseases
- polycystic ovary
- post operative scar revision
- post operative wound healing / pain
- post traumatic stress disorder (PTSD)
- postnasal drip
- premenstral syndrome (PMS)
- pre set head PL-touch
- pre-op
- prostate
- psoriasis
- punctures
- rash
- reflex sympathetic dystrophy (RSD)
- renal problems
- respiratory problems
- restless leg syndrome
- retinitis pigmentosa
- rheumatism
- ringworm
- road rash
- scar tissue
- sciatica
- sedation
- seizures
- shingles
- sinusitis
- skin
- sleep apnea
- small intesine
- smell - lack of
- sore throat
- soreness
- spasm
- spider veins
- spleen
- sprains
- spurs
- standars (neurological) setting
- stanard (up-regulation) setting
- staph infection
- stings
- stomach ulcer
- strep infections
- stress
- stroke
- sty
- subluxation
- sunburns
- swimmer's ear
- swollen ankles
- sympathetic calming
- tachycardia
- taste - lack of
- teeth
- tendonmyopathy (tendonitis)
- tension headaches
- theta waves
- thoratic outlet syndrome
- throat
- thrush
- thyroid (hyper)
- thyroid (hypo)
- tinnitus
- TMJ
- toenail fungus
- tonsilitis
- toothache
- ulcer
- ulcerative colotis
- up-regulation
- urinary tract infection
- varicose veins
- veins
- venereal warts
- viral infections
- voice
- vomiting
- water retention
- watery discharge from eye
- warts
- wounds
- yeast
Original Source: http://www.coldlasers.org/lllt-books/
Abstract
Photobiomodulation (PBM) also known as low-level level laser therapy is the use of red and near-infrared light to stimulate healing, relieve pain, and reduce inflammation. The primary chromophores have been identified as cytochrome c oxidase in mitochondria, and calcium ion channels (possibly mediated by light absorption by opsins). Secondary effects of photon absorption include increases in ATP, a brief burst of reactive oxygen species, an increase in nitric oxide, and modulation of calcium levels. Tertiary effects include activation of a wide range of transcription factors leading to improved cell survival, increased proliferation and migration, and new protein synthesis. There is a pronounced biphasic dose response whereby low levels of light have stimulating effects, while high levels of light have inhibitory effects. It has been found that PBM can produce ROS in normal cells, but when used in oxidatively stressed cells or in animal models of disease, ROS levels are lowered. PBM is able to up-regulate anti-oxidant defenses and reduce oxidative stress. It was shown that PBM can activate NF-kB in normal quiescent cells, however in activated inflammatory cells, inflammatory markers were decreased. One of the most reproducible effects of PBM is an overall reduction in inflammation, which is particularly important for disorders of the joints, traumatic injuries, lung disorders, and in the brain. PBM has been shown to reduce markers of M1 phenotype in activated macrophages. Many reports have shown reductions in reactive nitrogen species and prostaglandins in various animal models. PBM can reduce inflammation in the brain, abdominal fat, wounds, lungs, spinal cord.
2.1. Cytochrome c oxidase in mitochondria
Cytochrome c oxidase (CCO) is unit IV in the mitochondrial electron transport chain. It transfers one electron (from each of four cytochrome c molecules), to a single oxygen molecule, producing two molecules of water. At the same time the four protons required, are translocated across the mitochondrial membrane, producing a proton gradient that the ATP synthase enzyme needs to synthesize ATP. CCO has two heme centers (a and a3) and two copper centers (CuA and CuB). Each of these metal centers can exist in an oxidized or a reduced state, and these have different absorption spectra, meaning CCO can absorb light well into the NIR region (up to 950 nm) [9]. Tiina Karu from Russia was the first to suggest [10,11], that the action spectrum of PBM effects matched the absorption spectrum of CCO, and this observation was confirmed by Wong-Riley et al in Wisconsin [12]. The assumption that CCO is a main target of PBM also explains the wide use of red/NIR wavelengths as these longer wavelengths have much better tissue penetration than say blue or green light which are better absorbed by hemoglobin. The most popular theory to explain exactly why photon absorption by CCO could led to increase of the enzyme activity, increased oxygen consumption, and increased ATP production is based on photodissociation of inhibitory nitric oxide (NO) [13]. Since NO is non-covalently bound to the heme and Cu centers and competitively blocks oxygen at a ratio of 1:10, a relatively low energy photon can kick out the NO and allow a lot of respiration to take place [14].
2.2. Light gated ion channels and opsins
More recently it has become apparent that another class of photoreceptors, must be involved in transducing cellular signals, particularly responding to blue and green light. Thee photoreceptors have been proposed to be members of the family of light-sensitive G-protein coupled receptors known as opsins (OPN). Opsins function by photoisomerization of a cis-retinal co-factor leading to a conformational change in the protein. The most well known opsin is rhodopsin (OPN1), which is responsible for mediating vision in the rod and cone photoreceptor cells in the mammalian retina. There are other members of the opsin family (OPN2-5), which are expressed in many other tissues of the body including the brain [15]. One of the best-defined signaling events that occurs after light-activation of opsins, is the opening of light-gated ion channels such as members of the transient receptor potential (TRP) family of calcium channels [16]. TRP channels are now known to be pleiotropic cellular sensors mediating the response to a wide range of external stimuli (heat, cold, pressure, taste, smell), and involved in many different cellular processes [17]. Activation of TRP causes non-selective permeabilization (mainly of the plasma membrane) to calcium, sodium and magnesium [18]. It is now known that TRP channel proteins are conserved throughout evolution and are found in most organisms, tissues, and cell-types. The TRP channel superfamily is now classified into seven related subfamilies: TRPC, TRPM, TRPV, TRPA, TRPP, TRPML, and TRPN [19]. Light-sensitive ion channels are based on an opsin chromophore (isomerization of a cis-retinal molecule to the trans configuration) as illustrated in Drusophila photoreceptors [20].
We have shown that blue or green light (but not red or 810 nm NIR) increased intracellular calcium in adipose derived stem cells, that could be blocked by ion channel inhibitors [5].
2.3. Flavins and flavoproteins
There is another well-known family of biological chromophores called cryptochromes. These proteins have some sequence similarity to photolyases [21], which are blue light responsive enzymes that repair DNA damage in bacteria caused by UV exposure [22]. Cryptochromes rely on a flavin (flavin adenine dinucleotide, FAD) or a pterin (5,10-methenyltetrahydrofolic acid) to actually absorb the light (again usually blue or green). Cryptochromes have been studied mainly in plants and insects. Recent evidence has emerged that mammalian cryptochromes are important in regulation of the circadian clock. It is thought that human cryptochromes (CRY1 and CRY2) send signals via part of the optic nerve to the suprachiasmatic nucleus (SCN) in the brain, which is the master regulator of the CLOCK system to entrain biological responses to the light-dark cycle [23]. However the situation is complicated because retinal ganglion cells containing melanopsin (OPN4) are also involved in photoentrainment [24]. Studies are still ongoing to investigate this redundancy [25].
It should be emphasized that compared to CCO and mitochondria, evidence is still emerging concerning the extent to which opsins, cryptochomes and light-gated ion channels (which may be widely expressed in many different cell types) could be responsible for PBM effects. If their role is significant it is likely to be in the blue and green spectral regions. Further research will be necessary to explore their role in anti-inflammatory effects, wound healing and tissue regeneration.
2.4. Water as a chromophore and heat-gated ion channels
Since the biological effects of light continue to be observed, as the wavelength increases in the infra-red region (>1000 nm), beyond those known to be absorbed by CCO, it is now thought likely that an alternative chromophore must be responsible. The obvious candidate for this alternative chromophore is water molecules whose absorption spectrum has peaks at 980 nm, and also at most wavelengths longer than 1200 nm. Moreover, water is by the far the most prevalent molecule in biological tissue (particularly considering its low molecule weight = 18). At present the proposed mechanism involves selective absorption of IR photons by structured water layers (also known as interfacial water) [26] or water clusters [27], at power levels that are insufficient to cause any detectable bulk-heating of the tissue. A small increase in vibrational energy by a water cluster formed in or on a sensitive protein such as a heat-gated ion channel, could be sufficient to perturb the tertiary protein structure thus opening the channel and allowing modulation of intracellular calcium levels [28]. Pollack has shown that interfacial water can undergo charge separation when it absorbs visible or NIR light [29]. This charge separation (equivalent to localized pH changes) could affect the conformation of proteins [30]. It has also been suggested that PBM could reduce the viscosity of interfacial water within the mitochondria, and allow the F0F1 ATP synthase, which rotates as a nanomotor to turn faster [31]. It should be noted here that the first regulatory approvals of PBM were gained as a 510 K device “equivalent to an non-heating IR lamp” [32]. While the involvement of water as a chromophore may still be considered hypothetical it is difficult to think of another explanation for the beneficial of PBM at wavelengths between 1000 nm all the way to 10,000 nm (carbon dioxide laser).
3.1. PBM increases ROS in normal cells
When PBM stimulates CCO activity in normal healthy cells, the resulting increase in mitochondrial membrane potential (MMP) above normal baseline levels, leads to a brief and rather modest increase in generation of reactive oxygen species (ROS) [33]. However this brief burst of ROS caused by 3 J/cm2 of 810 nm laser (Figure 2A) was shown to be sufficient to activate the redox-sensitive transcription factor, NF-kB in embryonic fibroblasts [34] (Figure 2B). Addition of the anti-oxidant N-acetyl-cysteine to the cells could block the NK-kB activation (Figure 2C), but not the increase in cellular ATP caused by the mitochondrial stimulation (Figure 2D). In primary cultured cortical neurons [35], 810 nm laser produced a biphasic dose response in ATP production (Figure 3A) and MMP (Figure 3B) with a maximum at 3 J/cm2. At a high dose (30 J/cm2) the MMP was actually lowered below baseline. Interestingly the dose-response curve between fluence (J/cm2) and ROS production showed two different maxima (Figure 3C). One of these maxima occurred at 3 J/cm2 where the MMP showed its maximum increase. The second maximum in ROS production occurred at 30 J/cm2 where the MMP had been reduced below baseline. At a value between these two fluences (10 J/cm2) a dose at which the MMP was approximately back to baseline, there was not much ROS generation. These data are very good examples of the “biphasic dose response” or “Arndt-Schulz curve” which is often discussed in the PBM literature [7,8].
Thus it appears that ROS can be generated within mitochondria when the MMP is increased above normal values and also when it is decreased below normal values. It remains to be seen whether these two kinds of PBM-generated ROS are identical or not. One intriguing possibility is that whether the ROS generated by PBM is beneficial or detrimental may depend on the rate at which it is generated. If superoxide is generated in mitochondria at a rate that allows superoxide dismutase (SOD) to detoxify it to hydrogen peroxide, then the uncharged H2O2 can diffuse out of the mitochondria to activate beneficial signaling pathways, while if superoxide is generated at a rate or at levels beyond the ability of SOD to deal with it, then the charged superoxide may build up inside mitochondria and damage them.
3.2. PBM reduces ROS in oxidative stressed cells and tissues
Notwithstanding, the ability of PBM to produce a burst of ROS in normal cells, it is well-accepted that PBM when as a treatment for tissue injury or muscle damage is able to reduce markers of oxidative stress [36,37,38]. How can these apparently contradictory findings be reconciled? A study attempted to answer this question [39]. Primary cultured cortical neurons were treated with one of three different interventions, all of which were chosen from literature methods of artificially inducing oxidative stress in cell culture. The first was cobalt chloride (CoCl2), which is used as a mimetic for hypoxia and works by a Fenton reaction producing hydroxyl radicals [40]. The second was direct treatment with hydrogen peroxide. The third was treatment with the mitochondrial complex I inhibitor, rotenone [41]. All three of these different treatments increased the intracellular mitochondrial ROS as judged by Cell-Rox Red (Figure 4A), and at the same time lowered the MMP as measured by tetramethyl-rhodamine methyl ester (TMRM) (Figure 4B). PBM (3 J/cm2 of 810 nm laser) raised the MMP back towards baseline, while simultaneously reducing the generation of ROS in oxidatively stressed cells (while slightly increasing ROS in normal cells). In control cells (no oxidative stress), PBM increased MMP above baseline and still produced a modest increase in ROS.
Since most laboratory studies of PBM as a therapy have looked at various animal models of disease or injury, it is not surprising that most workers have measured reduction in tissue markers of oxidative stress (TBARS) after PBM [36,42]. There have been a lot of studies looking at muscles. In humans, especially in athletes, high-level exercise produces effects in muscles characterized by delayed-onset muscle soreness, markers of muscle damage (creatine kinase), inflammation and oxidative stress.
One cellular study by Macedo et al [43] used muscle cells isolated from muscular dystrophy mice (mdx LA 24) and found that 5 J/cm2 of 830 nm increased the expression levels of myosin heavy chain, and intracellular [Ca2+]i. PBM decreased H2O2 production and 4-HNE levels and also GSH levels and GR and SOD activities. The mdx cells showed significant increase in the TNF-α and NFκB levels, which were reduced by PBM.
While it is highly likely that the effects of PBM in modulating ROS are involved in the anti-inflammatory effects of PBM, it would be dangerous to conclude that that is the only explanation. Other signaling pathways (nitric oxide, cyclic AMP, calcium) are also likely to be involved in reduction of inflammation.
As mentioned above we found [34] that PBM (3 J/cm2 of 810 nm laser) activated NF-kB in embryonic fibroblasts isolated from mice that had been genetically engineered to express firefly luciferase under control of an NF-kB promoter. Although it is well-known that NF-kB functions as a pro-inflammatory transcription factor, but on the other hand it is also well known that in clinical practice or in laboratory animal studies) PBM has a profound anti-inflammatory effect in vivo. This gives rise to another apparent contradiction that must be satisfactorily resolved.
4.2. PBM reduces levels of pro-inflammatory cytokines in activated inflammatory cells
Part of the answer to the apparent contradiction highlighted above, was addressed in a subsequent paper [44]. We isolated primary bone marrow-derived dendritic cells (DCs) from the mouse femur and cultured them with GM-CSF. When these cells were activated with the classical toll-like receptor (TLR) agonists, LPS (TLR4) and CpG oligodeoxynucleotide (TLR9), they showed upregulation of cell-surface markers of activation and maturation such as MHC class II, CD86 and CD11c as measured by flow cytometry. Moreover IL12 was secreted by CpG-stimulated DCs. PBM (0.3 or 3 J/cm2 of 810 nm laser) reduced all the markers of activation and also the IL12 secretion. Figure 5.
Yamaura et al [45] tested PBM (810 nm, 5 or 25 J/cm2) on synoviocytes isolated from rheumatoid arthritis patients. They applied PBM before or after addition of tumor necrosis factor-α (TNF-α). mRNA and protein levels of TNF-α and interleukins (IL)-1beta, and IL-8 were reduced (especially by 25 J/cm2).
Hwang et al [46] incubated human annulus fibrosus cells with conditioned medium obtained from macrophages (THP-1 cells) containing proinflammatory cytokines IL1β, IL6, IL8 and TNF-α. They compared 405, 532 and 650 nm at doses up to 1.6 J/cm2. They found that all wavelengths reduced IL8 expression and 405 nm also reduced IL6.
The “Super-Lizer” is a Japanese device that emits linear polarized infrared light. Imaoka et al [47] tested it against a rat model of rheumatoid arthritis involving immunizing the rats with bovine type II collagen, after which they develop autoimmune inflammation in multiple joints. The found reductions in IL20 expression in histological sections taken from the PBM-treated joints and also in human rheumatoid fibroblast-like synoviocyte (MH7A) stimulated with IL1β.
Lim et al [48] studied human gingival fibroblasts (HGF) treated with lipopolysaccharides (LPS) isolated from Porphyromonas gingivalis. They used PBM mediated by a 635 nm LED and irradiated the cells + LPS directly or indirectly (transferring medium from PBM treated cells to other cells with LPS). Both direct and indirect protocols showed reductions in inflammatory markers (cyclooxygenase-2 (COX2), prostaglandin E2 (PGE2), granulocyte colony-stimulating factor (GCSF), regulated on activated normal T-cell expressed and secreted (RANTES), and CXCL11). In the indirect irradiation group, phosphorylation of C-Raf and Erk1/2 increased. In another study [49] the same group used a similar system (direct PBM on HGF + LPS) and showed that 635 nm PBM reduced IL6, IL8, p38 phosphorylation, and increased JNK phosphorylation. They explained the activation of JNK by the growth promoting effects of PBM. Sakurai et al reported [50] similar findings using HGF treated with Campylobacter rectus LPS and PBM (830 nm up to 6.3 J/cm2) to reduce levels of COX2 and PGE2. In another study [51] the same group showed a reduction in IL1β in the same system.
4.3. Effects of PBM on macrophage phenotype
Another very interesting property of PBM is its ability to change the phenotype of activated cells of the monocyte or macrophage lineage. These cells can display two very different phenotypes depending on which pathological situation the cells are faced with. The M1 phenotype (classically activated) applies to macrophages that are faced with a situation in which bacteria or other pathogens need to be killed, or alternatively tumor cells need to be destroyed. Inducible nitric oxide synthase is a hallmark of the M1 phenotype and nitric oxide secretion is often measured. On the other hand the M2 phenotype (alternatively activated) applies to macrophages that are involved in disposal of cellular or protein debris and stimulation of healing by angiogenesis. The M2 phenotype produces arginase, an enzyme that inhibits NO production and allows them to produce ornithine, a precursor of hydroxyproline and polyamines [52]. The markers of these two phenotypes of activated macrophage have some aspects in common, but also show many aspects that are very different [53]. It should be noted that this concept of M1 and M2 activation states, applies to other specialized macrophage type cells that are resident in different tissues, such as microglia in brain [54], alveolar macrophages in lung [55], Kuppfer cells in liver [56], etc.
Fernandes et al used J774 macrophage-like cells activated with interferon-γ and LPS to produce a MI phenotype and compared 660 nm and 780 nm laser. They found that both wavelengths reduced TNF-α, COX-2 and iNOS expression, with the 780 nm being somewhat better [57]. Silva et al used RAW264.7 macrophages to test two wavelengths (660 nm and 808 nm) at a range of fluences (11-214 J/cm2) [58]. They found increases in NO release with 660 nm at the higher fluences. von Leden et al carried out an interesting study looking at the effects of PBM on microglia and their interaction with cortical neurons [59]. They used both primary microglia isolated from mouse brains and the BV2 mouse microglial cell line and compared four fluences (0.2, 4, 10, and 30 J/cm2, at 808 nm. Fluences between 4 and 30 J/cm2 induced expression of M1 markers in microglia. Markers of the M2 phenotype, including CD206 and TIMP1, were observed at lower energy densities of 0.2–10 J/cm2. In addition, co-culture of PBM or control-treated microglia with primary neuronal cultures demonstrated a dose-dependent effect of PBM on microglial-induced neuronal growth and neurite extension. This suggests that the benefits of PBM on neuroinflammation may be more pronounced at lower overall doses. The same group went on to show that M1 activated macrophages receiving PBM (660 nm laser) showed significant decreases in CCL3, CXCL2 and TNFα mRNA expression 4 h after irradiation [60]. However, 24 h after irradiation, M1 macrophages showed increased expression of CXCL2 and TNFα genes. M1 activated macrophages irradiated with 780 nm showed a significant decrease in CCL3 gene expression 4h after irradiation. These data could explain the anti-inflammatory effects of LLLT in wound repair.
This section will cover some of the most important medical indications where PBM has been shown in laboratory studies to be effective (at least partly) by its pronounced anti-inflammatory effects. Figure 6 shows a graphical summary of the anti-inflammatory applications of PBM in experimental animal models.
5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.
5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.
5.1. Wound healing
Many papers have demonstrated the efficacy of PBM in stimulating wound healing. In animal models these studies have generally been on acute wounds [61], while in clinical trials they are often been concerned with chronic non-healing wounds such as diabetic ulcers [62]. Gupta et al [63] tested PBM using a superpulsed 904 nm laser on burn wounds in rats. They found faster healing, reduced inflammation (histology), decreased expression of TNF-α and NF-kB, and up-regulated expression of VEGF, FGFR-1, HSP-60, HSP-90, HIF-1α and matrix metalloproteinases-2 and 9 compared to controls. It is intriguing to speculate that the effects of PBM on wound healing (especially the use of for chronic non-healing wounds) could involve both pro-inflammatory effects and anti-inflammatory effects. This seemingly contradictory statement may be possible due to the recent discovery of resolvins and protectins, which are multifunctional lipid mediators derived from omega-3 polyunsaturated fatty acids [64]. If resolvins were produced as a result of the brief acute inflammation induced by application of PBM to chronic wounds, then it has been already shown that resolvins can hasten the healing of diabetic wounds in mice [65]. Resolvins have been shown to reduce tumor necrosis factor-α, interleukin-1β, and neutrophil platelet-endothelial cell adhesion molecule-1 in a mouse burn wound model [66].
5.2. Arthritis
In humans, arthritis is most often caused by a degenerative process occurring in osteoarthritis, or an autoimmune process occurring in rheumatoid arthritis. Both are characterized by pronounced inflammatory changes in the joint and even systemically. Different animal models are produced to mimic these diseases, but a common approach is to inject the sterile preparation of yeast cell walls known as zymosan into the knee joints of rats.
Castano et al [67] used this zymosan-induced arthritis model to study the effects of two different fluences of 810 nm laser (3 and 30 J/cm2) delivered at two different power densities (5 and 50 mW/cm2). PBM was delivered once a day for 5 days commencing after zymosan injection, and the swelling in the knee was measured daily. Prostagladin E2 (PGE2) was measured in the serum. They found that 3 out of the 4 sets of parameters were approximately equally effective in reducing swelling and PGE2, but the ineffective set of parameters was 3 J/cm2 delivered at 50 mW/cm2 which only took 1 min of illumination time. The conclusion was, that the illumination time was important in PBM, and if that time was too short, then the treatment could be ineffective.
Moriyama et al [68] used a transgenic mouse strain (FVB/N-Tg(iNOS-luc) that had been engineered to express luciferase under control of the inducible nitric oxide synthase promoter, to allow bioluminescence imaging of PBM of the zymosal-induced arthritis model in mice knees. They compared the same fluence of 635, 660, 690, and 905 nm (CW0 and 905 nm (short pulse). Animals younger than 15 weeks showed mostly reduction of iNOS expression, while older animals showed increased iNOS expression. Pulsed 905 nm also increased iNOS expression.
Pallotta et al [69] used a model where carageenan was injected into the rat knee and tested 810 nm laser at 1, 3, 6 or 10 J/cm2. Rats were sacrificed after 6 or 12 hours and the joint tissue removed. PBM was able to significantly inhibit the total number of leukocytes, as well as the myeloperoxidase activity. Vascular extravasation was significantly inhibited at the higher dose of energy of 10 J. Gene expression of both COX-1 and 2 were significantly enhanced by laser irradiation while PGE2 production was inhibited. These apparently contradictory results require more study to fully explain.
5.3. Muscles
One of the most robust applications of PBM, is its effects on muscles [70,71]. PBM can potentiate muscular performance especially when applied to the muscles 3 hours before exercise [72]. PBM can also make exercise-training regimens more effective. It is not therefore surprising that PBM can also help to heal muscle injuries, not to mention reducing muscle pain and soreness after excessive exercise. Many of the animal studies that have been done have looked at markers of inflammation and oxidative stress in muscle tissue removed from sacrificed animals. For instance, Silveira et al [73] caused a traumatic muscle injury by a single blunt-impact to the rat gastrocnemius muscle. PBM (850 nm, 3 or 5 J/cm2) was initiated 2, 12, and 24 h after muscle trauma, and repeated for five days. The locomotion and muscle function was improved by PBM. TBARS, protein carbonyls, superoxide dismutase, glutathione peroxidase, and catalase, were increased after muscle injury, these increases were prevented by PBM. PBM prevented increases in IL-6 and IL-10 and reversed the trauma-induced reduction in BDNF and VEGF.
5.4. Inflammatory pain
There have been many studies that have looked at the effects of PBM on pain in animal models. Some studies have looked at sensitivity to pain [74] using the von Frey filaments (a graded set of fibers of increasing stiffness and when the animal feels the pressure it withdraws its foot [75]).
Some studies have looked at animal models of neuropathic pain such as the “spared nerve injury” [76]. This involves ligating two out of three branches of the sciatic nerve in rats and causes long lasting (>6 months) mechanical allodynia [77]. Kobelia Ketz et al found improvements in pain scores with PBM (980 nm applied to affected hind paw 1 W, 20 s, 41 cm above skin, power density 43.25 mW/cm2, dose 20 J). They also found lower expression of the proinflammatory marker (Iba1) in microglia in the dorsal root ganglion, gracile nucleus, dorsal column and dorsal horn. The M1/M2 balance of the macrophage phenotype was switched from M1 to M2 by PBM, as judged by relative staining with anti-CD86 (M1) and anti-CD206 (M2).
Martins et al looked at the effect of PBM on a model of inflammatory pain [42]. This involved injecting complete Freund's adjuvant (CFA) into the mouse paw, and produces hyperalgesia and elevated cytokine levels (TNF-α, IL-1β, IL-10). They found that LEDT (950-nm, 80 mW/cm2, 1, 2 or 4 J/cm2) applied to the plantar aspect of the right hind limb, reduced pain, increased the levels of IL-10 prevented TBARS increase in both acute and chronic phases, reduced protein carbonyl levels and increased SOD and CAT activity in the acute phase only.
5.5. Lung inflammation
Aimbire and his laboratory in Brazil have carried out several studies on the use of PBM to reduce acute lung inflammation (ALI) in various animal models. In a mouse model of lung inflammation caused either by inhalation of lipolysaccharide or intranasal administration of TNFα they analyzed the bronchoalveolar lavage fluid (BALF). PBM (660 nm, 4.5 J/cm2) was administered to the skin over the right upper bronchus 15 min after ALI induction. PBM attenuated the neutrophil influx and lowered TNFα in BALF. In alveolar macrophages, PBM increased cAMP and reduced TNFα mRNA.
They also studied a different model of ALI caused by intestinal ischemia and reperfusion (I/R), that produces an analogue of acute respiratory distress syndrome (ARDS) [78]. Rats were subjected to superior mesenteric artery occlusion (45 min) and received PBM (660 nm, 7.5 J/cm2) carried out by irradiating the rats on the skin over the right upper bronchus for 15 and 30 min, and rats were euthanized 30 min, 2, or 4 h later. PBM reduced lung edema, myeloperoxisdase activity, TNF-α and iNOS, LLLT increased IL-10 in the lungs of animals subjected to I/R.
A third animal model was related to asthma [79]. Mice were sensitized to ovalbumin (OVA), and then challenged by a single 15-min exposure to aerosolized OVA. PBM was applied as above (660 nm, 30 mW, 5.4 J). Bronchial hyper-responsiveness (as measured by dose response curves to acetylcholine) was reduced by PBM as well as reductions in eosinophils and eotaxin. PBM also diminished expression of intracellular adhesion molecule and Th2 cytokines, as well as signal transducer and activator of transduction 6 (STAT6) levels in lungs from challenged mice. Recently Rigonato-Oliveira et al. presented a study that concluded that the reduced lung inflammation and the positive effects of PBM on the airways appear to be mediated by increased secretion of the anti-inflammatory cytokine IL-10, and reduction of mucus in the airway [80].
5.6. Traumatic brain injury
In recent years the use of PBM as a treatment for traumatic brain injury [81,82], and other brain disorders including stroke, neurodegenerative diseases and even psychiatric disorders has increased markedly [83]. It is thought that the actions of NIR light shone on the head and penetrating into the brain are multi-factorial, but one clear effect is the anti-inflammatory action of transcranial PBM. This was shown by a series of mouse experiments conducted by Khuman et al [84]. They used the controlled cortical impact model of TBI and delivered PBM (800 nm) was applied directly to the contused parenchyma or transcranially in mice beginning 60–80 min after CCI. Injured mice treated with 60 J/cm2 (500 mW/cm2 × 2 min) had improved latency to the hidden platform and probe trial performance in the Morris water maze. PBM in open craniotomy mice reduced the number of activated microglia in the brain at 48 h (21.8 ± 2.3 versus 39.2 ± 4.2 IbA-1 + cells/field).
5.7. Spinal cord injury
Spinal cord injury (SCI) is another promising area of central nervous system injury that could be benefited by PBM. Veronez et al [85] used a rat model of SCI involving a contusion produced by a mechanical impactor (between the ninth and tenth thoracic vertebrae), with a pressure of 150 kdyn. Three different doses of PBM (808-nm laser) were tested: 500 J/cm2, 750 J/cm2 and 1000 J/cm2 delivered daily for seven days. Functional preformance and tactile sensitivity were improved after PBM, at 1000 J/cm2. PBM at 750 and 1000 J/cm2 reduced the lesion volume and also reduced markers of inflammation (lower CD-68 protein expression).
5.8. Autoimmune diseases
Experimental autoimmune encephalomyelitis (EAE) is the most commonly studied animal model of multiple sclerosis (MS), a chronic autoimmune demyelinating disorder of the central nervous system. Immunomodulatory and immunosuppressive therapies currently approved for the treatment of MS slow disease progression, but do not prevent it. Lyons et al [86] studied a mouse model of EAE involving immunization with myelin oligodendrocyte glycoprotein (MOG35-55). They treated the female C57BL/6 mice with PBM (670 nm) for several days in different regimens. In addition to improved muscular function, they found down-regulation of inducible nitric oxide synthase (iNOS) gene expression in the spinal cords of mice as well as an up-regulation of the Bcl-2 anti-apoptosis gene, an increased Bcl-2:Bax ratio, and reduced apoptosis within the spinal cord of animals over the course of disease. 670 nm light therapy failed to ameliorate MOG-induced EAE in mice deficient in iNOS, confirming a role for remediation of nitrosative stress in the amelioration of MOG-induced EAE by 670 nm mediated photobiomodulation.
5.9. Abdominal fat
Yoshimura et al [87] looked at a mouse model of obesity and type 2 diabetes [87]. Four weeks old male adult C57BL/6 mice were fed a hypercaloric high-fat diet (40% calories derived from fat) for eight weeks to induce obesity and hyperglycemia. Over a period of four weeks mice were exposed to six irradiation sessions using an 843 nm LED (5.7 J cm−2, 19 mW cm−2). Non-irradiated control mice had areas of inflammation in their abdominal fat almost five times greater than the PBM group. The PBM group had significantly lower blood glucose levels 24 hours after the last session.
Amongst the many hundreds of reports of clinical applications of PBMT, we will highlight a few here, which seem to be especially relevant to inflammation, and inflammatory disorders.
6.1. Achilles tendinopathy
Bjordal et al in Norway carried out a randomized, placebo controlled trial of PBM (904 nm, 5.4 J per point, 20 mW/cm2) for activated Achilles tendinitis [88]. In addition to clinical assessment, they used microdialysis measurement of peritendinous prostaglandin E2 concentrations. Doppler ultrasonography measurements at baseline showed minor inflammation shown by increased intratendinous blood flow, and a measurable resistive index. PGE2 concentrations were significantly reduced with PBM vs placebo. The pressure pain threshold also increased significantly.
6.2. Thyroiditis
Chavantes and Chammas in Brazil have studied PBM for chronic autoimmune thyroiditis. An initial pilot trial [89] used 10 applications of PBM (830 nm, 50 mW, 38–108 J/cm2), twice a week, using either the punctual technique (8 patients) or the sweep technique (7 patients). Patients required a lower dosage of levothyroxine, and showed an increased echogenicity by ultrasound. The next study [90] was a randomized, placebo-controlled trial of 43 patients with a 9-month follow-up. In addition to improved thyroid function they found reduced autoimmunity evidenced by lower thyroid peroxidase antibodies (TPOAb), and thyroglobulin antibodies (TgAb). A third study [91] used color Doppler ultrasound to show improved normal vascualrization in the thyroid parenchyma. Finally [92] they showed a statistically significant increase in serum TGF-β1 levels 30 days post-intervention in the PBM group, thus confirming the anti-inflammatory effect. Recently a long-term follow up study of these thyroiditis patients (6 years later) was presented showing that PBM was safe in the long term and demonstrated lasting benefits [93].
6.3. Muscles
PBM for muscles aims to benefit athletic performance and training, to reduce delayed onset muscle soreness (DOMS), as well as to ameliorate signs of muscle damage (creatine kinase) after intense or prolonged exercise. Moreover PBM can also be used to treat frank muscle damage caused by muscle strains or trauma. The International Olympic Committee and the World Anti-Doping Agency cannot ban light therapy for athletes considering (1) the intensity is similar to sunlight, and (2) there is no forensic test for light exposure. There have been several clinical trials carried out in Brazil in athletes such as elite runners [94], volleyball players [95] and rugby players [96]. Ferraresi et al conducted a case-controlled study in a pair of identical twins [97]. They used a flexible LED array (850 nm, 75 J, 15 sec) applied to both quadriceps femoris muscles (real to one twin and sham to the other) immediately after each strength training session (3 times/wk for 12 weeks) consisting of leg press and leg extension exercises with load of 80% and 50% of the 1-repetition maximum test, respectively. PBM increased the maximal load in exercise and reduced fatigue, creatine kinase, and visual analog scale (DOMS) compared to sham. Muscle biopsies were taken before and after the training program and showed that PBM decreased inflammatory markers such as interleukin 1β and muscle atrophy (myostatin). Protein synthesis (mammalian target of rapamycin) and oxidative stress defense (SOD2, mitochondrial superoxide dismutase) were up-regulated.
6.4. Psoriasis
Psoriasis is a chronic autoimmune skin disease. Psoriasis is characterized by the abnormally excessive and rapid growth of keratinocytes (instead of being replaced every 28–30 days as in normal skin, in psoriatic skin they are replaced every 3–5 days). This hyperproliferation is caused by an inflammatory cascade in the dermis involving dendritic cells, macrophages, and T cells secreting TNF-α, IL-1β, IL-6, IL-17, IL-22, and IL-36γ [98]. PBM has been used for psoriasis because of its anti-inflammatory effects, which is a different approach from UV phototherapy which tends to kill circulating T-cells. Ablon [99] tested PBM using LEDs (830 nm, 60 J/cm2 and 633 nm, 126 J/cm2) in two 20-min sessions over 4 or 5 weeks, with 48 h between sessions in 9 patients with chronic treatment-resistant psoriasis. Clearance rates at the end of the follow-up period ranged from 60% to 100%. Satisfaction was universally very high.
Choi et al [100] tested PBM in case report of a patient with another inflammatory skin disease called acrodermatitis continua, who also had a 10-yr history of plaque psoriasis on her knees and elbows. As she was pregnant and not suited for pharmacological therapy, she received treatment with PBM (broad-band polarized light, 480–3,400 nm, 10 J/cm2). In two weeks (after only 4 treatments), the clinical resolution was impressive and no pustules were found. Topical methylprednisolone aceponate steroid cream was switched to a moisturizer, and she was treated twice or once a week with PBM until a healthy baby was delivered.
6.5. Arthritis
As can be seen from the animal studies section, arthritis is one of the most important clinical indications for PBM [101,102]. The two most common forms of arthritis are osteoarthritis (degenerative joint disease that mostly affects the fingers, knees, and hips) and rheumatoid arthritis (autoimmune joint inflammation that often affects the hands and feet). Osteoarthritis (OA) affects more than 3.8% of the population while rheumatoid arthritis (RA) affects about 0.24%. Both types have been successfully treated with PBM. Cochrane systematic reviews found for good evidence for its effectiveness in RA [103], and some evidence in the case of OA [104]. Most clinical studies have used pain scales and range of movement scores to test the effectiveness, rather than measures of inflammation which are difficult to carry out in human subjects.
Barabas and coworkers [105] made an attempt by testing PBM on ex vivo samples of synovial tissue surgically removed from patients receiving knee joint replacement. Synovial membrane samples received exposure to PBM (810 nm, 448 mW, 25 J/cm2, 1 cm2 area). PBM caused an increase in mitochondrial heat shock protein 1 60 kD, and decreases in calpain small subunit 1, tubulin alpha-1C, beta 2,vimentin variant 3, annexin A1, annexin A5, cofilin 1,transgelin, and collagen type VI alpha 2 chain precursor all significantly decreased compared to the control
6.6. Alopecia areata
Alopecia areata (AA) is one of the three common types of hair loss, the other two being androgenetic alopecia (AGA, male pattern baldness) and chemotherapy induced alopecia. AA is a common autoimmune disease resulting from damage caused to the hair follicles (HFs) by T cells. Evidence of autoantibodies to anagen stage HF structures is found in affected humans and experimental mouse models. Biopsy specimens from affected individuals demonstrate a characteristic peri- and intrafollicular inflammatory infiltrate around anagen-stage HFs consisting of activated CD4 and CD8 T lymphocytes [106]. PBM is an excellent treatment for hair loss in general and AGA in particular [107,108]. Yamazaki et al [109] reported the use of the “Super-Lizer” delivering linear-polarized light between 600–1600 nm at a power of 1.26 W to the areas of hair loss on the scalp (4-s pulses delivered at 1-s intervals for 3 min every 1 or 2 weeks until hair growth was observed). Regrowth of vellus hairs was achieved on more than 50% ofthe involved areas in all 15 cases. The frequency of irradiation until regrowth ranged from one to 14 times and the duration of SL treatment was 2 weeks to 5 months.
7. Conclusion and Future Studies
The clinical applications of PBM have been increasing apace in recent years. The recent adoption of inexpensive large area LED arrays, that have replaced costly, small area laser beams with a risk of eye damage, has accelerated this increase in popularity. Advances in understanding of PBM mechanisms of action at a molecular and cellular level, have provided a scientific rationale for its use for multiple diseases. Many patients have become disillusioned with traditional pharmaceutical approaches to a range of chronic conditions, with their accompanying distressing side-effects and have turned to complementary and alternative medicine for more natural remedies. PBM has an almost complete lack of reported adverse effects, provided the parameters are understood at least at a basic level. The remarkable range of medical benefits provided by PBM, has led some to suggest that it may be “too good to be true”. However one of the most general benefits of PBM that has recently emerged, is its pronounced anti-inflammatory effects. While the exact cellular signaling pathways responsible for this anti-inflammatory action are not yet completely understood, it is becoming clear that both local and systemic mechanisms are operating. The local reduction of edema, and reductions in markers of oxidative stress and pro-inflammatory cytokines are well established. However there also appears to be a systemic effect whereby light delivered to the body, can positively benefit distant tissues and organs.
There is a lot of scope for further work on PBM and inflammation. The intriguing benefits of PBM on some autoimmune diseases, suggests that this area may present a fertile area for researchers. There may be some overlap between the ability of PBM to activate and mobilize stem cells and progenitor cells, and its anti-inflammatory action, considering that one of the main benefits of exogenous stem cell therapy has been found to be its anti-inflammatory effect. The versatile benefits of PBM on the brain and the central nervous system, encourages further study of its ability to reduce neuroinflammation. Chronic diseases of the modern age involving systemic inflammation such as type II diabetes, obesity, Alzheimer's disease, cardiovascular disease and endothelial dysfunction are again worth investigating in the context of PBM.
Original Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5523874/
Original Source: https://pubmed.ncbi.nlm.nih.gov/31647776/
Abstract
Multiple sclerosis (MS) is an autoimmune demyelinating inflammatory disease characterized by recurrent episodes of T cell-mediated immune attack on central nervous system (CNS) myelin, leading to axon damage and progressive disability. The existing therapies for MS are only partially effective and are associated with undesirable side effects. Low-level laser therapy (LLLT) has been clinically used to treat inflammation, and to induce tissue healing and repair processes. However, there are no reports about the effects and mechanisms of LLLT in experimental autoimmune encephalomyelitis (EAE), an established model of MS. Here, we report the effects and underlying mechanisms of action of LLLT (AlGaInP, 660 nm and GaAs, 904 nm) irradiated on the spinal cord during EAE development. EAE was induced in female C57BL/6 mice by immunization with MOG35–55 peptide emulsified in complete Freund’s adjuvant. Our results showed that LLLT consistently reduced the clinical score of EAE and delayed the disease onset, and also prevented weight loss induced by immunization. Furthermore, these beneficial effects of LLLT seem to be associated with the down-regulation of NO levels in the CNS, although the treatment with LLLT failed to inhibit lipid peroxidation and restore antioxidant defense during EAE. Finally, histological analysis showed that LLLT blocked neuroinflammation through a reduction of inflammatory cells in the CNS, especially lymphocytes, as well as preventing demyelination in the spinal cord after EAE induction. Together, our results suggest the use of LLLT as a therapeutic application during autoimmune neuroinflammatory responses, such as MS.
Introduction
Multiple sclerosis (MS) is an inflammatory chronic autoimmune and neurodegenerative disorder of the human central nervous system (CNS), in which encephalitogenic Th1 and Th17 lymphocytes induce a response against components of myelin [1–3 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747
Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145
Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407 ]. Inflammation, gliosis and axonal injury are additional prominent neuropathological characteristics, as is the clinical evolution from intermittent attacks to slow, steady progressive worsening [4 Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, some evidence points to an important role for nitric oxide (NO) in the pathogenesis of MS and to its contribution to the various facets of the disorder, including inflammation, oligodendrocytes injury, changes in synaptic transmission, axonal degeneration and neuronal death [5 Smith, K. J., and H. Lassmann. 2002. The role of nitric oxide in multiple sclerosis. Lancet Neurol. 1: 232–241[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
Patients with MS typically present between the ages of 20 and 40 years, with affected women outnumbering men 2:1, and the progressive phase of disease manifests at any time between 5 and 35 years after onset [4 Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. MS causes a multitude of symptoms, including visual disturbances, spasticity, weakness, impairment of walking, coordination difficulties, tremor/ataxia, sensory problems and bladder disturbances [1 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747[Crossref], [PubMed], [Web of Science ®], [Google Scholar],6 McFarland, H. F., and R. Martin. 2007. Multiple sclerosis: a complicated picture of autoimmunity. Nat. Immunol. 8: 913–919[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, “invisible” symptoms, such as fatigue, neuropathic pain and cognitive deficits, are also common [7–9 Shi, J., C. B. Zhao, T. L. Vollmer, et al. 2008. APOE epsilon 4 allele is associated with cognitive impairment in patients with multiple sclerosis. Neurology 70: 185–190
Rao, S. M., G. J. Leo, L. Bernardin, and F. Unverzagt. 1991. Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41: 685–691
Rao, S. M. 1995. Neuropsychology of multiple sclerosis. Curr. Opin. Neurol. 8: 216–220 ] and are detectable even before a definitive diagnosis of MS is made. These sensorial, cognitive and emotional symptoms related to MS strongly impact on family, social and work activities, as well as their quality of life [10 Engel, C., B. Greim, and U. K. Zettl. 2007. Diagnostics of cognitive dysfunctions in multiple sclerosis. J. Neurol. 254: II30–II34[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. MS manifests in several forms, like as: (i) clinically isolated syndrome (CIS) is the first manifestation of MS-like signs and symptoms, usually followed by another attack at which a clinical diagnosis of MS is made; (ii) relapsing remitting MS (RRMS), which is characterized by sudden relapses punctuated by short- or long-term remissions; (iii) secondary progressive MS (SPMS), which has a progressive course resulting in severe, irreversible debilitation and (iv) primary progressive MS (PPMS), which is a progressive type of MS without an initial relapsing and remitting period [11 Confavreux, C., and S. Vukusic. 2006. Natural history of multiple sclerosis: a unifying concept. Brain J. Neurol. 129: 606–616[Crossref], [Web of Science ®], [Google Scholar]]. The economic cost of MS associated with relapses and subsequent disability is considerable. For instance, a multicenter study initially carried out in five European countries examined the costs associated with MS, and the annual cost for those with expanded disability status scale (EDSS) ≤3 ranged from E 13.534 to E 22.561 increasing to E 28.524–E 43.984 for EDSS 4–6.5 and E 39.592–E 65.395 for EDSS ≥7 [12 Karampampa, K., A. Gustavsson, C. Miltenburger, and B. Eckert. 2012. Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from five European countries. Mult. Scler. 18: 7–15[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and loss of earnings was the biggest contributor to indirect costs [13 O'Connell, K., S. B. Kelly, E. Fogarty, et al. 2014. Economic costs associated with an MS relapse. Mult. Scler. Relat. Disord. 3: 678–683[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In this context, phototherapy, especially laser, has been widely used in research of different tissues, such as tendons, nerves, skin tissue, bones, muscles and CNS [14–18 Baroni, B. M., R. Rodrigues, B. B. Freire, et al. 2015. Effect of low-level laser therapy on muscle adaptation to knee extensor eccentric training. Eur. J. Appl. Physiol. 115: 639–647
Barbosa, R. I., A. M. Marcolino, R. R. de Jesus Guirro, et al. 2010. Comparative effects of wavelengths of low-power laser in regeneration of sciatic nerve in rats following crushing lesion. Lasers Med. Sci. 25: 423–430
Batista, J. D., S. Sargenti-Neto, P. Dechichi, et al. 2015. Low-level laser therapy on bone repair: is there any effect outside the irradiated field? Lasers Med. Sci. 30: 1569–1574
Allahverdi, A., D. Sharifi, M. A. Takhtfooladi, et al. 2015. Evaluation of low-level laser therapy, platelet-rich plasma, and their combination on the healing of Achilles tendon in rabbits. Lasers Med. Sci. 30: 1305–1313
Hartzell, T. L., R. Rubinstein, and M. Herman. 2012. Therapeutic modalities – an updated review for the hand surgeon. J. Hand Surg. 37: 597–621 ].
Low-level laser therapy (LLLT) has been considered as an adjuvant clinical treatment [19 Carrasco, T. G., M. O. Mazzetto, R. G. Mazzetto, and W. MestrinerJr. 2008. Low intensity laser therapy in temporomandibular disorder: a phase II double-blind study. Cranio 26: 274–281[PubMed], [Google Scholar],20 Gavish, L., L. S. Perez, P. Reissman, and S. D. Gertz. 2008. Irradiation with 780 nm diode laser attenuates inflammatory cytokines but upregulates nitric oxide in lipopolysaccharide-stimulated macrophages: implications for the prevention of aneurysm progression. Lasers Surg. Med. 40: 371–378[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and its photomodulating, analgesic and direct interference effects on the neuroinflammatory process have drawn the attention of many researchers. LLLT can modulate a broad-spectrum of cellular processes, including: (i) protection from cell and tissue death; (ii) stimulation of healing and repair of injuries and (iii) reduction of pain, swelling and inflammation [21 Chung, H., T. Dai, S. K. Sharma, et al. 2012. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 40: 516–533[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. It has been reported that the effects of laser irradiation – called photobiomodulation – are related to light fluence [22 Wang, F., T. S. Chen, D. Xing, et al. 2005. Measuring dynamics of caspase-3 activity in living cells using FRET technique during apoptosis induced by high fluence low-power laser irradiation. Lasers Surg. Med. 36: 2–7[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Evidence suggests that red or near-infra-red light (at wavelengths that can penetrate tissue) is absorbed by mitochondrial chromophores, especially cytochrome c oxidase, leading to increased cellular respiration and ATP formation, and modulation of oxidative stress and NO production that together lead to the activation of signaling pathways and gene transcription [23 Chen, A. C., P. R. Arany, Y. Y. Huang, et al. 2011. Low-level laser therapy activates NF-kB via generation of reactive oxygen species in mouse embryonic fibroblasts. PLoS One 6: e22453[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The effect of LLLT on the brain has also been extensively investigated. Transcranially applied LLLT has been shown to have beneficial effects on Alzheimer’s disease (AD) mouse models, and on rats and rabbits post-stroke [24–26 Oron, A., U. Oron, J. Chen, et al. 2006. Low-level laser therapy applied transcranially to rats after induction of stroke significantly reduces long-term neurological deficits. Stroke 37: 2620–2624
De Taboada, L., J. Yu, S. El-Amouri, et al. 2011. Transcranial laser therapy attenuates amyloid-beta peptide neuropathology in amyloid-beta protein precursor transgenic mice. J. Alzheimers Dis. 23: 521–535
Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436 ]. Furthermore, LLLT-regulated microglial function through Src kinase – a non-receptor tyrosine kinase that is activated by oxidative events [27 Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] – and reduced long-term neurological deficits after traumatic brain injury (TBI) [28 Xuan, W., F. Vatansever, L. Huang, et al. 2013. Transcranial low-level laser therapy improves neurological performance in traumatic brain injury in mice: effect of treatment repetition regimen. PLoS One 8: e53454[Crossref], [PubMed], [Web of Science ®], [Google Scholar],29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. However, there have been no reports about the effects of irradiation on the autoimmune neuroinflammatory diseases, such as MS. Therefore, in the present study, we investigated the therapeutic potential of LLLT on experimental autoimmune encephalomyelitis (EAE) disease progression, an established model of MS. Most importantly, we attempted to elucidate some of the mechanisms through which LLLT modulates the pro-inflammatory environment of CNS.
Methods
Experimental animals
Experiments were conducted using female C57BL/6 mice (6–10 weeks of age). The mice were kept in groups of four to six animals per cage, maintained under controlled temperature (22 ± 1 °C) with a 12-h light/dark cycle (lights on at 07:00 h), and were given free access to food and water. All procedures used in the present study followed the Guide for the Care and Use of Laboratory Animals (NIH publication no. 85–23) and were approved by the Animal Ethics Committee of the Universidade Federal de Santa Catarina (CEUA-UFSC, protocol number PP00956) and Universidade do Extremo Sul Catarinense (CEUA-UNESC, protocol number 042/2014-1).
EAE induction and clinical evaluation
Active EAE was induced by subcutaneous immunization with 200 µg of myelin oligodendrocytes glycoprotein (MOG) peptide, amino acids 35–55 and 500 µg Mycobacterium tuberculosis extract H37Ra in complete Freund’s adjuvant oil, as previously described [30 Stromnes, I. M., and J. M. Goverman. 2006. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1: 1810–1819[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. All animals were also injected intraperitoneally on days 0 and 2 with 300 ng of Pertussis toxin. Non-immunized (naive) and EAE non-irradiated animals were used as controls. Mice were monitored and scored daily for clinical disease severity according to the standard 0–5 EAE grading scale: 0, unaffected; 1, tail limpness; 2, failure to right upon attempt to roll over; 3, partial paralysis; 4, complete paralysis and 5, moribund. The mean of the clinical scores and body weight (a parameter of health) of all mice within a given treatment group was determined daily, thereby yielding the mean clinical score and body weight change for that treatment group.
Laser treatment
The animals were randomly divided into four groups: (I) not immunized and untreated – naïve group; (II) immunized and untreated – EAE group; (III) immunized and treated with AlGaInP LLLT (660 nm) and (IV) immunized and treated with GaAs LLLT (904 nm). A two-laser diode (Ibramed™, São Paulo, Brazil) was used with the following parameters: (i) 660-nm wavelength (AsGaInP), mean power of 30 mW, continuous regime and beam area of 0.06 cm2. The laser irradiation was delivered with a fluency of 10 J/cm2 and energy of 0.6 J, with exposure time of 20 s for each position; (ii) 904-nm wavelength (GaAs), peak power of 70 W, pulsed regime (time of pulse 60 ns) and beam area of 0.10 cm2. The laser irradiation was delivered with a fluency of 3 J/cm2. The animals were irradiated during 30 days (starting on day 0 until day 30 post-immunization), with a total of six position of irradiation per day – laser radiation was timed to contact in six points located 0.5 cm distance between the points. The laser focus was positioned on the spinal cord at an angle of 90° to the skin according to a contact-point technique, and the gauging of the laser emission was conducted before and after completion of the experiments.
Biochemical assays
Nitric oxide
NO release was quantified using the Griess assay [31 Pang, Q., X. Hu, X. Li, et al. 2015. Behavioral impairments and changes of nitric oxide and inducible nitric oxide synthase in the brains of molarless KM mice. Behav. Brain Res. 278: 411–416[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. After EAE induction, the production of NO was determined by an assay for nitrite. Eight mice of each group were euthanized, and the inguinal lymph nodes, spinal cords and spleen were extracted. The lymph node, spinal cord and spleen were rapidly separated on an ice plate and weighed. The samples were incubated with Griess reagent (1% sulfanilamide in 0.1 mol/L HCl and 0.1% N-(1-naphthyl) ethylenediamine dihydrochloride) at room temperature for 10 min, and optical density of the assay samples was measured spectrophotometrically at 540 nm.
Oxidative damage to lipids
The levels of 2-thiobarbituric acid-reactive species (TBARS) are expressed as malondialdehyde (MDA) equivalents, as previously described [32 Draper, H. H., and M. Hadley. 1990. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186: 421–431[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Briefly, spinal cord and inguinal lymph nodes were mixed with 1 mL of 10% trichloroacetic acid and 1 mL of 0.67% thiobarbituric acid. Subsequently, the mixture was heated in a boiling water bath for 15 min. The amount of TBARS was determined by measuring absorbance at 532 nm, and the results are given in nanomoles of TBARS per milligram of protein.
Glutathione (GSH) levels
Samples of spinal cord were collected and maintained at −80 °C for at least 48 h. The sample was homogenized with 200 μL of 0.02 M EDTA. The homogenate was mixed with 25 μL of 10% trichloroacetic acid, and was homogenized three times over 15 min, followed by centrifugation (15 min× 1500g × 4 °C). The supernatant was added to 200 μL of 0.2 M TRIS buffer, pH 7.4 and 500 μM DTNB. Color development resulting from the reaction between DTNB and thiols reached a maximum in 5 min and was stable for more than 30 min. Absorbance was read at 412 nm after 10 min. A standard GSH curve was formed. The results are expressed as GSH per mg of protein [33 Borghi, S. M., A. C. Zarpelon, F. A. Pinho-Ribeiro, et al. 2014. Role of TNF-alpha/TNFR1 in intense acute swimming-induced delayed onset muscle soreness in mice. Physiol. Behav. 128: 277–287[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
Histopathological examination and assessment
For histopathological analysis, 30 days after EAE induction, animals were sacrificed and each portion of the lumbar spinal cord (L3–L5) was removed and fixed immediately in 10% neutral formalin buffer [formalin:phosphate buffer (0.01 M, pH 7.4) = 1:1] for 24 h. The spinal cord portions were subsequently processed by routine paraffin embedding, sectioned (5 -μm thickness) and mounted on glass slides. A deparaffinization protocol was carried out through a xylene-free method as previously described [34 Falkeholm, L., C. A. Grant, A. Magnusson, and E. Moller. 2001. Xylene-free method for histological preparation: a multicentre evaluation. Lab. Invest. 81: 1213–1221[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Hematoxylin–eosin (H&E)- or luxol fast blue (LFB)-stained slides were observed for immune cell infiltration and demyelination area, respectively. The settings used for image acquisition were identical for both control and experimental tissues, and representative images are presented. Four ocular fields per section (six to nine mice per group) were captured and a threshold optical density that best discriminated the nuclear staining of inflammatory cells (hematoxylin-eosin) or myelin (luxol fast blue) was obtained using NIH ImageJ 1.36 b imaging software (NIH, Bethesda, MD) and applied to all experimental groups The total pixel intensity was determined, and the data are expressed as optical density (O.D.).
ELISA assay
Spinal cord segments were homogenized in phosphate buffer containing 0.05% Tween® 20, 0.1 mM phenylmethylsulphonyl fluoride, 0.1 mM benzethonium chloride, 10 mM EDTA and 20 UI aprotinin A. The homogenate was centrifuged at 3000g for 10 min, and the supernatants were stored at −70 °C until further analysis. IFN-γ, IL-17 and IL-1β levels were estimated with ELISA kits from R&D Systems (Minneapolis, MN) according to the manufacturer’s recommendations.
Drugs and reagents
Pertussis toxin, phosphate-buffered saline (PBS) and complete Freund’s adjuvant oil were all purchased from Sigma Chemical Co. (St. Louis, MO). The MOG35–55 peptide (MEVGWYRSPFSRVVHLYRNGK) was obtained from EZBiolab (Carmel, IN) and the M. tuberculosis extract H37Ra from Difco Laboratories (Detroit, MI). The anti-mouse-IL-17, IFN-γ, IL-1β DuoSet kits were obtained from R&D Systems (Minneapolis, MN). Other reagents were all of analytical grade and were obtained from different commercial sources.
Statistical analysis
Results are presented as means ± SEM of measurements made on 6–9 mice per group per experiment, and are representative of one/two independent experiments without overlapping samples by evaluating the reproducibility of these results. One-way ANOVA followed by the Newman–Keuls test was used to compare the groups at each time-point when the parameters were measured at different times after the EAE induction. p values less than 0.05 (p < 0.05) were considered significant. The statistical analyses were performed using GraphPad Prism 4 Software (GraphPad Software Inc., San Diego, CA).
Results
LLLT alleviates symptoms and delays disease onset in EAE mice
C57BL/6 mice immunized with MOG35–55 developed EAE clinical symptoms after 7 days and reached a maximum mean clinical on day 30, when the incidence of clinical EAE was 100% and the average score was around 3.5 ± 0.5 (Figure 1A and Table 1). To test the prophylactic efficacy of laser during EAE, treatment starts from day 0 of induction. Compared with the untreated EAE group, AlGaInP 10 J/cm2 or GaAs 3 J/cm2 treatment significantly delayed disease onset (p < 0.001; Table 1) and decreased disease severity as measured by the mean maximal clinical score (2.0 ± 0.2 and 2.5 ± 0.5, respectively), with inhibition of 68 ± 2% (AlGaInP 10 J/cm2, Figure 1A and B) and 54 ± 5% (GaAs 3 J/cm2) (p < 0.0001; F = 48.05), based on the area under the curve (AUC), compared with the EAE-untreated group (Figure 1A and B; Table 1).
Figure 1. Low-level laser therapy attenuates the EAE disease process in C57BL/6 mice. Active EAE was induced in C57BL/6 mice by immunization with MOG35–55 on day 0. The clinical score (A), area under the curve (AUC) (B), body weight change (C) and delta (Δ) body weight gain or loss at the peak of disease (day 30 post-induction) (D) were evaluated in the naive group, the control group (EAE), in mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and in mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after immunization. The clinical symptoms were scored every day in a blinded manner and are expressed as the mean clinical score or as the AUC. Data points are presented as the mean ± SEM. Values of ##p < 0.001 versus naive group and **p < 0.001 versus EAE group (one-way ANOVA followed by post-hoc Newman–Keuls).

As previously described, animals with EAE tend to have a reduced body weight as a result of anorexia and deficient fluid uptake, which fit well with the severity of the clinical score [35 Mix, E., H. Meyer-Rienecker, and U. K. Zettl. 2008. Animal models of multiple sclerosis for the development and validation of novel therapies – potential and limitations. J. Neurol. 255: 7–14[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Next, we evaluated whether LLLT prevents the body weight change that is induced by EAE in mice. As expected, after EAE induction, a significant body weight loss was observed in the EAE mice compared with the naïve group (Figure 1C and D). Interestingly, a significant body weight gain was found in the EAE plus AlGaInP 10 J/cm2 (10 ± 2.5%; Figure 1D) group and the EAE plus GaAs 3 J/cm2 group (11 ± 3.0%; Figure 1D) (p < 0.01; F = 6.3) when compared with the EAE group.
LLLT down-regulates NO levels in the CNS and peripheral lymphoid tissue without affecting lipid peroxidation or the antioxidant defense during EAE
Excess amounts of NO are harmful for CNS function and are implicated in the pathophysiology of many neurologic diseases, such as MS, and the EAE model, in which NO is overproduced, mainly by innate immunity cells, such as macrophages and microglia [36–38 Ghasemi, M., and A. Fatemi. 2014. Pathologic role of glial nitric oxide in adult and pediatric neuroinflammatory diseases. Neurosci. Biobehav. Rev. 45: 168–182
Das, U. N. 2012. Is multiple sclerosis a proresolution deficiency disorder? Nutrition 28: 951–958
Miller, E. 2012. Multiple sclerosis. Adv. Exp. Med. Biol. 724: 222–238 ]. Thus, we investigated the effect of LLLT on the level of NO in the CNS and secondary lymphoid tissue of EAE-treated and untreated animals. In agreement with clinical signs, the concentration of NO in the spinal cord of the EAE mice was significantly increased (52 ± 25 µmol/mg of protein) compared with the control animals (Figure 2A). In contrast, both AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment down-regulated the NO level in the CNS of the EAE-treated animals, with a mean of 10 ± 5 and 15 ± 10 µmol/mg of protein, respectively (Figure 2A; p < 0.01; F = 7.15). Moreover, this upregulation was attenuated with LLLT (AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment) in the spleen tissue after EAE induction (p < 0.05 and p < 0.01 versus the healthy group; Figure 2C). However, compared with the untreated EAE group, LLLT did not significantly modulate NO in the lymph node (Figure 2B). In addition, LLLT failed to inhibit lipid peroxidation (Figure 3A and B; p < 0.08; F = 2.80 and p < 0.7; F = 0.38) or to restore the antioxidant defense (Figure 3C and D; p < 0.31; F = 1.28 and p < 0.45; F = 0.91) after EAE induction in the spinal cord and lymph node, respectively.
Figure 2. Low-level laser therapy selectively inhibits NO level in the CNS and peripheral lymphoid tissue of EAE mice. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA. The spinal lumbar cords (A), inguinal lymph nodes (B) and spleen (C) were obtained from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after immunization. The NO production was analyzed using the Griess assay. Data are presented as means ± SEM of 6–9 mice per group and are representative of two independent experiments. #p < 0.05 versus naïve group and **p < 0.001 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

Figure 3. Low-level laser therapy ameliorates EAE without affecting lipid peroxidation or the antioxidant defense. Animals were immunized with MOG35–55 peptide/CFA and pertussis toxin. Lumbar spinal cord and inguinal lymph node samples were collected from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm), 30 days after EAE induction for the determination of TBARS (panels A and B) and GSH (panels C and D) levels, respectively. Results are presented as means ± SEM of 6–9 mice/group, and are representative of two separate experiments.

LLLT limits the infiltration of immune cells to the CNS
The hallmark of EAE disease is the infiltration of inflammatory cells into the CNS, leading to neuronal and oligodendrocyte damage [39 Bogie, J. F., P. Stinissen, and J. J. Hendriks. 2014. Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol. 128: 191–213[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Therefore, we aimed to determine the effect of LLLT on the infiltration of inflammatory cells into the CNS after EAE induction. As shown in Figure 4, no inflammatory foci were detected in the naïve lumbar spinal cord; however, the untreated EAE mice showed profound infiltration of immune cells into the CNS, particularly in the white matter region (Figure 4A and B). Interestingly, treatment with AlGaInP 10 J/cm2 significantly reduced the infiltration of these inflammatory cells into the CNS (Figure 4A and B; p < 0.02; F = 4.36). In contrast, treatment with GaAs 3 J/cm2 only resulted in a moderate inhibition (Figure 4).
Figure 4. Low-level laser therapy blocks infiltration of mononuclear cells into the CNS during EAE pathology. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA plus pertussis toxin. At the peak of disease (day 30), animals were killed and the lumbar spinal cords from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm) were harvested for infiltration studies. Infiltration of mononuclear cells into spinal cords sections was examined by H&E staining (A), with magnification ×40, ×100 and ×400. Graphical representation of the inflammatory cells evaluated in the lumbar spinal cord (B). Specifically, four alternate 5 -µm sections (six to nine animals/group) of the white matter of the lumbar spinal cord were obtained between L4 and L6. Detail: inflammatory foci in the white matter after EAE induction. Data are presented as means ± SEM. #p < 0.05 versus naïve group and *p < 0.05 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

LLLT reduces the demyelination area
To investigate whether clinical improvement was accompanied by decreased neuropathology, we examined the demyelination area in longitudinal sections of the lumbar region of spinal cords by LFB staining 30 days post-immunization. Histological analysis of the spinal cord tissue sections from the healthy control mice showed an intact myelin sheath (Figure 5), whereas typical demyelination was observed in the EAE mice (Figure 5A and B). Again, AlGaInP 10 J/cm2 treatment remarkably attenuated CNS demyelination in the EAE mice (Figure 5A and B), while GaAs 3 J/cm2 failed to inhibit the demyelination area induced by EAE (Figure 5A and B). These data suggest the clinical relevance of LLLT, especially AlGaInP 10 J/cm2, in reducing EAE severity.
Figure 5. Low-level laser therapy inhibits CNS demyelination during EAE development. Active EAE was induced in the C57BL/6 mice with MOG35–55/CFA plus pertussis toxin. At the peak of disease (day 30), animals were killed and the lumbar spinal cords from the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm) were harvested for demyelination studies. Demyelination areas in spinal cord sections were examined by luxol fast blue (LFB) staining (A), with magnification ×40 and ×100. Graphical representation of the CNS demyelination in lumbar spinal cord (B). Specifically, four alternate 5 -µm sections (six to nine animals/group) of the white matter of the lumbar spinal cord were obtained between L4 and L6. Detail: CNS demyelination in the white matter after EAE induction. Data are presented as means ± SEM. #p < 0.05 versus naïve group and *p < 0.05 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).

LLLT attenuated production of pro-inflammatory cytokines during EAE pathology
To initiate CNS inflammation, myelin-specific T cells, especially Th17 and Th1 subsets, must be activated in the periphery, gain access to the CNS and then be reactivated by central APCs presenting self-antigen, initiating a cascade of events, including the secretion of cytokines/chemokines, which recruit macrophages to the sites of T-cell activation [3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, pro-inflammatory mediators secreted by macrophages/microglia, such as IL-1β, are important for both perpetuating inflammation and contributing to CNS tissue damage in EAE [40 Kuchroo, V. K., A. C. Anderson, H. Waldner, et al. 2002. T cell response in experimental autoimmune encephalomyelitis (EAE): role of self and cross-reactive antigens in shaping, tuning, and regulating the autopathogenic T cell repertoire. Ann. Rev. Immunol. 20: 101–123[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Here, pronounced increase in IL-17, IFN-γ and IL-1β levels was observed in the spinal cord after EAE-immunization (Figure 6). AlGaInP 10 J/cm2 and GaAs 3 J/cm2 treatment markedly inhibited the upregulation of IL-17 (Figure 6A), IFN-γ (Figure 6B) and IL-1β (Figure 6C) in the CNS after EAE induction.
Figure 6. Low-level laser therapy inhibits production of pro-inflammatory cytokines during EAE pathology. The spinal cord was extracted and processed to estimate the levels of IL-17 (A), IFN-γ (B) and IL-1β (C) by ELISA in the naive group, the control group (EAE), from mice pre-treated with AlGaInP 10 J/cm2 (660 nm) and from mice pre-treated with GaAs 3 J/cm2 (904 nm). Data are presented as means ± SEM of 6–9 mice per group. #p < 0.05 and ##p < 0.01 versus naïve group; *p < 0.05 and **p < 0.001 versus EAE group (one-way ANOVA with Newman–Keuls post-hoc test).
Discussion
MS is the prototypic autoimmune inflammatory disorder of the CNS for which no cure is presently known. T cells have a pivotal role in orchestrating the complex cascade of events in MS, which include chronic inflammation, primary demyelination and axonal damage. The adverse events associated with the widely used IFN-β, glatiramer acetate, fingolimod, and, more recently, dimethyl fumarate justifying the search for alternative and less detrimental therapies.
Herein, we show that LLLT reduced the clinical score of EAE and delayed the disease onset through down-regulation of NO levels in the CNS and peripheral lymphoid tissue. Interestingly, a significant body weight gain was found in the EAE plus AlGaInP group and the EAE plus GaAs group, when compared with the EAE group, which could be due to the modulation of leptin levels. In fact, recently, Burduli demonstrated that the combined treatment by means of low-intensity laser irradiation is accompanied by the normalization of the plasma leptin level, suppression of the inflammatory process and a significant improvement of the quality of life of the patients suffering from rheumatoid arthritis [41 Burduli, N. N., and N. M. Burduli. 2015. [The influence of intravenous laser irradiation of the blood on the dynamics of leptin levels and the quality of life of the patients presenting with rheumatoid arthritis]. Vopr. Kurortol. Fizioter. Lech. Fiz. Kult. 92: 11–13[PubMed], [Google Scholar]]. Therefore, further experiments are required to confirm whether or not LLLT modulates the leptin pathway during the development of EAE. In addition, these beneficial effects of LLLT seem to be associated with a block of the entry of the inflammatory cells (especially lymphocytes) into the CNS, as well as immune cell migration, the demyelinating process and production of pro-inflammatory cytokines, after EAE induction (see proposed scheme in Figure 7). These results are in accord with studies in rodent models demonstrating that LLLT: (i) improves cognitive functions in the progressive stages of a mouse model of AD [26 Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]; (ii) recovers short- and long-term (56 days) neurobehavioral functions and reduces brain lesion volume after TBI [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] and (iii) reduces the delayed-type hypersensitivity reaction to ovalbumin by down-regulation of pro-inflammatory mediators [42 Oliveira, R. G., A. P. Ferreira, A. J. Cortes, et al. 2013. Low-level laser reduces the production of TNF-alpha, IFN-gamma, and IL-10 induced by OVA. Lasers Med. Sci. 28: 1519–1525[Crossref], [PubMed], [Web of Science ®], [Google Scholar],43 de Oliveira, R. G., F. M. Aarestrup, C. Miranda, et al. 2010. Low-level laser therapy reduces delayed hypersensitivity reaction to ovalbumin in Balb/C mice. Photomed. Laser Surg. 28: 773–777[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Taken together, these studies demonstrate the biological effects of LLLT with different parameters, confirming the ample therapeutic window of LLLT in different clinical conditions. In the literature, there are a large number of experimental studies with LLLT, although few parameters are described in detail, which results in the comparison and consequent understanding of the mechanisms involved being difficult. In the present study, we used two wavelengths – 660 and 904 nm. In agreement with our data, Enwemeka reported that only 30% of published papers using LLLT reveal consistent information to determine the dose, or even reported inaccurate data [44 Enwemeka, C. S. 2008. Standard parameters in laser phototherapy. Photomed. Laser Surg. 26: 411[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Thus, further studies are required to verify the effectiveness of LLLT in MS.
Figure 7. Schematic representation of low-level laser therapy (LLLT) anti-inflammatory and immunosuppressive effects in an experimental model of MS. Preventive treatment with LLLT during the induction phase of EAE, an experimental model of MS, inhibits development and progression of disease, besides neuroinflammation and demyelinating process in the CNS. Together, LLLT immunomodulatory correlates to inhibition of NO and cytokines levels in the spinal cord after EAE induction. LLLT, low-level laser therapy; EAE, experimental autoimmune encephalomyelitis; MS, multiple sclerosis; BBB, blood–brain barrier; CNS, central nervous systems; MOG, myelin oligodendrocytes glycoprotein; Th cell, T helper lymphocyte.

It has been suggested that LLLT may affect the inflammatory system, although the basis for the immunosuppressive effects of LLLT is still unknown. It is possible that LLLT irradiation changes RNA expression at the level of mRNA or protein synthesis of pro-inflammatory mediators, such as IL-2, TNF-α, IFN-γ, ICAM-1 and COX-2, as well as enhancing peripheral endogenous opioid in rats [45–47 Mafra de Lima, F., M. S. Costa, R. Albertini, et al. 2009. Low level laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat bronchi segments in E. coli lipopolysaccharide-induced airway inflammation by a NF-kappaB dependent mechanism. Lasers Surg. Med. 41: 68–74
Hagiwara, S., H. Iwasaka, A. Hasegawa, and T. Noguchi. 2008. Pre-Irradiation of blood by gallium aluminum arsenide (830 nm) low-level laser enhances peripheral endogenous opioid analgesia in rats. Anesth. Analg. 107: 1058–1063
Matsumoto, M. A., R. V. Ferino, G. F. Monteleone, and D. A. Ribeiro. 2009. Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med. Sci. 24: 195–201 ], from immune cells. In fact, recently, Mozzati et al. demonstrated that superpulsed laser irradiation blocked down-regulation of IL-1β, IL-6, IL-10 and COX-2, and that this was associated with a reduction in the inflammatory process after tooth extraction [48 Mozzati, M., G. Martinasso, N. Cocero, et al. 2011. Influence of superpulsed laser therapy on healing processes following tooth extraction. Photomed. Laser Surg. 29: 565–571[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Specific wavelengths of light trigger different inflammatory pathways of immune cells, such as antigen-presenting cells (APCs, e.g. macrophages) [49 Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], which leads to increased infiltration into the tissues. The ability of macrophages to act as phagocytes is also modulated by the application of LLLT [49 Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The ability of LLLT to drain lymphatic cells can be explained by the direct effects of laser light on the production of cytokines, because laser light can penetrate to 50 mm below the tissue surface [50 Uebelhoer, N. S., and E. V. Ross. 2008. Introduction. Update on lasers. Semin. Cutan. Med. Surg. 27: 221–226[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Accumulated evidence now suggests that in the induction phase of EAE and MS disease (day 0–day 7), encephalitogenic T cells in the periphery become activated by a viral or another infectious antigen [1 Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Here, we hypothesize that LLLT applied during the induction phase of EAE increased phagocytic activity, and thus reduced antigen presentation in draining lymphatic cells and consistently inhibited activation of encephalitogenic Th1 and Th17 cells during the presentation of myelin antigens in peripheral lymphoid organs. Consequently, these cells failed to differentiate, proliferate and migrate to the CNS effectively, an effect that abrogated the development of EAE. In agreement with our data, Farfara et al. showed that laser-induced CD11b-positive phagocytotic monocyte cells were associated with a significant reduction of brain amyloid load following a short period of treatment [26 Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436[Crossref], [PubMed], [Web of Science ®], [Google Scholar]].
After peripheral activation, CD4+ T cells effectively enter the subarachnoid space by crossing the blood-cerebrospinal fluid (CSF) barrier in either the choroid plexus or the meningeal venules [2 Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145[Crossref], [PubMed], [Web of Science ®], [Google Scholar],3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, inside the CNS, the T cells are re-activated by MHC class II-expressing microglia, which express myelin epitopes [3 Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. These T cells are capable of producing pro-inflammatory mediators, such as cytokines and reactive oxygen species (ROS). The level of ROS, especially NO, is enhanced in MS [51 Koch, M., G. S. Ramsaransing, A. V. Arutjunyan, et al. 2006. Oxidative stress in serum and peripheral blood leukocytes in patients with different disease courses of multiple sclerosis. J. Neurol. 253: 483–487[Crossref], [PubMed], [Web of Science ®], [Google Scholar]] and consequently causes increased permeability of the blood–brain barrier (BBB) [52 Kuhlmann, C. R., R. Tamaki, M. Gamerdinger, et al. 2007. Inhibition of the myosin light chain kinase prevents hypoxia-induced blood–brain barrier disruption. J. Neurochem. 102: 501–507[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Similarly, opening of the BBB and oxidative stress are known to be involved in the pathogenesis of EAE, the animal model of MS [53 van Horssen, J., G. Schreibelt, J. Drexhage, et al. 2008. Severe oxidative damage in multiple sclerosis lesions coincides with enhanced antioxidant enzyme expression. Free Radic. Biol. Med. 45: 1729–1737[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In this study, we extended and enriched these findings by demonstrating that LLLT inhibited the NO level in the CNS and peripheral lymphoid tissue, especially, spleen after EAE induction. On the other hand, LLLT did not modulate the production of NO in the inguinal lymph nodes, which could be due to either the EAE mice having less NO-producing cells or to a decreased NO-producing capability on a per cell basis in the regional lymph nodes. Thus, future studies will need to clarify this hypothesis, as well as to investigate whether LLLT could modulate, directly, these cells in lymph nodes after EAE induction. Interestingly, the beneficial effect of LLLT can be partially explained based on the rapid elevation of ATP content, as previously demonstrated after laser irradiation in the ischemic heart [54 Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Furthermore, increases in total antioxidants, angiogenesis, heat-shock protein content and anti-apoptotic activity following LLLT were previously found for ischemic heart and skeletal muscles [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar],54 Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301[Crossref], [PubMed], [Web of Science ®], [Google Scholar],55 Avni, D., S. Levkovitz, L. Maltz, and U. Oron. 2005. Protection of skeletal muscles from ischemic injury: low-level laser therapy increases antioxidant activity. Photomed. Laser Surg. 23: 273–277[Crossref], [PubMed], [Web of Science ®], [Google Scholar]], and can be suggested as possible processes that are also attenuated by LLLT in the EAE model.
Additionally, much attention has been paid to therapeutic strategies aimed at controlling microglia-mediated neurotoxicity. Recently, it has been debated whether He–Ne (632.8 nm) LLLT can activate a number of signaling pathways, including MAPK/ERK, Src, Akt and RTK/PKCs signaling pathways [56 Zhang, J., D. Xing, and X. Gao. 2008. Low-power laser irradiation activates Src tyrosine kinase through reactive oxygen species-mediated signaling pathway. J. Cell. Physiol. 217: 518–528[Crossref], [PubMed], [Web of Science ®], [Google Scholar],57 Zhang, L., D. Xing, X. Gao, and S. Wu. 2009. Low-power laser irradiation promotes cell proliferation by activating PI3K/Akt pathway. J. Cell. Physiol. 219: 553–562[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. A study by Song et al. employed a microglial activation model (BV2 cells plus lipopolysaccharide) and evaluated the LLLT-induced neuroprotective effect. They found that LLLT prevents Toll-like receptor (TLR)-mediated pro-inflammatory responses in microglia, characterized by down-regulation of pro-inflammatory cytokine expression and NO production [27 Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Results reported here indicate, for the first time to our knowledge, that LLLT preventive treatment produced a marked reduction in inflammatory cell recruitment into the spinal cord and effectively prevented demyelination areas in the EAE mice. According to our data, the neuroinflammatory process results in neuronal injury that may impair function in the CNS, and these results suggest a neuroprotective effect of LLLT, which can be observed in terms of reduced EAE development and severity of clinical scores.
The present study also indicates a possible preferable mode of laser use for LLLT application after EAE immunization. The 660-nm wavelength (AsGaInP 10 J/cm2) in continuous-pulse mode demonstrated a better outcome in the percentage of mice showing complete recovery compared with the 904-nm wavelength (GaAs 3 J/cm2). In agreement with our data, Oron et al. (LLLT 808 nm, GaAlAs) described the superiority of the 100-Hz laser compared to the 600-Hz frequency after closed-head injury (CHI), and suggested that this difference may be associated with a resonance effect between pulsed light and brain waves (such as α-waves and θ-waves) [29 Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. In addition, there is a higher elevation in ATP content in the rabbit brain when laser energy was applied in the 100-Hz mode compared with 600 Hz [58 Lapchak, P. A., and L. De Taboada. 2010. Transcranial near infrared laser treatment (NILT) increases cortical adenosine-5′-triphosphate (ATP) content following embolic strokes in rabbits. Brain Res. 1306: 100–105[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Thus, we can propose that the AsGaInP 10-J/cm2 mode is perhaps the preferable mode with which to obtain a beneficial effect after autoimmune and neurodegenerative diseases, such as MS. Finally, in agreement with our data, Hudson et al. used the LLLT at 808 and 980 nm (1 W/cm2), which was projected through bovine tissue samples ranging in thickness from 18 to 95 mm and power density measurements were taken for each wavelength at the various depths. Thus, the authors concluded that 808 nm of light penetrates as much as 54% deeper than 980 nm light in bovine tissue, although we have not found any data with another tissue, such as bone, skin, nerves or MS [59 Hudson, D. E., D. O. Hudson, J. M. Wininger, and B. D. Richardson. 2013. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed. Laser Surg. 31: 163–168[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. Moreover, Byrnes et al. showed that LLLT at 810 nm can penetrate deep into the body and promote neuronal regeneration and functional recovery for spinal cord injury (SCI) [60 Byrnes, K. R., R. W. Waynant, I. K. Ilev, et al. 2005. Light promotes regeneration and functional recovery and alters the immune response after spinal cord injury. Lasers Surg. Med. 36: 171–185[Crossref], [PubMed], [Web of Science ®], [Google Scholar]]. The noninvasive nature and almost complete absence of side effects encourage further studies in neuroscience. Usually every laser and light source has a therapeutic window, narrow or wide for a particular lesion or condition. In laser treatment side effects can be divided into: (i) immediate effects, which occur either immediately or within a few minutes or hours of laser treatment, occasionally related to improper technique and rarely related to an accident; (ii) late effects, excess fluence leads to epidermal erythema, superficial burn or deep dermal burn with incident scarring depending on the extent of injury and pigmentary changes and (iii) sequelae, which rarely occurs. Importantly, these adverse effects can be prevented or minimised by proper patient and lesion selection, proper parameter selection, test shots and stepping down on fluence [61 Patil, U. A., and L. D. Dhami. 2008. Overview of lasers. Indian J. Plastic Surg. 41: S101–S113[Crossref], [PubMed], [Google Scholar]].
In summary, the present study indicates that LLLT applied daily post-EAE induction to C57BL/6 mice markedly inhibits clinical signs, neuroinflammation and oxidative damage induced by encephalitogenic T lymphocytes and microglia in the CNS. Thus, LLLT may be a promising non-pharmacological disease-modifying therapy for the treatment of autoimmune conditions, such as MS.
References
- Sospedra, M., and R. Martin. 2005. Immunology of multiple sclerosis. Ann. Rev. Immunol. 23: 683–747
,
- Steinman, L. 2007. A brief history of T(H)17, the first major revision in the T(H)1/T(H)2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13: 139–145
,
- Goverman, J. 2009. Autoimmune T cell responses in the central nervous system. Nat. Rev. Immunol. 9: 393–407
,
- Ransohoff, R. M., D. A. Hafler, and C. F. Lucchinetti. 2015. Multiple sclerosis – a quiet revolution. Nat. Rev. Neurol. 11: 134–142
,
- Smith, K. J., and H. Lassmann. 2002. The role of nitric oxide in multiple sclerosis. Lancet Neurol. 1: 232–241
,
- McFarland, H. F., and R. Martin. 2007. Multiple sclerosis: a complicated picture of autoimmunity. Nat. Immunol. 8: 913–919
,
- Shi, J., C. B. Zhao, T. L. Vollmer, et al. 2008. APOE epsilon 4 allele is associated with cognitive impairment in patients with multiple sclerosis. Neurology 70: 185–190
,
- Rao, S. M., G. J. Leo, L. Bernardin, and F. Unverzagt. 1991. Cognitive dysfunction in multiple sclerosis. I. Frequency, patterns, and prediction. Neurology 41: 685–691
,
- Rao, S. M. 1995. Neuropsychology of multiple sclerosis. Curr. Opin. Neurol. 8: 216–220
,
- Engel, C., B. Greim, and U. K. Zettl. 2007. Diagnostics of cognitive dysfunctions in multiple sclerosis. J. Neurol. 254: II30–II34
,
- Confavreux, C., and S. Vukusic. 2006. Natural history of multiple sclerosis: a unifying concept. Brain J. Neurol. 129: 606–616
,
- Karampampa, K., A. Gustavsson, C. Miltenburger, and B. Eckert. 2012. Treatment experience, burden and unmet needs (TRIBUNE) in MS study: results from five European countries. Mult. Scler. 18: 7–15
,
- O'Connell, K., S. B. Kelly, E. Fogarty, et al. 2014. Economic costs associated with an MS relapse. Mult. Scler. Relat. Disord. 3: 678–683
,
- Baroni, B. M., R. Rodrigues, B. B. Freire, et al. 2015. Effect of low-level laser therapy on muscle adaptation to knee extensor eccentric training. Eur. J. Appl. Physiol. 115: 639–647
,
- Barbosa, R. I., A. M. Marcolino, R. R. de Jesus Guirro, et al. 2010. Comparative effects of wavelengths of low-power laser in regeneration of sciatic nerve in rats following crushing lesion. Lasers Med. Sci. 25: 423–430
,
- Batista, J. D., S. Sargenti-Neto, P. Dechichi, et al. 2015. Low-level laser therapy on bone repair: is there any effect outside the irradiated field? Lasers Med. Sci. 30: 1569–1574
,
- Allahverdi, A., D. Sharifi, M. A. Takhtfooladi, et al. 2015. Evaluation of low-level laser therapy, platelet-rich plasma, and their combination on the healing of Achilles tendon in rabbits. Lasers Med. Sci. 30: 1305–1313
,
- Hartzell, T. L., R. Rubinstein, and M. Herman. 2012. Therapeutic modalities – an updated review for the hand surgeon. J. Hand Surg. 37: 597–621
,
- Carrasco, T. G., M. O. Mazzetto, R. G. Mazzetto, and W. MestrinerJr. 2008. Low intensity laser therapy in temporomandibular disorder: a phase II double-blind study. Cranio 26: 274–281
,
- Gavish, L., L. S. Perez, P. Reissman, and S. D. Gertz. 2008. Irradiation with 780 nm diode laser attenuates inflammatory cytokines but upregulates nitric oxide in lipopolysaccharide-stimulated macrophages: implications for the prevention of aneurysm progression. Lasers Surg. Med. 40: 371–378
,
- Chung, H., T. Dai, S. K. Sharma, et al. 2012. The nuts and bolts of low-level laser (light) therapy. Ann. Biomed. Eng. 40: 516–533
,
- Wang, F., T. S. Chen, D. Xing, et al. 2005. Measuring dynamics of caspase-3 activity in living cells using FRET technique during apoptosis induced by high fluence low-power laser irradiation. Lasers Surg. Med. 36: 2–7
,
- Chen, A. C., P. R. Arany, Y. Y. Huang, et al. 2011. Low-level laser therapy activates NF-kB via generation of reactive oxygen species in mouse embryonic fibroblasts. PLoS One 6: e22453
,
- Oron, A., U. Oron, J. Chen, et al. 2006. Low-level laser therapy applied transcranially to rats after induction of stroke significantly reduces long-term neurological deficits. Stroke 37: 2620–2624
,
- De Taboada, L., J. Yu, S. El-Amouri, et al. 2011. Transcranial laser therapy attenuates amyloid-beta peptide neuropathology in amyloid-beta protein precursor transgenic mice. J. Alzheimers Dis. 23: 521–535
,
- Farfara, D., H. Tuby, D. Trudler, et al. 2015. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J. Mol. Neurosci. 55: 430–436
,
- Song, S., F. Zhou, and W. R. Chen. 2012. Low-level laser therapy regulates microglial function through Src-mediated signaling pathways: implications for neurodegenerative diseases. J. Neuroinflamm. 9: 219
,
- Xuan, W., F. Vatansever, L. Huang, et al. 2013. Transcranial low-level laser therapy improves neurological performance in traumatic brain injury in mice: effect of treatment repetition regimen. PLoS One 8: e53454
,
- Oron, A., U. Oron, J. Streeter, et al. 2012. Near infrared transcranial laser therapy applied at various modes to mice following traumatic brain injury significantly reduces long-term neurological deficits. J. Neurotrauma 29: 401–407
,
- Stromnes, I. M., and J. M. Goverman. 2006. Active induction of experimental allergic encephalomyelitis. Nat. Protoc. 1: 1810–1819
,
- Pang, Q., X. Hu, X. Li, et al. 2015. Behavioral impairments and changes of nitric oxide and inducible nitric oxide synthase in the brains of molarless KM mice. Behav. Brain Res. 278: 411–416
,
- Draper, H. H., and M. Hadley. 1990. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 186: 421–431
,
- Borghi, S. M., A. C. Zarpelon, F. A. Pinho-Ribeiro, et al. 2014. Role of TNF-alpha/TNFR1 in intense acute swimming-induced delayed onset muscle soreness in mice. Physiol. Behav. 128: 277–287
,
- Falkeholm, L., C. A. Grant, A. Magnusson, and E. Moller. 2001. Xylene-free method for histological preparation: a multicentre evaluation. Lab. Invest. 81: 1213–1221
,
- Mix, E., H. Meyer-Rienecker, and U. K. Zettl. 2008. Animal models of multiple sclerosis for the development and validation of novel therapies – potential and limitations. J. Neurol. 255: 7–14
,
- Ghasemi, M., and A. Fatemi. 2014. Pathologic role of glial nitric oxide in adult and pediatric neuroinflammatory diseases. Neurosci. Biobehav. Rev. 45: 168–182
,
- Das, U. N. 2012. Is multiple sclerosis a proresolution deficiency disorder? Nutrition 28: 951–958
,
- Miller, E. 2012. Multiple sclerosis. Adv. Exp. Med. Biol. 724: 222–238
,
- Bogie, J. F., P. Stinissen, and J. J. Hendriks. 2014. Macrophage subsets and microglia in multiple sclerosis. Acta Neuropathol. 128: 191–213
,
- Kuchroo, V. K., A. C. Anderson, H. Waldner, et al. 2002. T cell response in experimental autoimmune encephalomyelitis (EAE): role of self and cross-reactive antigens in shaping, tuning, and regulating the autopathogenic T cell repertoire. Ann. Rev. Immunol. 20: 101–123
,
- Burduli, N. N., and N. M. Burduli. 2015. [The influence of intravenous laser irradiation of the blood on the dynamics of leptin levels and the quality of life of the patients presenting with rheumatoid arthritis]. Vopr. Kurortol. Fizioter. Lech. Fiz. Kult. 92: 11–13
,
- Oliveira, R. G., A. P. Ferreira, A. J. Cortes, et al. 2013. Low-level laser reduces the production of TNF-alpha, IFN-gamma, and IL-10 induced by OVA. Lasers Med. Sci. 28: 1519–1525
,
- de Oliveira, R. G., F. M. Aarestrup, C. Miranda, et al. 2010. Low-level laser therapy reduces delayed hypersensitivity reaction to ovalbumin in Balb/C mice. Photomed. Laser Surg. 28: 773–777
,
- Enwemeka, C. S. 2008. Standard parameters in laser phototherapy. Photomed. Laser Surg. 26: 411
,
- Mafra de Lima, F., M. S. Costa, R. Albertini, et al. 2009. Low level laser therapy (LLLT): attenuation of cholinergic hyperreactivity, beta(2)-adrenergic hyporesponsiveness and TNF-alpha mRNA expression in rat bronchi segments in E. coli lipopolysaccharide-induced airway inflammation by a NF-kappaB dependent mechanism. Lasers Surg. Med. 41: 68–74
,
- Hagiwara, S., H. Iwasaka, A. Hasegawa, and T. Noguchi. 2008. Pre-Irradiation of blood by gallium aluminum arsenide (830 nm) low-level laser enhances peripheral endogenous opioid analgesia in rats. Anesth. Analg. 107: 1058–1063
,
- Matsumoto, M. A., R. V. Ferino, G. F. Monteleone, and D. A. Ribeiro. 2009. Low-level laser therapy modulates cyclo-oxygenase-2 expression during bone repair in rats. Lasers Med. Sci. 24: 195–201
,
- Mozzati, M., G. Martinasso, N. Cocero, et al. 2011. Influence of superpulsed laser therapy on healing processes following tooth extraction. Photomed. Laser Surg. 29: 565–571
,
- Dube, A., H. Bansal, and P. K. Gupta. 2003. Modulation of macrophage structure and function by low level He–Ne laser irradiation. Photochem. Photobiol. Sci. 2: 851–855
,
- Uebelhoer, N. S., and E. V. Ross. 2008. Introduction. Update on lasers. Semin. Cutan. Med. Surg. 27: 221–226
,
- Koch, M., G. S. Ramsaransing, A. V. Arutjunyan, et al. 2006. Oxidative stress in serum and peripheral blood leukocytes in patients with different disease courses of multiple sclerosis. J. Neurol. 253: 483–487
,
- Kuhlmann, C. R., R. Tamaki, M. Gamerdinger, et al. 2007. Inhibition of the myosin light chain kinase prevents hypoxia-induced blood–brain barrier disruption. J. Neurochem. 102: 501–507
,
- van Horssen, J., G. Schreibelt, J. Drexhage, et al. 2008. Severe oxidative damage in multiple sclerosis lesions coincides with enhanced antioxidant enzyme expression. Free Radic. Biol. Med. 45: 1729–1737
,
- Oron, U., T. Yaakobi, A. Oron, et al. 2001. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation 103: 296–301
,
- Avni, D., S. Levkovitz, L. Maltz, and U. Oron. 2005. Protection of skeletal muscles from ischemic injury: low-level laser therapy increases antioxidant activity. Photomed. Laser Surg. 23: 273–277
,
- Zhang, J., D. Xing, and X. Gao. 2008. Low-power laser irradiation activates Src tyrosine kinase through reactive oxygen species-mediated signaling pathway. J. Cell. Physiol. 217: 518–528
,
- Zhang, L., D. Xing, X. Gao, and S. Wu. 2009. Low-power laser irradiation promotes cell proliferation by activating PI3K/Akt pathway. J. Cell. Physiol. 219: 553–562
,
- Lapchak, P. A., and L. De Taboada. 2010. Transcranial near infrared laser treatment (NILT) increases cortical adenosine-5′-triphosphate (ATP) content following embolic strokes in rabbits. Brain Res. 1306: 100–105
,
- Hudson, D. E., D. O. Hudson, J. M. Wininger, and B. D. Richardson. 2013. Penetration of laser light at 808 and 980 nm in bovine tissue samples. Photomed. Laser Surg. 31: 163–168
,
- Byrnes, K. R., R. W. Waynant, I. K. Ilev, et al. 2005. Light promotes regeneration and functional recovery and alters the immune response after spinal cord injury. Lasers Surg. Med. 36: 171–185
,
- Patil, U. A., and L. D. Dhami. 2008. Overview of lasers. Indian J. Plastic Surg. 41: S101–S113
,
Original Source: https://www-tandfonline-com.colorado.idm.oclc.org/doi/full/10.3109/08916934.2015.1124425?scroll=top&needAccess=true
Background: Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and β-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Aβ). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Aβ brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Abstract: Abstract Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and β-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Aβ). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Aβ brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24994540
Background: This paper focuses on brain mitochondrial respiration as a therapeutic target for neuroprotection and cognitive enhancement. We propose that improving brain mitochondrial respiration is an important future direction in research and treatment of Alzheimer's disease (AD) and other conditions associated with cognitive impairment and neurodegeneration. The central thesis is that supporting and improving brain mitochondrial respiration constitutes a promising neurotherapeutic principle, with potential applications in AD as well as in a wide variety of neuropsychological conditions. We propose three different interventional approaches to improve brain mitochondrial respiration based on (a) pharmacology, (b) photobiomodulation and (c) nutrition interventions, and provide detailed examples for each type of intervention. First, low-dose USP methylene blue is described as a pharmacological intervention that can successfully increase mitochondrial respiration and result in memory enhancement and neuroprotection. Second, transcranial low-level light/laser therapy with near-infrared light is used to illustrate a photobiomodulation intervention with similar neurometabolic mechanisms of action as low-dose methylene blue. Finally, a nutrition intervention to improve mitochondrial respiration is proposed by increasing ketone bodies in the diet. The evidence discussed for each intervention supports a fundamental neurotherapeutic strategy based on improving oxidative energy metabolism while at the same time reducing the pro-oxidant tendencies of the nervous system. Targeting brain mitochondrial respiration with these three types of interventions is proposed as part of a holistic neurotherapeutic approach to improve brain energy metabolism and antioxidant defenses. This strategy represents a promising new bioenergetics direction for treatment of AD and other neuropsychological disorders featuring cognitive impairment and neurodegeneration.
Abstract: Abstract This paper focuses on brain mitochondrial respiration as a therapeutic target for neuroprotection and cognitive enhancement. We propose that improving brain mitochondrial respiration is an important future direction in research and treatment of Alzheimer's disease (AD) and other conditions associated with cognitive impairment and neurodegeneration. The central thesis is that supporting and improving brain mitochondrial respiration constitutes a promising neurotherapeutic principle, with potential applications in AD as well as in a wide variety of neuropsychological conditions. We propose three different interventional approaches to improve brain mitochondrial respiration based on (a) pharmacology, (b) photobiomodulation and (c) nutrition interventions, and provide detailed examples for each type of intervention. First, low-dose USP methylene blue is described as a pharmacological intervention that can successfully increase mitochondrial respiration and result in memory enhancement and neuroprotection. Second, transcranial low-level light/laser therapy with near-infrared light is used to illustrate a photobiomodulation intervention with similar neurometabolic mechanisms of action as low-dose methylene blue. Finally, a nutrition intervention to improve mitochondrial respiration is proposed by increasing ketone bodies in the diet. The evidence discussed for each intervention supports a fundamental neurotherapeutic strategy based on improving oxidative energy metabolism while at the same time reducing the pro-oxidant tendencies of the nervous system. Targeting brain mitochondrial respiration with these three types of interventions is proposed as part of a holistic neurotherapeutic approach to improve brain energy metabolism and antioxidant defenses. This strategy represents a promising new bioenergetics direction for treatment of AD and other neuropsychological disorders featuring cognitive impairment and neurodegeneration. Copyright © 2013 Elsevier Inc. All rights reserved.
Methods: Copyright © 2013 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24316434
Original Source: https://www.scientificamerican.com/article/an-hour-of-light-and-sound-a-day-might-keep-alzheimers-at-bay/
Abstract
Alzheimer’s disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Background
Many diseases, including AD, are characterized with aberrant cellular activities resulting from increased oxidative stress and inflammation (1, 2). In AD, Aβ when aggregated to its oligomeric form has the most cytotoxic effects (3). Toxic Aβ increases oxidative stress and triggers inflammatory responses in glial cells through increased expression of inflammatory proteins, such as interleukin-1β (IL-1β) and inducible nitric oxide synthase (iNOS) (4).
Low level light with wavelengths ranging from 632.8 to 904 nm have been reported to attenuate inflammation and oxidative stress in a number of models (5, 6). For example, we have previously demonstrated that a low level energy laser at 632.8 nm attenuated oxidative stress and inflammation in primary astrocytes induced by Aβ (5). However, the use of laser or LED light as a clinical therapy for the treatment of AD brains is limited due to the low penetration of light in tissues (7).
In this study, we demonstrate a new light delivery method by using a recently engineered nanoparticle, BRET-Qdot. This light delivery nanoparticle has been used in a number of in vivo applications (8) and exploits the F?ster resonance energy transfer from an engineered enzyme, Renilla Reniformis luciferase (Luc8) (9). When activated with its substrate, coelenterazine-h (coel-h), Luc8 is known to emit at 480 nm to a CdSe or CdTe quantum dot, which then re-emits the light at 800 nm. A typical BRET-Qdot construct incorporates 3 to 6 Luc8 molecules conjugated to the surface of CdTe quantum dots (Figure 1). Conceptually, this light delivery approach could potentially resolve difficulties in providing light from external sources to the central areas of the brain.
Methods
To test the efficacy of BRET-Qdots (Zymera, San Jose, CA, USA) as proposed, we used primary cortical rat astrocytes (Invitrogen, Grand Island, NY, USA) as in our previous study (5). The maximum fluency and duration of the BRET-Qdots’ emitted light was observed using a PIXIS 512 CCD camera (Princeton Instruments, Trenton, NJ, USA) in conjunction with a cold mirror with a 0° AOI (Edmund Optics Inc., Barrington, NJ, USA) to reflect >90% visible light. Then, the BRET-Qdots were used as the pretreatment prior to a 5 μM Aβ (American Peptide, Sunnyvale, CA, USA) exposure to astrocyte, followed by the characterizations of superoxide anion accumulation using fluorescence microscopy of dihydroethidium (DHE), and the expressions of IL-1β and iNOS using Western blot analysis. Detailed methodological information can be found in the Supplementary Materials.
Results
BRET-Qdot with coel-h attenuates Aβ-induced superoxide anion production
We test if the superoxide anion induced by Aβ can be reduced by 800 nm-emitting BRET-Qdots which preceded Aβ treatment. Figure 2 shows that exposing astrocytes to 5 μM Aβ for 2 h increased superoxide anion production by 40% as compared to the untreated control. Light pre-treatment using 66.7 pM BRET-Qdots activated with 2.5 μM coel-h reduces Aβ-induced superoxide anion production to untreated control levels. Either BRET-Qdots or coel-h individually reduces superoxide anion production on average compared to Aβ-treated cultures, but not statistically significant.
BRET-Qdot with coel-h pretreatment attenuates Aβ-induced inflammatory marker expression
Similarly to the superoxide anion experiment, we investigate if the light pretreatment with 66.7 pM BRET-Qdot activated with 2.5 μM coel-h reduce the inflammation contributed by 5 μM Aβ as measured by IL-1β and iNOS expression levels (Figure 3). Incubating astrocytes with 5 μM Aβ for 18 h increased IL-1β and iNOS expression by 30 and 50%, respectively. BRET-Qdot-based light pre-treatment lowered Aβ-induced expression of IL-1β to that of the untreated control, while Aβ-induced iNOS expression was reduced to below untreated control levels. Neither BRET-Qdot nor coel-h alone showed the same reduction in IL-1β or iNOS expression.
Discussion
Our previous study with astrocytes showed that application of 632.8 nm laser caused a decrease in oxidative stress and inflammation (5). However, red to NIR light cannot be transmitted through the scalp to the brain more than a few centimeters (7). This makes non-invasive LLLT for AD nearly impossible.
In this present study, we demonstrated the ability of a light-generating nanoparticle to attenuate Aβ-induced oxidative stress and inflammatory responses in astrocytes. The application of this light delivery method based on light-generating nanoparticles demonstrated a novel method for applying LLLT.
Some studies on the application of quantum dots in biological systems may consider the possible toxic effects of quantum dots due to their heavy metal content, nanoparticulate nature, or coating composition . Nevertheless, investigations of quantum dots in vivo have shown retention of the material in tissues from days to weeks with no major complications (10). Here we also show that exposing astrocytes to BRET-Qdots with coel-h for 2 and 12 h did not significantly lower cell viability as compared with control. (see Supplementary Materials, Fig. S2)
Results here show the advantages of light-generating nanoparticles as a concept to be further developed for in vivo light-delivery for LLLT. CdSe/CdTe quantum dots may be used further in animal models as non-toxic, NIR-emitting nanoparticles become available. In addition, even though this study only covered Aβ-induced inflammation and oxidative stress, as studied in the AD pathology, the LLLT delivered by light-generating BRET-dots may also be applied to other cellular systems and diseases involving oxidative stress and inflammation, such as diabetes (6). A light-generating nanoparticle-based therapy could potentially create the ability to offer LLLT in an in vivo setting to reduce the onset of these other pernicious illnesses.
Intro: Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Background: Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT.
Abstract: Abstract Alzheimer's disease (AD) is the sixth leading cause of age-related death with no effective intervention yet available. Our previous studies have demonstrated the potential efficacy of Low Level Laser Therapy (LLLT) in AD cell models by mitigating amyloid-β peptide (Aβ)-induced oxidative stress and inflammation. However, the penetration depth of light is still the major challenge for implementing LLLT in animal models and in the clinical settings. In this study, we present the potential of applying Bioluminescence Resonance Energy Transfer to Quantum Dots (BRET-Qdots) as an alternative near infrared (NIR) light source for LLLT. Our results show that BRET-Qdot-emitted NIR suppresses Aβ-induced oxidative stress and inflammatory responses in primary rat astrocytes. These data provide a proof of concept for a nanomedicine platform for LLLT. FROM THE CLINICAL EDITOR: Low Level Laser Therapy has already been demonstrated to mitigate amyloid-β peptide induced oxidative stress and inflammation, a key driver of Alzheimer's disease. The major issue in moving this forward from cell cultures to live animals and potentially to human subjects is light penetration depth. In this novel study, BRET-Qdots were used as an alternative near infrared light source with good efficacy, paving the way to the development of a nanomedicine platform. Copyright © 2014 Elsevier Inc. All rights reserved.
Methods: Low Level Laser Therapy has already been demonstrated to mitigate amyloid-β peptide induced oxidative stress and inflammation, a key driver of Alzheimer's disease. The major issue in moving this forward from cell cultures to live animals and potentially to human subjects is light penetration depth. In this novel study, BRET-Qdots were used as an alternative near infrared light source with good efficacy, paving the way to the development of a nanomedicine platform.
Results: Copyright © 2014 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/24200521
12.1. INTRODUCTION
Significant changes in blood flow or in the integrity of cerebral vessels are believed to cause cerebrovascular disease (CVD) and to contribute to dementias including Alzheimer’s disease [1]. Stroke, the most serious form of CVD, is one of the leading causes of death and adult disability worldwide. Acute treatments for stroke, however, are severely limited. Neuroprotective drugs under development show promise at halting the ischemic cascade, but as yet, no such compound has received federal approval in the United States. One of the biggest limitations to this development is the lack of understanding of the mechanisms by which cerebral vessels react to factors such as ischemia, inflammation, blood pressure changes, metabolic demands, and trauma [2]. In order to address these fundamental questions, functional brain imaging techniques such as fMRI and intrinsic signal optical imaging (ISOI) have emerged as tools to visualize and quantify cerebral hemodynamics.
In the neuroscience community, ISOI has long been used to study the organization and functional architecture of different cortical regions in animals and humans [3–5] (see other chapters in this book). Three sources of ISOI signals that affect the intensity of diffusely reflected light derive from characteristic physiologic changes in the cortex. For functional neuronal activation, these have been observed to occur over a range of timescales, including (1) light scattering changes, both fast (over 10 s of milliseconds) and slow (i.e., > ~0.5 s) (2) early (~0.5–2.5 s) absorption changes from alterations in chromophore redox status, i.e., the oxy/deoxy-hemoglobin ratio (known as the “initial dip” period), and (3), slower (~2–10 s) absorption changes due to blood volume increase (correlated with the fMRI BOLD signal). Light scattering changes have been attributed to interstitial volume changes resulting from cellular swelling, organelle swelling due to ion and water movement, capillary expansion, and neurotransmitter release [6,7]. The slower absorption factors have been demonstrated to correlate with the changes in metabolic demand and subsequent hemodynamic cascades following neuronal activation [4,8,9].
Using animal models of acute and chronic brain injury, ISOI has been used to quantify the acute hemodynamic events in response to stroke, including focal ischemia and cortical spreading depression (CSD) [10–21]. Researchers have also used ISOI to locate and quantify the spatial extent of the stroke injury, including ischemic core, penumbra, and healthy tissue zones [18,22]. CSD also plays a key role in migraine headache, and recent laser speckle imaging studies have revealed the neurovascular coupling mechanism to the transmission of headache pain [23,24].
To fully understand the underlying mechanisms in vascular changes associated with cerebrovascular diseases such as stroke, an optical imaging technique that has the capability to rapidly separate absorption from scattering effects can enhance the information content of traditional ISOI, enabling (1) more accurate quantitation of hemodynamic function, (2) isolation of the electro-chemical changes characterized by light scattering, and (3) longitudinal chronic injury studies of function where structural reorganization due to neovascularization can cause significant alterations in scattering [25,26].
Quantitative diffuse optical methods [27] such as spatially-resolved reflectance, diffuse optical spectroscopy (DOS), and tomography (DOT), and diffuse correlation spectroscopy (DCS) possess exquisite sensitivity to these functional and structural alterations associated with brain injury, and have been applied to the study of CSD [11,15,28]. DOS and DOT utilize the near-infrared spectral region (600–1000 nm) to separate and quantify the multispectral absorption (μa) and reduced scattering coefficients (μs′), providing quantitative determination of several important biological chromophores such as deoxy-hemoglobin (HbR), oxy-hemoglobin (HbO2), water (H2O), and lipids. Concentrations of these chromophores represent the direct metrics of tissue function such as blood volume fraction, tissue oxygenation, and edema. Additionally, the scattering coefficient contains important structural information about the size and density of scatterers and can be used to assess tissue composition (exctracellular matrix proteins, cell nuclei, mitochondria) as well as follow the process of tissue remodeling (wound healing, cancer progression). DOS utilizes a limited number of source-detector positions, e.g., 1–2, but often employs broadband content in temporal and spectral domains [29]. In contrast, DOT typically utilizes a limited number of optical wavelengths (e.g., 2–6) and a narrow temporal bandwidth, but forms higher resolution images of subsurface structures by sampling a large number of source-detector “views.” To achieve maximal spatial resolution, the ideal DOT design would employ thousands of source-detector pairs and wavelengths. However, several engineering considerations including measurement time and instrument complexity currently limit the practicality of this approach.
In this chapter we present the basic principles of a new, noncontact quantitative optical imaging technology, modulated imaging (MI) [30–32], and provide examples of MI performance in 2 rat models of brain injury, cortical spreading depression (CSD) and stroke. MI enables both DOS and DOT concepts with high spatial (<1 mm) and temporal resolution (<1 s) in a simple, scan-free platform. MI is capable of both separating and spatially-resolving optical absorption and scattering parameters, allowing wide-field quantitative mapping of tissue optical properties. While compatible with time-modulation methods, MI alternatively uses spatially modulated illumination for imaging of tissue constituents. Periodic illumination patterns of various spatial frequencies are projected over a large area of a sample. The diffusely reflected image is modified from the illumination pattern due to the turbidity of the sample. Typically, sine-wave illumination patterns are used. The demodulation of these spatially modulated waves characterizes the modulation transfer function (MTF) of the material, and embodies the sample optical property information.
12.2. METHODS AND INSTRUMENTATION
12.2.1. Modulated Imaging Spectroscopy
The MI instrument platform was introduced originally by Cuccia et al. [30] Based on this design, we have developed a custom multispectral near-infrared (NIR) MI spectroscopy system capable of imaging between 650 and 1000 nm. A diagram of this system is shown in Figure 12.1.

FIGURE 12.1
Modulated imaging platform. QTH—quartz tungsten halogen; L1—aspheric condenser; H—hybrid hot mirror; DMD—digital micromirror device; L2—projection lens; L3—camera lens; LCTF—liquid crystal tunable (more...)
Broadband NIR illumination is provided by an intensity-stabilized 250 W quartz-tungsten-halogen (QTH) lamp (Oriel QTH Source with Light Intensity Controller, Newport Corporation-Oriel Instruments, Stratford, Connecticut). Light is collimated and refocused with a pair of aspheric F/#0.7 optical lens systems (Oriel Aspherab). A custom-sized 3.5 in square hybrid hot mirror (Reynard Corporation, i.e., R00670-00) was placed between the lenses to limit the illumination to wavelengths below 1000 nm. Light engine optics taken from a digital projector (NEC HT1000) serve to homogenize and direct the light onto a 0.7 in digital micromirror device (DMD Discovery™ 1100 with ALP Accessory Package, ViALUX, Germany). Grayscale spatial sinusoid patterns are projected at 400 Hz using the ViALUX software development toolkit, which generates the necessary pulse-width modulation of binary sub-frames to produce a specified grayscale bit-depth (1–8 bits). Finally, a fixed focal length (f = 100 mm) projection lens illuminates the tissue at a slight angle from normal with a 15 × 25 mm illumination field. Detection was performed at normal incidence using a CRI Nuance™ camera system, which combines a 12-bit CCD camera and a liquid crystal tunable filter (LCTF; λ = 650–1100 nm, Δλ = 10 nm). To avoid specular reflection, crossed linear polarizers are used in the illumination and detection arms. For this system, the former is a 1.5 in diameter NIR linear polarizer (Meadowlark Optics, VLM-200-IR-R) placed immediately after the projection lens, and the first stage of the Nuance LCTF serves as the latter. The DMD, CCD, and LCTF are controlled via USB by a laptop computer, and synchronized using LabVIEW software (LabVIEW 8, National Instruments), enabling fast acquisition of a series of patterns with various spatial frequencies.
12.2.2. SFD Measurement, Calibration, and Modeling
A detailed description of SFD measurement, calibration, and diffusion modeling is provided by Cuccia [32]. In this work, we modeled diffuse reflectance using a transport-based White Monte Carlo (WMC) method [33,34]. Previously, we have found that compared with Monte Carlo, (1) diffusion predictions over- and underestimate low- and high-frequency diffuse reflectance, respectively, and (2) the quantitative accuracy of diffusion degrades with decreasing albedo [32]. Due to the moderate albedo of brain tissue (μs′/μa ~ 10–20), we chose to analyze all brain data with the WMC approach. This homogeneous tissue model is a significant simplification of the multilayered rat brain, and more work is necessary to accurately model this complex system. We discuss further the consequences of our simple model in Section 12.2.5.
12.2.3. Optical Property Inversion Methods
In this chapter, we use two inversion methods to calculate the absorption and reduced scattering from measurements of diffuse reflectance. When high measurement precision is desired, we use a “sweep” in spatial frequency space, producing an overdetermined set of diffuse reflectance measurements, which can be fitted to our WMC forward model predictions using least-squares minimization. This method is performed for all spatially averaged region analysis of optical properties and chromophores. When increased acquisition and/or processing speed is desired, we alternatively use a rapid two-frequency lookup table method based on cubic spline interpolation [32]. This data can be achieved with a minimal 3-phase, single frequency image set (by demodulating and averaging the images to obtain AC and DC amplitude maps, respectively). On typical personal computers this approach is capable of millions of inverse lookup calculations per second, and is therefore used to calculate all high-resolution images including time sequences. The signal-to-noise ratio (and thus the measurement precision) of either approach is limited by the data sampling, with the two-frequency method having a lower precision with the tradeoff of higher acquisition and processing speed.
12.2.4. Spectral Analysis-Chromophore Calculation
The quantitative absorption coefficient is assumed to be a linear (Beer’s law) summation of individual chromophore absorption contributions:
where ci and ?i(λ) represent chromophore concentrations and molar extinction coefficients, respectively. Using reported extinction coefficients of HbO2/HbR35 and H2O,36 we can invert Equation 12.1 and calculate tissue chromophore concentration separately at each pixel by linear least-squares fitting to the multispectral absorption images. Total hemoglobin (HbT) and oxygen saturation (StO2) can then be calculated as HbT = HbR + HbO2 and StO2= HbO 2/(HbR + HbO2) * 100, respectively.
12.2.5. Optical Property Mapping: Resolution Versus Quantitation
On a pixel-by-pixel basis, diffuse reflectance versus spatial frequency is fitted to the WMC forward model to extract the local absorption and reduced scattering optical property contrast. This process is repeated for each wavelength, resulting in multi-spectral absorption and scattering spectra at each pixel. The measured contrast from discrete absorbers and scatterers on millimeter and submillimeter spatial scales, however, will possess partial volume effects in all three spatial dimensions. This is due to the physical light transport length scales in tissue, limiting the true x-y resolution of optical property contrast to many detector pixels [32]. This phenomenon is not unique to MI, but present in all planar reflectance imaging measurements of turbid media. Absorption and scattering are calculated using a homogeneous reflectance model, extracting a locally averaged sampling of optical property contrast. Based on simulations of the tissue MTF for varying optical properties [32], we expect the resulting image resolution to scale directly with the transport length, l* = (μa + μs′)− 1, and the spatial frequency of illumination. In this chapter, we place quantitative emphasis on average optical properties and chromophores measured over a field of view that is greater than l*. Spatial maps and videos of these parameters are displayed and referred to as “contrast maps,” with the caveat that high resolution features will exhibit degraded quantitative accuracy.
12.2.6. In Vivo Rat CSD Experiments
12.2.6.1. Animal Preparation
MI spectroscopy measurements were performed on an in vivo Wistar rat model with a thinned-skull preparation. All procedures were performed in accordance with approved IACUC protocol guidelines. The animals were anesthetized, placed in a stereotaxic frame, their skulls thinned and glass coverslip applied. This preparation is described in detail by Masino et al. [37] The resulting thinned skulls allowed direct imaging of the cortex over a 5 × 7 mm field-of-view (whisker barrel cortex, centered at the C2 location). In order to investigate the sensitivity of MI toward studying acute cortical injury, we induced cortical spreading depression (CSD) by applying 1 M KCl solution to the surface of the cortex through a perforated section of skull and dura, located approximately 3 mm above the camera’s imaging field.
12.2.6.2. MI Measurement Protocol
For each of three animals, our MI measurement protocol was twofold. Prior to CSD induction, baseline spatial modulation data were acquired at 6 spatial frequencies (3-phase projections each) from 0 to 0.26 mm−1, at 10 nm intervals over the entire range between 650 and 980 nm. Depending on the wavelength, image acquisition times ranged from 200 ms to 4 s, with total spectral imaging time of approximately 30 s per spatial pattern. The entire measurement (34 wavelengths, 3 phases, 6 frequencies) was repeated three times for statistical averaging yielding an entire measurement time of approximately 30 min.
Next, rapid dynamic measurements were performed, beginning 1 min prior to K+Cl− administration. Here, a significantly reduced data set was chosen in order to achieve high temporal resolution. Two spatial frequencies (0 and 0.26 mm−1) were acquired with three phase projection images, as described in Section 12.2.2, at each of four wavelengths (680, 730, 780, and 830 nm). The resulting 12 images took in total 6 s, permitting a repetition rate of 10 measurements per minute. The animals were followed for a period of 10 min for rats 1 and 2, and a period of 30 min for rat 3.
All images in this study were smoothed by 2D convolution with a Gaussian filter function (FWHM = 3 pixels), and baseline repetitions were averaged prior to data processing. Additionally, time-series data were post-processed by smoothing slightly in time (Gaussian FWHM of 2 timepoints = 12 s).
12.2.6.3. Spatial Frequency Sensitivity Analysis
Because of the differential absorption sensitivity at low and high frequencies, optimal optical property separation is achieved when a large range of frequencies is used [31]. In Figure 12.2a, we depict this differential sensitivity using diffuse reflectance (MTF) predictions versus frequency, increasing μa by 100% from 0.02 (black line) to 0.04mm−1 (gray line). This is done for two values of μs′, 0.6 (solid lines) to 1.2mm−1 (dashed lines), simulating a 100% change in scattering. Notice that the low frequencies have a significant reflectance change due to absorption, while high frequency reflectance remains nearly unchanged. Conversely, reflectance changes due to scattering are observed at all spatial frequencies. In Figure 12.2b, we further visualize this by plotting the reflectance sensitivity to 1% changes in absorption and scattering. Whereas DC reflectance is equivalently sensitive to a fractional change in either absorption or scattering, at high spatial frequencies absorption contrast is lost while scattering contrast is retained. For instance, notice that at our maximum measurement frequency of 0.26 mm−1 the reflectance is roughly 24 times more sensitive to scattering compared to absorption (ΔRd = 0.56 μs′ versus 0.024 * 10−3 for μa). This plays an important role in Section 12.3.2 during our discussion of dynamic scattering measurement.

FIGURE 12.2
(a) Reflectance contrast in absorption and scattering covering a typical range of brain optical properties. (b) The frequency-dependent sensitivity to absorption (black line) and scattering (gray line), respectively. Reflectance at fx = 0.26 mm−1 (more...)
In realistic heterogeneous tissues, a tradeoff exists between maximizing the frequency range for optical property accuracy and obtaining similar sampling volumes. As tissue is a low-pass spatial filter, high frequencies are attenuated quickly with depth. Using diffusion-based forward modeling, we have estimated mean sampling depths at 650 nm using measured average background optical properties of brain tissue. This was done by predicting the depth sensitivity to contrast from a planar perturbation in absorption, given a background fluence profile from spatial frequencies 0 and 0.26 mm−1. Based on these results, we observe qualitatively similar depth sampling, with mean depth sampling ranging between 2.5 mm and 1.2 mm (for fx = 0 and 0.26 mm−1, respectively). In all cases maximal sensitivity was found in the first 1–2 mm, where cortical hemodynamic changes occur.
12.3. RESULTS AND DISCUSSION
12.3.1. Baseline MI Spectroscopy
In Figure 12.3a we show a grayscale planar reflectance image of the cortical region of rat 1 at 650 nm. A dotted-line box denotes the region-of-interest (ROI) used for analysis, selected for its uniform illumination and the absence of cerebral bruising. The Monte Carlo-model fitting of spatial frequency data allows calculation of the absorption and reduced scattering coefficients. In Figure 12.3b we show the spatially averaged diffuse reflectance at 650 nm and the corresponding multi-frequency fit. Excellent agreement is observed between measurement data and the model-based fit, with derived μa and μs′ coefficients of 0.033 and 0.70 mm−1, respectively.

FIGURE 12.3
(a) Reflectance map for rat 1, showing the 3.8 × 5.9 mm region chosen for quantitative analysis. (b) Sample MTF reflectance data (squares) and fit (solid line) at 650 nm. (c) Recovered optical property maps (above) and corresponding image histogram (more...)
Analysis of multifrequency reflectance data separately at each pixel results in spatial maps of absorption and reduced scattering contrast. In Figure 12.3c, we plot the μa and μs′ maps recovered at 650 nm for rat 1. Note the strong absorption in the vein region, due to a large absorption by HbR at this wavelength. Below the images, we show histogram distributions of the corresponding quantitative maps above, indicating the degree of spatial variation in recovered optical properties. The mean and standard deviation for the pixel-wise μa and μs ′ were 0.030 ± 0.007 mm−1 and 0.63 ± 0.13 mm−1, respectively. These statistical results are in good agreement with the spatially averaged reflectance fit from Figure 12.3b, suggesting that our simple pixel-wise fitting approach yields optical properties similar to that calculated using a global analysis.
By mapping the absorption coefficient at multiple wavelengths, we can perform quantitative spectral imaging of tissue. In Figure 12.4, we summarize the baseline spectroscopy results for all three animals. In Figure 12.4a we show the μa (left) and μs′ (right) coefficients versus wavelength (circles) recovered from spatially averaged fitting. Data for rat 1 is shown in black (rat 2 in dark gray; rat 3 in light gray). Note the distinct spectral features in absorption, resulting from oxy- and deoxy-hemoglobin (HbO2, HbR), and water (H2O) absorption. The calculated scattering coefficient generally decays with increasing wavelength, and the results from a power law (μs ′ = A·λ(nm) −b, solid lines) fit are shown. A small residual coupling is observed between measured scattering and absorption spectral features. In particular, the scattering at the shortest and longest wavelengths appears to be underestimated by 5–10%, occurring where the corresponding absorption is highest (due to HbR and H2O, absorption features, respectively). Based on our experiments in layered tissue phantoms [38], we believe this effect is primarily due to frequency-dependent probing volumes in the presence of depth-heterogeneous structures.

FIGURE 12.4
(a) Average μa (left) and μs′(right) spectra over entire ROI (circles). HbO 2, HbR, and H2O concentrations are determined by subsequent least-squares fitting (solid lines) of molar extinction coefficients to the absorption. Data (more...)
Simultaneous linear fitting of the absorption to known extinction coefficients yields measures of chromophore concentration. Shown in Figure 12.4a, multispectral fitting (solid line) for rat 1 yields HbO2, HbR, H2O, HbT and StO2 values of 56.3 μM, 33.2 μM, 63.9%, 89.6 μM, and 56.3%, respectively. Tabulated results of chromophore values for all three animals are shown in Figure 12.4b. Lipid absorption near 930 nm was not apparent in the μa spectrum, and when included in the spectral analysis was not found to significantly affect the results. The small absorption “bump” at 900–910 nm is an artifact of imperfect phantom calibration due to the presence of a sharp, strong silicone absorption peak that is present in the phantom.
We note that the solution for chromophore concentration is well-determined when the number of wavelengths is at least equal to the number of chromophores. Therefore, as few as two wavelengths can be used to separate HbO2 and HbR (if a constant value of H2O is assumed). Repeating the above analysis with 780 and 830 nm only (assuming H2O = 65%) yields results for HbO2 and HbR within 10% of those from full spectral fitting. Repeating the above analyses using a simple diffusion-based model provided qualitatively similar results for absorption and scattering spectra, but in general was found to overestimate the absorption coefficient by 10–25%.
Absorption spectra at each pixel can be separately analyzed to yield spatial maps of local HbO2, HbR, and H2O distribution, shown in Figure 12.5. Notice the high concentration of HbR over the large superficial draining vessel (venous) regions, also reflected in the StO2 image, highlighting the effect of tissue oxygen extraction. Conversely, notice that the high albedo regions with less structural detail are highly oxygenated, with StO2 levels between 60 and 70%. Lastly, the H2O map reveals a relatively homogeneous distribution of water.

FIGURE 12.5
Chromophore fits to absorption spectra at each pixel yield maps of local HbO2, HbR, and H2O concentration (left). Total hemoglobin (HbT) and oxygen saturation (StO2) maps can then be calculated from HbO2 and HbR.
12.3.2. Dynamic MI Spectroscopy of CSD
We performed measurements of CSD in each of the three rats, as described in Section 12.2.3. The results are presented as follows. We first present data for a single animal, choosing rat 3 for its long observation period of 30 minutes. Three ROIs are selected for analysis, and baseline MI spectroscopy results are reported for each of these regions. Next, the observed dynamic time courses of diffuse reflectance, optical properties, and chromophore concentrations are shown for each ROI. We then present the full spatio-temporal dynamic contrast data for rat 3 (2D + time) in the form of “snapshot” images.
Figure 12.6 summarizes the baseline spectroscopy measurements for rat 3. In Figure 12.6a, we show three regions of interest superimposed on the DC reflectance map, chosen to highlight three different characteristic temporal profiles observed within the field of view. In Figure 12.6b we show the baseline spectral fits for each of these regions, and in Figure 12.6c we tabulate the resulting calculated chromophore concentrations. In general, Region A (black) is a high albedo region lacking any large blood vessels, whereas Regions B (dark gray) and C (light gray) include high-absorption blood vessels and mild cerebral bruising from surgery. These differences are apparent in their recovered absorption spectra and fits, with on average 27% higher HbT, and 32% lower saturation in the vascular regions. Also, 7% higher H2O is found in Regions B and C, which may indicate increased edema due to bruising.

FIGURE 12.6
Regionwise spectral analysis of rat 1 baseline data including the respective (A) ROIs, (B) spectral absorption data (circles) and fit (lines), and (C) tabulated recovered chromophore data for each region
In Figures 12.7–12.9 (for regions A–C, respectively), we present the temporal dynamics of CSD in each ROI of rat 3 as measured by MI. In part (a) of each figure, we plot the multispectral diffuse reflectance changes at fx = 0 mm−1 (DC, top) and fx = 0.26 mm−1 (AC, bottom). In part (b), we plot the recovered Δμa (top) and Δμs′ (bottom) optical properties at each wavelength. While absolute values of diffuse reflectance and optical properties are measured separately at each time point, for visualization purposes all data are displayed as a change from that prior to KCl administration. Absolute optical property values at t = 0 (not shown) demonstrate excellent agreement (~5–10%) with full multifrequency baseline data.

FIGURE 12.7
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx =.26 mm−1, bottom) for Region A of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) coefficients. (more...)

FIGURE 12.8
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx = 0.26 mm−1, bottom) for Region B of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) (more...)

FIGURE 12.9
(A) Multispectral diffuse reflectance at DC (fx = 0 mm−1, top) and DC (fx = 0.26 mm−1, bottom) for Region C of rat 3 over approximately 30 min. (B) Corresponding recovered multispectral absorption (top) and reduced scattering (bottom) (more...)
Looking first at the reflectance time courses of Figure 12.7a (Region A), we see in general a series of three CSD events over the 30 minutes, with each transient event occurring for approximately 4.3 minutes. The first event occurs at minute 2.9 after KCl application, indicating an initial latency between the insult and the first resulting spreading depression wave. Reflectance contrast is present in both DC and AC frequency components, but with markedly different signatures. Generally, the DC time course shows a slow, gradual decay, punctuated by sharp, wavelength-dependent spikes/dips (for short/long wavelengths, respectively). Alternatively, the AC signature contains three sets of transient dips consistent across all wavelengths, with final values leveling off progressively lower than baseline. Discussed in detail in the following paragraph, we believe these AC changes are due primarily a result of optical scattering and may be related to neuronal depolarization. The corresponding derived optical properties in Figure 12.7b reflects this, with μs′ trends tracking directly with the measured AC reflectance. As expected, μa trends reveal similar wavelength-dependence of the DC reflectance (with opposite polarity), reflecting changes in HbO2 and HbR.
In Section 12.2.3.3 we noted that the diffuse reflectance at fx = 0.26 mm−1 is 23 times more sensitive to scattering changes compared to absorption. In this context, we propose that the observed magnitude of the CSD-induced AC reflectance changes can only be explained by changes in optical scattering. To concretely illustrate this point, we pick as an example the observed 780 nm AC diffuse reflectance dip in Figure 12.7a at t = 3.7 min of -0.003. Here, the corresponding change in reduced scattering in Figure 12.7b, Δμs′, is calculated to be −0.03 mm−1. In order for this change to instead be due to an absorption-only event, μa would need to increase by 121% from baseline (from 0.038 to 0.084 mm−1). This increase would also need to be accompanied by a drop in Rd (fx = 0 mm−1) of 0.12 (33%), whereas the actual observed DC reflectance only drops by 0.008 (<1%) and thus cannot explain the change. Secondly, we note that the three sets of AC reflectance dips occur consistently across all four wavelengths. While an approximate 120% increase in HbT could induce this decrease at high frequency, it would also require a large broad-wavelength decrease in the DC reflectance. We instead observe during these events that the DC increases at short wavelengths while the DC decreases at long wavelengths, suggesting primarily an exchange between HbO2 and HbR volume fractions, as opposed to a dramatic HbT change.
Regions A–C (Figures 12.7–12.9) were chosen to highlight three different time signatures observed in the field of view during the CSD dynamics. The most contrasting feature between all three regions is the measured AC reflectance and the derived scattering coefficient. In Region B (Figure 12.8), each CSD event appears to cause a biphasic scattering change, with a sharp increase and then decrease, whereas a monophasic dip was observed in Region A (Figure 12.7). Region C (Figure 12.9) appears even more complex with a triphasic rise-dip-rise temporal profile. We observe that Regions A to C are located with increasing proximity to the CSD induction point (3 mm above the imaging field).
Because fractional changes in scattering and absorption have an equal (and opposite) effect on DC reflectance (see Section 12.2.3.3), any scattering (i.e., pathlength) changes measured here could be misinterpreted as absorption events with traditional ISOI analyses (i.e., DC reflectance only). In our observations, the measured scattering change of up to −0.05 mm−1 would be interpreted as an increase in absorption of up to +0.005 mm−1, more than the maximum measured absorption change for wavelengths 730, 780, or 830 nm in any of the three regions. In order to account for differential pathlength changes, Kohl et al. proposed a multispectral model [39], which they used to differentiate dynamic scattering and absorption changes using ISOI. This approach improves ISOI accuracy, and has been generally adopted as the method of choice for quantitative functional imaging. For dynamic measurements, we see MI as an improvement over this approach as it alternatively uses frequency domain measurements at a single wavelength to derive absolute scattering and absorption coefficients. This potentially provides a simplified single-wavelength measurement apparatus for detection of scattering, and also avoids potential mis-estimation of background optical properties.
Light scattering changes induced by spreading depression have been reported previously, and a comprehensive review is provided by Somjen. With in vivo spatially resolved reflectance measurements, Kohl et al. [11] separated absorption from scattering and observed a biphasic scattering response similar to that of Region A. With simultaneous laser scattering and electrophysiological measurements, both Jarvis et al. and Tao et al. found a strong correlation between electrical and optical scattering changes [12,13,40]. Tao et al. noted spatial heterogeneity in the dynamic spreading depression (SD) waveform related to the proximity to the SD induction site, similar to our results.
Using linear spectral analysis of absorption at all four wavelengths, we calculated the time-dependent chromophore concentration for Regions A, B, and C, presented in Figure 12.10A,B,C, respectively. In each region, the calculated baseline concentrations of H2O were assumed to be constant. All three regions exhibit remarkably similar trends in HbR, HbO2, HbT, and StO2. This similarity is not clear in the DC traces of Figures 12.7–12.9, further highlighting the benefit of accurate separation of μa and μs′. Focusing on the first CSD event, there is a very consistent signature of: (1) a 2-minute latency post-KCl administration, (2) a 30-second period of decreasing StO2 (3) a dramatic spike in both StO2 (3–10%) and HbT (2–4 μM) with rise and decay times of approximately 1 minute each. For each region, the final StO2 is approximately 5–10% lower than baseline, while the HbT restores to baseline values. This process repeats again twice more, except that the phase (2) desaturation appears to be absent. Additionally, in the “vessel” Region 3, we observe a gradual increase in HbT over the 30 minutes, indicating chronic blood pooling.

FIGURE 12.10
Recovered HbR, HbO2, HbT, and STO2, for ROIs A, B, and C (top, middle, and bottom), recovered by analysis of the multispectral absorption coefficients from Figures 12.7–12.9b (top).
We show in Figure 12.11 the spatio-temporal evolution of both chromophore concentration and scattering changes from the first SD wave in rat 3. These are depicted in the form of a time derivative, i.e., (C(tn + 1) − C(tn))/(tn + 1 − tn), where C represents concentration/saturation/scattering values and tn represents time of acquisition for data point n. This visualization is appealing as it highlights the changes with high contrast [18]. From left to right, we show HbO2, HbR, HbT, StO2, and μs′. Notice the wave in scattering which propagates from top right to bottom left, at a rate of approximately 3 mm/min. An increase, or “spike” in scattering is observed initially in the top right hand corner, in close proximity to the location of KCl administration. Note the large spikes in HbT and StO2 due to vascular activity from depression wave propagation through the measurement field. We observe a transient increase in saturation and blood volume. Over the longer time periods, however, we observe a slow, sustained trend toward hypoxia in the vein regions.

FIGURE 12.11
Spatio-temporal evolution of the hemodynamic and neural scattering response during a single spontaneous CSD event in rat 3. For visualization, a time derivative of the image sequence is displayed to highlight changes.
The spatio-temporal evolution of the scattering coefficient in Figure 12.11 reveals a spatially defined scattering wave (reduction in μs′) that precedes hemodynamic changes. The scattering drop is presumed to be a consequence of neuronal depolarization accompanying CSD. This observed wave pattern has been shown previously with reflectance ISOI and attributed to blood volume changes [18]. Interestingly, the scattering depolarization wave is clearly followed in space and time by the increase in deoxyhemoglobin (HbR), decrease in saturation (StO2), and drop in oxyhemoglobin (HbO2); changes that are consistent with depolarization-induced neural tissue oxygen consumption.
12.3.3. Dynamic MI Spectroscopy of Stroke
In order to assess the sensitivity of MI to stroke, we conducted preliminary studies in a rat middle cerebral artery occlusion (MCAo) model, the most commonly involved artery in ischemic strokes. The left MCA was surgically cauterized using monopolar cautery or ligated to produce a permanent stroke. Figure 12.12 shows pre-versus post-MCAo results for a representative animal. Data were acquired at 5 wavelengt
Original Source: https://www.ncbi.nlm.nih.gov/books/NBK20233/
Low-level laser therapy rescues dendrite atrophy via upregulating BDNF expression: implications for Alzheimer's disease.
Meng C1, He Z, Xing D. - J Neurosci. 2013 Aug 14;33(33):13505-17. doi: 10.1523/JNEUROSCI.0918-13.2013. () 744Background: Downregulation of brain-derived neurotrophic factor (BDNF) in the hippocampus occurs early in the progression of Alzheimer's disease (AD). Since BDNF plays a critical role in neuronal survival and dendrite growth, BDNF upregulation may contribute to rescue dendrite atrophy and cell loss in AD. Low-level laser therapy (LLLT) has been demonstrated to regulate neuronal function both in vitro and in vivo. In the present study, we found that LLLT rescued neurons loss and dendritic atrophy via upregulation of BDNF in both Aβ-treated hippocampal neurons and cultured APP/PS1 mouse hippocampal neurons. Photoactivation of transcription factor CRE-binding protein (CREB) increased both BDNF mRNA and protein expression, since knockdown CREB blocked the effects of LLLT. Furthermore, CREB-regulated transcription was in an ERK-dependent manner. Inhibition of ERK attenuated the DNA-binding efficiency of CREB to BDNF promoter. In addition, dendrite growth was improved after LLLT, characterized by upregulation of Rac1 activity and PSD-95 expression, and the increase in length, branching, and spine density of dendrites in hippocampal neurons. Together, these studies suggest that upregulation of BDNF with LLLT by activation of ERK/CREB pathway can ameliorate Aβ-induced neurons loss and dendritic atrophy, thus identifying a novel pathway by which LLLT protects against Aβ-induced neurotoxicity. Our research may provide a feasible therapeutic approach to control the progression of AD.
Abstract: Abstract Downregulation of brain-derived neurotrophic factor (BDNF) in the hippocampus occurs early in the progression of Alzheimer's disease (AD). Since BDNF plays a critical role in neuronal survival and dendrite growth, BDNF upregulation may contribute to rescue dendrite atrophy and cell loss in AD. Low-level laser therapy (LLLT) has been demonstrated to regulate neuronal function both in vitro and in vivo. In the present study, we found that LLLT rescued neurons loss and dendritic atrophy via upregulation of BDNF in both Aβ-treated hippocampal neurons and cultured APP/PS1 mouse hippocampal neurons. Photoactivation of transcription factor CRE-binding protein (CREB) increased both BDNF mRNA and protein expression, since knockdown CREB blocked the effects of LLLT. Furthermore, CREB-regulated transcription was in an ERK-dependent manner. Inhibition of ERK attenuated the DNA-binding efficiency of CREB to BDNF promoter. In addition, dendrite growth was improved after LLLT, characterized by upregulation of Rac1 activity and PSD-95 expression, and the increase in length, branching, and spine density of dendrites in hippocampal neurons. Together, these studies suggest that upregulation of BDNF with LLLT by activation of ERK/CREB pathway can ameliorate Aβ-induced neurons loss and dendritic atrophy, thus identifying a novel pathway by which LLLT protects against Aβ-induced neurotoxicity. Our research may provide a feasible therapeutic approach to control the progression of AD.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/23946409
Photobiomodulation by low-power laser irradiation attenuates Aβ-induced cell apoptosis through the Akt/GSK3β/β-catenin pathway.
Liang J1, Liu L, Xing D. - Free Radic Biol Med. 2012 Oct 1;53(7):1459-67. doi: 10.1016/j.freeradbiomed.2012.08.003. Epub 2012 Aug 11. () 1145Background: Apoptosis induced by amyloid β peptide (Aβ) is thought to associate with the pathogenesis of Alzheimer disease (AD). Accumulating evidence shows that low-power laser irradiation (LPLI) is capable of reducing Aβ-induced apoptosis. However, the underlying mechanisms remain unclear. In this study, we report a novel molecular mechanism by which LPLI attenuates Aβ(25-35)-induced apoptosis through the Akt/GSK3β/β-catenin pathway. We found that Akt activated by LPLI interacted with GSK3β and phosphorylated it on Ser9 in the presence of Aβ(25-35), which resulted in the inhibition of GSK3β. Furthermore, LPLI increased the nuclear translocation of β-catenin and enhanced its T cell factor/lymphocyte enhancer factor-dependent transcriptional activity via the Akt/GSK3β pathway to promote cell survival upon treatment with Aβ(25-35.) Our data demonstrate that LPLI has a prosurvival effect on Aβ-induced apoptosis and may be an effective therapeutic strategy in treating AD by targeting GSK3β.
Abstract: Abstract Apoptosis induced by amyloid β peptide (Aβ) is thought to associate with the pathogenesis of Alzheimer disease (AD). Accumulating evidence shows that low-power laser irradiation (LPLI) is capable of reducing Aβ-induced apoptosis. However, the underlying mechanisms remain unclear. In this study, we report a novel molecular mechanism by which LPLI attenuates Aβ(25-35)-induced apoptosis through the Akt/GSK3β/β-catenin pathway. We found that Akt activated by LPLI interacted with GSK3β and phosphorylated it on Ser9 in the presence of Aβ(25-35), which resulted in the inhibition of GSK3β. Furthermore, LPLI increased the nuclear translocation of β-catenin and enhanced its T cell factor/lymphocyte enhancer factor-dependent transcriptional activity via the Akt/GSK3β pathway to promote cell survival upon treatment with Aβ(25-35.) Our data demonstrate that LPLI has a prosurvival effect on Aβ-induced apoptosis and may be an effective therapeutic strategy in treating AD by targeting GSK3β. Copyright © 2012 Elsevier Inc. All rights reserved.
Methods: Copyright © 2012 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/22917976
Low-level light therapy improves cortical metabolic capacity and memory retention.
Rojas JC1, Bruchey AK, Gonzalez-Lima F. - J Alzheimers Dis. 2012;32(3):741-52. doi: 10.3233/JAD-2012-120817. () 1182Background: Cerebral hypometabolism characterizes mild cognitive impairment and Alzheimer's disease. Low-level light therapy (LLLT) enhances the metabolic capacity of neurons in culture through photostimulation of cytochrome oxidase, the mitochondrial enzyme that catalyzes oxygen consumption in cellular respiration. Growing evidence supports that neuronal metabolic enhancement by LLLT positively impacts neuronal function in vitro and in vivo. Based on its effects on energy metabolism, it is proposed that LLLT will also affect the cerebral cortex in vivo and modulate higher-order cognitive functions such as memory. In vivo effects of LLLT on brain and behavior are poorly characterized. We tested the hypothesis that in vivo LLLT facilitates cortical oxygenation and metabolic energy capacity and thereby improves memory retention. Specifically, we tested this hypothesis in rats using fear extinction memory, a form of memory modulated by prefrontal cortex activation. Effects of LLLT on brain metabolism were determined through measurement of prefrontal cortex oxygen concentration with fluorescent quenching oximetry and by quantitative cytochrome oxidase histochemistry. Experiment 1 verified that LLLT increased the rate of oxygen consumption in the prefrontal cortex in vivo. Experiment 2 showed that LLLT-treated rats had an enhanced extinction memory as compared to controls. Experiment 3 showed that LLLT reduced fear renewal and prevented the reemergence of extinguished conditioned fear responses. Experiment 4 showed that LLLT induced hormetic dose-response effects on the metabolic capacity of the prefrontal cortex. These data suggest that LLLT can enhance cortical metabolic capacity and retention of extinction memories, and implicate LLLT as a novel intervention to improve memory.
Abstract: Abstract Cerebral hypometabolism characterizes mild cognitive impairment and Alzheimer's disease. Low-level light therapy (LLLT) enhances the metabolic capacity of neurons in culture through photostimulation of cytochrome oxidase, the mitochondrial enzyme that catalyzes oxygen consumption in cellular respiration. Growing evidence supports that neuronal metabolic enhancement by LLLT positively impacts neuronal function in vitro and in vivo. Based on its effects on energy metabolism, it is proposed that LLLT will also affect the cerebral cortex in vivo and modulate higher-order cognitive functions such as memory. In vivo effects of LLLT on brain and behavior are poorly characterized. We tested the hypothesis that in vivo LLLT facilitates cortical oxygenation and metabolic energy capacity and thereby improves memory retention. Specifically, we tested this hypothesis in rats using fear extinction memory, a form of memory modulated by prefrontal cortex activation. Effects of LLLT on brain metabolism were determined through measurement of prefrontal cortex oxygen concentration with fluorescent quenching oximetry and by quantitative cytochrome oxidase histochemistry. Experiment 1 verified that LLLT increased the rate of oxygen consumption in the prefrontal cortex in vivo. Experiment 2 showed that LLLT-treated rats had an enhanced extinction memory as compared to controls. Experiment 3 showed that LLLT reduced fear renewal and prevented the reemergence of extinguished conditioned fear responses. Experiment 4 showed that LLLT induced hormetic dose-response effects on the metabolic capacity of the prefrontal cortex. These data suggest that LLLT can enhance cortical metabolic capacity and retention of extinction memories, and implicate LLLT as a novel intervention to improve memory.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/22850314
670 nm laser light and EGCG complementarily reduce amyloid-β aggregates in human neuroblastoma cells: basis for treatment of Alzheimer's disease?
Sommer AP1, Bieschke J, Friedrich RP, Zhu D, Wanker EE, Fecht HJ, Mereles D, Hunstein W. - Photomed Laser Surg. 2012 Jan;30(1):54-60. doi: 10.1089/pho.2011.3073. Epub 2011 Oct 26. () 1485Background: The aim of the present study is to present the results of in vitro experiments with possible relevance in the treatment of Alzheimer's disease (AD).
Abstract: Abstract OBJECTIVE: The aim of the present study is to present the results of in vitro experiments with possible relevance in the treatment of Alzheimer's disease (AD). BACKGROUND DATA: Despite intensive research efforts, there is no treatment for AD. One root cause of AD is the extra- and intracellular deposition of amyloid-beta (Aβ) fibrils in the brain. Recently, it was shown that extracellular Aβ can enter brain cells, resulting in neurotoxicity. METHODS: After internalization of Aβ(42) into human neuroblastoma (SH-EP) cells, they were irradiated with moderately intense 670-nm laser light (1000 Wm(-2)) and/or treated with epigallocatechin gallate (EGCG). RESULTS: In irradiated cells, Aβ(42) aggregate amounts were significantly lower than in nonirradiated cells. Likewise, in EGCG-treated cells, Aβ(42) aggregate amounts were significantly lower than in non-EGCG-treated cells. Except for the cells simultaneously laden with Aβ(42) and EGCG, there was a significant increase in cell numbers in response to laser irradiation. EGCG alone had no effect on cell proliferation. Laser irradiation significantly increased ATP levels in Aβ(42)-free cells, when compared to nonirradiated cells. Laser-induced clearance of Aβ(42) aggregates occurred at the expense of cellular ATP. CONCLUSIONS: Irradiation with moderate levels of 670-nm light and EGCG supplementation complementarily reduces Aβ aggregates in SH-EP cells. Transcranial penetration of moderate levels of red to near-infrared (NIR) light has already been amply exploited in the treatment of patients with acute stroke; the blood-brain barrier (BBB) penetration of EGCG has been demonstrated in animals. We hope that our approach will inspire a practical therapy for AD.
Methods: Despite intensive research efforts, there is no treatment for AD. One root cause of AD is the extra- and intracellular deposition of amyloid-beta (Aβ) fibrils in the brain. Recently, it was shown that extracellular Aβ can enter brain cells, resulting in neurotoxicity.
Results: After internalization of Aβ(42) into human neuroblastoma (SH-EP) cells, they were irradiated with moderately intense 670-nm laser light (1000 Wm(-2)) and/or treated with epigallocatechin gallate (EGCG).
Conclusions: In irradiated cells, Aβ(42) aggregate amounts were significantly lower than in nonirradiated cells. Likewise, in EGCG-treated cells, Aβ(42) aggregate amounts were significantly lower than in non-EGCG-treated cells. Except for the cells simultaneously laden with Aβ(42) and EGCG, there was a significant increase in cell numbers in response to laser irradiation. EGCG alone had no effect on cell proliferation. Laser irradiation significantly increased ATP levels in Aβ(42)-free cells, when compared to nonirradiated cells. Laser-induced clearance of Aβ(42) aggregates occurred at the expense of cellular ATP.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/22029866
Inhibition of Aβ(25-35)-induced cell apoptosis by low-power-laser-irradiation (LPLI) through promoting Akt-dependent YAP cytoplasmic translocation.
Zhang H1, Wu S, Xing D. - Cell Signal. 2012 Jan;24(1):224-32. doi: 10.1016/j.cellsig.2011.09.004. Epub 2011 Sep 14. () 1520Background: Deposition of amyloid-β-peptide (Aβ) in the brain is considered a pathological hallmark of Alzheimer's disease (AD). Our previous studies show that Yes-associated protein (YAP) is involved in the regulation of apoptosis induced by Aβ(25-35) through YAP nuclear translocation and its pro-apoptotic function is mediated by its interaction with p73. In the present study, we first found that Low-power laser irradiation (LPLI) promoted YAP cytoplasmic translocation and inhibited Aβ(25-35)-induced YAP nuclear translocation. Moreover, the cytoplasmic translocation was in an Akt-dependent manner. Activated Akt by LPLI phosphorylated YAP on ser127 (S127) and resulted in decreasing the interaction between YAP and p73, and in suppressing the proapoptotic gene bax expression following Aβ(25-35) treatment. Inhibition of Akt expression by siRNA significantly abolished the effect of LPLI. More importantly, LPLI could inhibit Aβ(25-35)-induced cell apoptosis through activation of Akt/YAP/p73 signaling pathway. Therefore, our findings first suggest that YAP may be a therapeutic target and these results directly point to a potential therapeutic strategy for the treatment of AD through Akt/YAP/p73 signaling pathway with LPLI.
Abstract: Abstract Deposition of amyloid-β-peptide (Aβ) in the brain is considered a pathological hallmark of Alzheimer's disease (AD). Our previous studies show that Yes-associated protein (YAP) is involved in the regulation of apoptosis induced by Aβ(25-35) through YAP nuclear translocation and its pro-apoptotic function is mediated by its interaction with p73. In the present study, we first found that Low-power laser irradiation (LPLI) promoted YAP cytoplasmic translocation and inhibited Aβ(25-35)-induced YAP nuclear translocation. Moreover, the cytoplasmic translocation was in an Akt-dependent manner. Activated Akt by LPLI phosphorylated YAP on ser127 (S127) and resulted in decreasing the interaction between YAP and p73, and in suppressing the proapoptotic gene bax expression following Aβ(25-35) treatment. Inhibition of Akt expression by siRNA significantly abolished the effect of LPLI. More importantly, LPLI could inhibit Aβ(25-35)-induced cell apoptosis through activation of Akt/YAP/p73 signaling pathway. Therefore, our findings first suggest that YAP may be a therapeutic target and these results directly point to a potential therapeutic strategy for the treatment of AD through Akt/YAP/p73 signaling pathway with LPLI. Copyright © 2011 Elsevier Inc. All rights reserved.
Methods: Copyright © 2011 Elsevier Inc. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/21945154
Long-term safety of single and multiple infrared transcranial laser treatments in Sprague-Dawley rats.
McCarthy TJ1, De Taboada L, Hildebrandt PK, Ziemer EL, Richieri SP, Streeter J. - Photomed Laser Surg. 2010 Oct;28(5):663-7. doi: 10.1089/pho.2009.2581. () 1903Background: Growing interest exists in the use of near-infrared laser therapies for the treatment of numerous neurologic conditions, including acute ischemic stroke, traumatic brain injury, Parkinson's disease, and Alzheimer's disease. In consideration of these trends, the objective of this study was to evaluate the long-term safety of transcranial laser therapy with continuous-wave (CW) near-infrared laser light (wavelength, 808 ± 10 nm, 2-mm diameter) with a nominal radiant power of 70 mW; power density, 2,230 mW/cm(2), and energy density, 268 J/cm(2) at the scalp (10 mW/cm(2) and 1.2 J/cm(2) at the cerebral cortical surface) in healthy Sprague-Dawley rats.
Abstract: Abstract BACKGROUND AND OBJECTIVE: Growing interest exists in the use of near-infrared laser therapies for the treatment of numerous neurologic conditions, including acute ischemic stroke, traumatic brain injury, Parkinson's disease, and Alzheimer's disease. In consideration of these trends, the objective of this study was to evaluate the long-term safety of transcranial laser therapy with continuous-wave (CW) near-infrared laser light (wavelength, 808 ± 10 nm, 2-mm diameter) with a nominal radiant power of 70 mW; power density, 2,230 mW/cm(2), and energy density, 268 J/cm(2) at the scalp (10 mW/cm(2) and 1.2 J/cm(2) at the cerebral cortical surface) in healthy Sprague-Dawley rats. MATERIALS AND METHODS: In this study, 120 anesthetized rats received sequential transcranial laser treatments to the right and left parietal areas of the head on the same day (minimum of 5 min between irradiation of each side), on either Day 1 or on each of Days 1, 3, and 5. Sixty anesthetized rats served as sham controls. Rats were evaluated 1 year after treatment for abnormalities in clinical hematology and brain and pituitary gland histopathology. RESULTS: No toxicologically important differences were found in the clinical hematology results between sham-control and laser-treated rats for any hematologic parameters examined. All values fell within historic control reference ranges for aged Sprague-Dawley rats. Similarly, brain and pituitary gland histopathology showed no treatment-related abnormalities or induced neoplasia. CONCLUSIONS: Single and multiple applications of transcranial laser therapy with 808-nm CW laser light at a nominal power density of 10 mW/cm(2) at the surface of the cerebral cortex appears to be safe in Sprague-Dawley rats 1 year after treatment.
Methods: In this study, 120 anesthetized rats received sequential transcranial laser treatments to the right and left parietal areas of the head on the same day (minimum of 5 min between irradiation of each side), on either Day 1 or on each of Days 1, 3, and 5. Sixty anesthetized rats served as sham controls. Rats were evaluated 1 year after treatment for abnormalities in clinical hematology and brain and pituitary gland histopathology.
Results: No toxicologically important differences were found in the clinical hematology results between sham-control and laser-treated rats for any hematologic parameters examined. All values fell within historic control reference ranges for aged Sprague-Dawley rats. Similarly, brain and pituitary gland histopathology showed no treatment-related abnormalities or induced neoplasia.
Conclusions: Single and multiple applications of transcranial laser therapy with 808-nm CW laser light at a nominal power density of 10 mW/cm(2) at the surface of the cerebral cortex appears to be safe in Sprague-Dawley rats 1 year after treatment.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/20961232
Low energy laser light (632.8 nm) suppresses amyloid-β peptide-induced oxidative and inflammatory responses in astrocytes.
Yang X1, Askarova S, Sheng W, Chen JK, Sun AY, Sun GY, Yao G, Lee JC. - Neuroscience. 2010 Dec 15;171(3):859-68. doi: 10.1016/j.neuroscience.2010.09.025. Epub 2010 Sep 25. () 1933Background: Oxidative stress and inflammation are important processes in the progression of Alzheimer's disease (AD). Recent studies have implicated the role of amyloid β-peptides (Aβ) in mediating these processes. In astrocytes, oligomeric Aβ induces the assembly of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complexes resulting in its activation to produce anionic superoxide. Aβ also promotes production of pro-inflammatory factors in astrocytes. Since low energy laser has previously been reported to attenuate oxidative stress and inflammation in biological systems, the objective of this study was to examine whether this type of laser light was able to abrogate the oxidative and inflammatory responses induced by Aβ. Primary rat astrocytes were exposed to Helium-Neon laser (λ=632.8 nm), followed by the treatment with oligomeric Aβ. Primary rat astrocytes were used to measure Aβ-induced production of superoxide anions using fluorescence microscopy of dihydroethidium (DHE), assembly of NADPH oxidase subunits by the colocalization between the cytosolic p47(phox) subunit and the membrane gp91(phox) subunit using fluorescent confocal microscopy, phosphorylation of cytosolic phospholipase A(2) cPLA(2) and expressions of pro-inflammatory factors including interleukin-1β (IL-1β) and inducible nitric-oxide synthase (iNOS) using Western blot Analysis. Our data showed that laser light at 632.8 nm suppressed Aβ-induced superoxide production, colocalization between NADPH oxidase gp91(phox) and p47(phox) subunits, phosphorylation of cPLA(2,) and the expressions of IL-1β and iNOS in primary astrocytes. We demonstrated for the first time that 632.8 nm laser was capable of suppressing cellular pathways of oxidative stress and inflammatory responses critical in the pathogenesis in AD. This study should prove to provide the groundwork for further investigations for the potential use of laser therapy as a treatment for AD.
Abstract: Abstract Oxidative stress and inflammation are important processes in the progression of Alzheimer's disease (AD). Recent studies have implicated the role of amyloid β-peptides (Aβ) in mediating these processes. In astrocytes, oligomeric Aβ induces the assembly of nicotinamide adenine dinucleotide phosphate (NADPH) oxidase complexes resulting in its activation to produce anionic superoxide. Aβ also promotes production of pro-inflammatory factors in astrocytes. Since low energy laser has previously been reported to attenuate oxidative stress and inflammation in biological systems, the objective of this study was to examine whether this type of laser light was able to abrogate the oxidative and inflammatory responses induced by Aβ. Primary rat astrocytes were exposed to Helium-Neon laser (λ=632.8 nm), followed by the treatment with oligomeric Aβ. Primary rat astrocytes were used to measure Aβ-induced production of superoxide anions using fluorescence microscopy of dihydroethidium (DHE), assembly of NADPH oxidase subunits by the colocalization between the cytosolic p47(phox) subunit and the membrane gp91(phox) subunit using fluorescent confocal microscopy, phosphorylation of cytosolic phospholipase A(2) cPLA(2) and expressions of pro-inflammatory factors including interleukin-1β (IL-1β) and inducible nitric-oxide synthase (iNOS) using Western blot Analysis. Our data showed that laser light at 632.8 nm suppressed Aβ-induced superoxide production, colocalization between NADPH oxidase gp91(phox) and p47(phox) subunits, phosphorylation of cPLA(2,) and the expressions of IL-1β and iNOS in primary astrocytes. We demonstrated for the first time that 632.8 nm laser was capable of suppressing cellular pathways of oxidative stress and inflammatory responses critical in the pathogenesis in AD. This study should prove to provide the groundwork for further investigations for the potential use of laser therapy as a treatment for AD. Copyright © 2010 IBRO. Published by Elsevier Ltd. All rights reserved.
Methods: Copyright © 2010 IBRO. Published by Elsevier Ltd. All rights reserved.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/20884337
Low-power laser irradiation inhibiting Abeta25-35-induced PC12 cell apoptosis via PKC activation.
Zhang L1, Xing D, Zhu D, Chen Q. - Cell Physiol Biochem. 2008;22(1-4):215-22. doi: 10.1159/000149799. Epub 2008 Jul 25. () 2733Background: Apoptosis is a contributing pathophysiological mechanism of Alzheimer's disease (AD). Recently, low-power laser irradiation (LPLI) has been applied to moderate AD, but the underlying mechanism remains unknown. In this study, the techniques of fluorescence resonance energy transfer (FRET) and real-time quantitative RT-PCR were used to investigate the anti-apoptotic mechanism of LPLI. Rat pheochromocytoma (PC12) cells were treated with amyloid beta 25-35 (Abeta(25-35)) for induction of apoptosis before LPLI treatment. The cell viability assays and morphological examinations show that low fluence of LPLI (0.156 J/cm(2)-0.624 J/cm(2)) could inhibit the cells apoptosis. An increase of PKC activation was dynamically monitored in the cells treated with PMA (specific activator of PKC), LPLI only or Abeta(25-35) followed by 5 min LPLI treatment, respectively. However, the effect of LPLI activating PKC could be inhibited by Go 6983 (specific inhibitor of PKC). Similar results were obtained by using Western blot analysis. Furthermore, LPLI involved an increase in mRNA of the cell survival member bcl-xl and a decrease in the up-regulation of cell death member bax mRNA caused by Abeta(25-35). Further data show that low fluence of LPLI could reverse the increased level of bax/bcl-xl mRNA ratio caused by Abeta(25-35) treatment. In addition, Go 6983 could inhibit the decreased level of bax/bcl-xl mRNA ratio. Taken together, these data clearly indicate that LPLI inhibited Abeta(25-35)-induced PC12 cell apoptosis via PKC-mediated regulation of bax/bcl-xl mRNA ratio.
Abstract: Abstract Apoptosis is a contributing pathophysiological mechanism of Alzheimer's disease (AD). Recently, low-power laser irradiation (LPLI) has been applied to moderate AD, but the underlying mechanism remains unknown. In this study, the techniques of fluorescence resonance energy transfer (FRET) and real-time quantitative RT-PCR were used to investigate the anti-apoptotic mechanism of LPLI. Rat pheochromocytoma (PC12) cells were treated with amyloid beta 25-35 (Abeta(25-35)) for induction of apoptosis before LPLI treatment. The cell viability assays and morphological examinations show that low fluence of LPLI (0.156 J/cm(2)-0.624 J/cm(2)) could inhibit the cells apoptosis. An increase of PKC activation was dynamically monitored in the cells treated with PMA (specific activator of PKC), LPLI only or Abeta(25-35) followed by 5 min LPLI treatment, respectively. However, the effect of LPLI activating PKC could be inhibited by Go 6983 (specific inhibitor of PKC). Similar results were obtained by using Western blot analysis. Furthermore, LPLI involved an increase in mRNA of the cell survival member bcl-xl and a decrease in the up-regulation of cell death member bax mRNA caused by Abeta(25-35). Further data show that low fluence of LPLI could reverse the increased level of bax/bcl-xl mRNA ratio caused by Abeta(25-35) treatment. In addition, Go 6983 could inhibit the decreased level of bax/bcl-xl mRNA ratio. Taken together, these data clearly indicate that LPLI inhibited Abeta(25-35)-induced PC12 cell apoptosis via PKC-mediated regulation of bax/bcl-xl mRNA ratio. Copyright 2008 S. Karger AG, Basel.
Methods: Copyright 2008 S. Karger AG, Basel.
Original Source: http://www.ncbi.nlm.nih.gov/pubmed/18769048
Preliminary results of highly localized plantar irradiation with low incident levels of mid-infrared energy which contributes to the prevention of dementia associated with underlying diabetes mellitus.
Ryotokuji K, Ishimaru K, Kihara K, Nakajima T, Otani S, Namiki Y - (Publication) 4421Stress-free Therapy Research Center, Ryotokuji University.
BACKGROUND AND AIMS: The incidence of vascular dementia (VD) and Alzheimer's disease (AD) has recently increased and the prevention of progression of these diseases is very difficult.
RESULTS: The application of pinpoint plantar long-wavelength infrared light irradiation (PP-LILI) to a patient's sole, at the point where the line drawn between the first and second metatarsal heads intersects with the vertical line from the medial malleolus, was effective in increasing blood flow to the facial artery, elevating high- density lipoprotein cholesterol (HDL-C) levels, and reducing insulin resistance.
CONCLUSIONS: We found that these effects of PP-LILI might be helpful for preventing VD and AD, conditions that are becoming a social problem in an aging Japanese society.
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=25941422
Low-Level Laser Therapy Ameliorates Disease Progression in a Mouse Model of Alzheimer's Disease.
Farfara D, Tuby H, Trudler D, Doron-Mandel E, Maltz L, Vassar RJ, Frenkel D, Oron U - (Publication) 4423Department of Neurobiology, George S. Wise Faculty of Life Sciences, Tel Aviv University, Tel Aviv, Israel.
Low-level laser therapy (LLLT) has been used to treat inflammation, tissue healing, and repair processes. We recently reported that LLLT to the bone marrow (BM) led to proliferation of mesenchymal stem cells (MSCs) and their homing in the ischemic heart suggesting its role in regenerative medicine. The aim of the present study was to investigate the ability of LLLT to stimulate MSCs of autologous BM in order to affect neurological behavior and beta-amyloid burden in progressive stages of Alzheimer's disease (AD) mouse model. MSCs from wild-type mice stimulated with LLLT showed to increase their ability to maturate towards a monocyte lineage and to increase phagocytosis activity towards soluble amyloid beta (Abeta). Furthermore, weekly LLLT to BM of AD mice for 2 months, starting at 4 months of age (progressive stage of AD), improved cognitive capacity and spatial learning, as compared to sham-treated AD mice. Histology revealed a significant reduction in Abeta brain burden. Our results suggest the use of LLLT as a therapeutic application in progressive stages of AD and imply its role in mediating MSC therapy in brain amyloidogenic diseases.
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=24994540
Photobiomodulation with near infrared light mitigates Alzheimer's disease- related pathology in cerebral cortex - evidence from two transgenic mouse models.
Purushothuman S, Johnstone DM, Nandasena C, Mitrofanis J, Stone J - (Publication) 4431Bosch Institute, University of Sydney NSW 2006, Australia. Daniel.Johnstone@sydney.edu.au
INTRODUCTION: Previous work has demonstrated the efficacy of irradiating tissue with red to infrared light in mitigating cerebral pathology and degeneration in animal models of stroke, traumatic brain injury, parkinsonism and Alzheimer's disease (AD). Using mouse models, we explored the neuroprotective effect of near infrared light (NIr) treatment, delivered at an age when substantial pathology is already present in the cerebral cortex.
METHODS: We studied two mouse models with AD- related pathologies: the K369I tau transgenic model (K3), engineered to develop neurofibrillary tangles, and the APPswe/PSEN1dE9 transgenic model (APP/PS1), engineered to develop amyloid plaques. Mice were treated with NIr 20 times over a four-week period and histochemistry was used to quantify AD- related pathological hallmarks and other markers of cell damage in the neocortex and hippocampus.
RESULTS: In the K3 mice, NIr treatment was associated with a reduction in hyperphosphorylated tau, neurofibrillary tangles and oxidative stress markers (4-hydroxynonenal and 8-hydroxy-2'- deoxyguanosine) to near wildtype levels in the neocortex and hippocampus, and with a restoration of expression of the mitochondrial marker cytochrome c oxidase in surviving neurons. In the APP/PS1 mice, NIr treatment was associated with a reduction in the size and number of amyloid-beta plaques in the neocortex and hippocampus.
CONCLUSIONS: Our results, in two transgenic mouse models, suggest that NIr may have potential as an effective, minimally-invasive intervention for mitigating, and even reversing, progressive cerebral degenerations.
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=24387311
Laser acupuncture improves memory impairment in an animal model of Alzheimer's disease.
Sutalangka C, Wattanathorn J, Muchimapura S, Thukham-Mee W, Wannanon P, Tong-Un T - (Publication) 4433Department of Physiology (Neuroscience Program), Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand; Integrative Complementary Alternative Medicine Research and Development Group, Khon Kaen University, Khon Kaen, Thailand.
The burden of Alzheimer's disease is continually rising globally, especially in the Asia-Pacific region. Unfortunately, the efficacy of the therapeutic strategy is still very limited. Because the effect of acupuncture at HT7 can improve learning and memory, the beneficial effect of laser acupuncture, a noninvasive form of acupuncture, at HT7 on memory improvement in patients with Alzheimer's disease has been a focus of research. To elucidate this issue, we used AF64A, a cholinotoxin, to induce memory impairment in male Wistar rats, which weighed 180-220 g. Then, the animals were treated with laser acupuncture either at HT7 or at a sham acupoint once daily for 10 minutes for a period of 14 days.
Spatial memory assessments were performed at 1, 7, and 14 days after AF64A administration and at the end of the experiment, and the changes in the malondialdehyde (MDA) level and in the superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GSH-Px), and acetylcholinesterase (AChE) activities in the hippocampus were recorded. The results showed that laser acupuncture significantly suppressed AChE activity in the hippocampus. Although laser acupuncture enhanced SOD and CAT activities, no reduction in MDA level in this area was observed. Therefore, laser acupuncture at HT7 is a potential strategy to attenuate memory impairment in patients with Alzheimer's disease. However, further research, especially on the toxicity of laser acupuncture following repetitive exposure, is essential.
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=24139462
Low-Level Laser Therapy Rescues Dendrite Atrophy via Upregulating BDNF Expression: Implications for Alzheimer's Disease.
Meng C, He Z, Xing D - (Publication) 4434MOE Key Laboratory of Laser Life Science and Institute of Laser Life Science, College of Biophotonics, South China Normal University, Guangzhou 510631, China.
Downregulation of brain-derived neurotrophic factor (BDNF) in the hippocampus occurs early in the progression of Alzheimer's disease (AD). Since BDNF plays a critical role in neuronal survival and dendrite growth, BDNF upregulation may contribute to rescue dendrite atrophy and cell loss in AD. Low- level laser therapy (LLLT) has been demonstrated to regulate neuronal function both in vitro and in vivo. In the present study, we found that LLLT rescued neurons loss and dendritic atrophy via upregulation of BDNF in both Abeta-treated hippocampal neurons and cultured APP/PS1 mouse hippocampal neurons. Photoactivation of transcription factor CRE-binding protein (CREB) increased both BDNF mRNA and protein expression, since knockdown CREB blocked the effects of LLLT. Furthermore, CREB-regulated transcription was in an ERK-dependent manner. Inhibition of ERK attenuated the DNA-binding efficiency of CREB to BDNF promoter. In addition, dendrite growth was improved after LLLT, characterized by upregulation of Rac1 activity and PSD-95 expression, and the increase in length, branching, and spine density of dendrites in hippocampal neurons. Together, these studies suggest that upregulation of BDNF with LLLT by activation of ERK/CREB pathway can ameliorate Abeta-induced neurons loss and dendritic atrophy, thus identifying a novel pathway by which LLLT protects against Abeta-induced neurotoxicity.
Our research may provide a feasible therapeutic approach to control the progression of AD. J Neurosci 2013 Aug 14 33(33) 13505-17
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=23946409
670 nm Laser Light and EGCG Complementarily Reduce Amyloid-beta Aggregates in Human Neuroblastoma Cells: Basis for Treatment of Alzheimer's Disease?
Sommer AP, Bieschke J, Friedrich RP, Zhu D, Wanker EE, Fecht HJ, Mereles D, Hunstein W - (Publication) 44501 Institute of Micro and Nanomaterials, Nanobionic Laboratory, University of Ulm , Ulm, Germany .
Abstract Objective: The aim of the present study is to present the results of in vitro experiments with possible relevance in the treatment of Alzheimer's disease (AD).
Background Data: Despite intensive research efforts, there is no treatment for AD. One root cause of AD is the extra- and intracellular deposition of amyloid-beta (Abeta) fibrils in the brain. Recently, it was shown that extracellular Abeta can enter brain cells, resulting in neurotoxicity.
Methods: After internalization of Abeta(42) into human neuroblastoma (SH-EP) cells, they were irradiated with moderately intense 670-nm laser light (1000 Wm (-2)) and/or treated with epigallocatechin gallate (EGCG).
Results: In irradiated cells, Abeta(42) aggregate amounts were significantly lower than in nonirradiated cells. Likewise, in EGCG-treated cells, Abeta(42) aggregate amounts were significantly lower than in non-EGCG-treated cells. Except for the cells simultaneously laden with Abeta(42) and EGCG, there was a significant increase in cell numbers in response to laser irradiation. EGCG alone had no effect on cell proliferation. Laser irradiation significantly increased ATP levels in Abeta(42)-free cells, when compared to nonirradiated cells. Laser- induced clearance of Abeta(42) aggregates occurred at the expense of cellular ATP.
Conclusions: Irradiation with moderate levels of 670-nm light and EGCG supplementation complementarily reduces Abeta aggregates in SH-EP cells. Transcranial penetration of moderate levels of red to near-infrared (NIR) light has already been amply exploited in the treatment of patients with acute stroke; the blood-brain barrier (BBB) penetration of EGCG has been demonstrated in animals. We hope that our approach will inspire a practical therapy for AD.
Original Source: https://www.ncbi.nlm.nih.gov/pubmed/?term=22029866
Light Therapy to Stem Cells: A New Therapeutic Approach in Regenerative Medicine – A Mini Review
Amir Oron1 and Uri Oron2* - 15 April 2015 (Publication) 4489 Abstract
Regenerative capacity following injury or an ischemic event is confined to non mammalian vertebrates. Mammals have a limited capacity to restore organs
following injury to organs like the liver and skeletal muscles but practically no ability to regenerate organs like the heart or brain following an ischemic event
or injury. We tried a new approach in cell based therapy to improve regeneration in various organs following ischemic injury. Low-level laser therapy (LLLT)
which has photobiostimulating effects on cells was delivered to autologous bone marrow (BM) that is enriched with stem cells and various progenitor cells, in
order to induce the cells in the BM for the benefit of the injured /ischemic organs. In a model of induced myocardial infarction (MI) in rats laser application
to the BM caused a marked and significant decrease (79%) in infarct size (scarring) 3 weeks post-MI. It was also found that a significantly higher density of
c-kit positive cells (a marker of mesenchymal stem cells) in the myocardium of laser-treated rats relative to non-treated rat’s post-MI. The novel approach
presented in this study, of the use of stem cells for cell therapy to the infracted heart, avoids the need to isolate millions of stem cells, to grow them in vitro and
to inject them back into the patient. In the same line of rationale we tried to find whether LLLT to the BM could be beneficial also to kidney impairment after
ischemic reperfusion injury (IRI) to the rat kidney. C-kit positive cell density in kidneys post-IRI and laser-treatment was significantly (p=0.05) 2.4-fold higher
compared to the non laser treated group. Creatinine, blood urea nitrogen, and cystatin-C levels were significantly lower in the laser-treated rats as compared
to non-treated ones. The effect of LLLT delivery to BM was also tested on Alzheimer’s disease (AD) mice in their late stage of the disease. Mice were given
multiple (every 10 days) LLLT to BM from age 4 to 6 months. It was found that in the treated AD-mice neurological tests (Fear and Cognitive tests) revealed
a significantly (p<0.05) better neurological performance and cognitive capacity compared to the non-treated AD mice. Furthermore, concomitantly with the
improved neurological performance, ß-amyloid density in the hippocampal region of the brains was revealed to be significantly less in the laser-treated mice
as compared to control. In conclusion, a novel approach, of applying LLLT to autologeous BM in order to induce stem cells that are consequently recruited to the
injured/ischemic organ leading to a marked beneficial effect post-ischemic event or degenerative process is presented. This approach is novel in the respect
that it is stimulating the patient’s own abilities to initiate a regenerative response in an organ by the utilization of light. The possibility that this approach can
also be applied to other ischemic/injured organs or organs undergoing degenerative processes (i.e. neurodegenerative diseases), with consequent beneficial
effects, cannot be ruled out
CELL THERAPY FOR INJURED/ISCHEMIC ORGANS
Clinical trials have lately been implemented in a growing
abundance due to the extensive research and new approaches of
cell based therapies for the reconstruction of impaired organs.
Regenerative capacity following injury or an ischemic event
is confined to non mammalian vertebrates. In particular, fish
and primitive amphibians can regenerate organs like the heart,
brain and limbs. However, mammals have a limited capacity
to restore organs following injury to organs like the liver and
skeletal muscles but practically no ability to regenerate organs
like the heart or brain following an ischemic event or injury.
The mammalian heart, including the human heart, for example,
has a very limited capacity to regenerate following damage or
an acute ischemic event like myocardial infarction (MI). This is
due to the very low level of cardiomyocyte proliferation and the
limited number of cells expressing stem-cell marker proteins.
Stem-cell-based therapy was suggested as a potential solution
to the above situation. In recent years, cell-based therapy for
cardiac repair in particular has undergone a rapid transition
from basic science research to clinical reality [1-3]. The general
outcome of the clinical trials was that the procedures and longterm
outcome post-stem-cell implantation to the heart via the
coronary arteries are safe. However, improvement in long-term
functional performance of the heart was either not achieved or
was marginal [1-3].
There are several central issues pertaining to the use of cell
implantation in stem-cell therapy: the number of implanted stem
cells has to be high since there is massive cell death following
implantation or injection of cells into the heart or the blood
circulation. Another central issue in stem-cell implantation for
organ repair is the creation of a receptive cell environment in the
ischemic organ. Several factors (e.g. inhibition of inflammation
and apoptosis, secretion of cell growth factors etc.) are necessary
for optimal cell implantation [4]. The injected cells may have to
migrate from the circulating blood to the ischemic niche. They
can then remain active and secrete growth factors, exerting a paracrine effect on the ischemic tissue [5]. Alternatively, they
may stimulate the small population of stem cells in the ischemic
organ (such as the heart), to proliferate and differentiate so as to
enhance cardiac repair post-MI [6]. Another issue is the timing
of injection of the stem cells to the infarcted heart and effect of
MI (inflammatory phase) on the BM [7]. Photobiostimulation
of cells in the bone marrow (BM), that is enriched with various
progenitor cells, by low level laser therapy (LLLT) may suggest a
new approach that may overcome some of the above limitation.
This new approach will be discussed in the present mini review
[8-11].
LOW LEVEL LASER THERAPY FOR THE ISCHEMIC
HEART
In general LLLT has been found to modulate various biological
processes, such as increasing mitochondrial respiration and ATP
synthesis, facilitating wound healing, and promoting the process
of skeletal muscle regeneration and angiogenesis [12,13]. It
was previously shown that LLLT can enhance skeletal muscle
regeneration following partial excision in the rat hind limb
muscles when the laser was delivered directly to the injured
organ multiple times (for 2 min each time) following injury [14].
This phenomenon was even more prominent following cold
injury to the frog skeletal muscles indicating that enhancement
of regeneration by LLLT is probably a general phenomenon in
vertebrates and maybe more effective in cold blooded animals
which innately have a lower metabolic rate in their cells [13-15].
In an experimental model of the infarcted heart in rats and dogs, it
was demonstrated that LLLT (Diode –Ga-Al-As 810nm at a power
density of 5 mW/cm2 for 120sec duration of laser exposure
comprising 0.6 J/cm2), application directly to the infarcted area
in the heart at optimal power parameters significantly reduces
infarct size (scar tissue formation) [16,17]. This phenomenon
was partially attributed to a significant elevation in ATP content,
heat shock proteins, vascular endothelial growth factor (VEGF),
and angiogenesis in the ischemic zone of the laser-irradiated
rats, as compared to non-irradiated rats [16,17]. The mechanism
associated with the photobiostimulation by LLLT is not yet
clearly understood [12]. There is evidence that cytochrome c
oxidase and perhaps also plasma membranes in cells function as
photoacceptors of the photons, and thereafter a cascade of events
occur in the mitochondria, leading to effects on various processes
like ATP production, up-regulation of VEGF, etc [12].
The effect of photobiostimulation on stem cells or progenitor
cells has not been extensively studied [18-21]. It was previously
shown that laser application (Diode laser at 50mW/cm2 for
100sec, energy density 0.5 mW/cm2) to the mesenchymal stem
cells isolated from bone marrow or cardiac stem cells causes a
significant increase in their proliferation in vitro [20]. Based on
previous studies that showed an increase in cytoprotective effect
on the ischemic heart following LLLT, a new approach was taken
to apply laser irradiation to stem cells grown in culture prior
to their implantation to the infarcted heart as a cell therapy for
heart repair [21]. In that study it was demonstrated that MSCs
that were laser treated prior to their implantation to the rat
infarcted heart caused a significant reduction in infarct size
as compared to MSCs that were injected to the heart without
prior laser treatment. This phenomenon was also associated
with significant elevation of vascular endothelial growth factor
(VEGF) in the myocardium of the rats that received the lasertreated
MSCs. In a recent study [8] the possibility of recruiting
autologous stem cells stimulated by LLLT in the BM to the
infarcted heart was addressed. The rationale behind the attempt
to use LLLT to induce the “crude” BM in the bone was, and still is,
that one cannot significantly affect the complex process post-MI
or ischemic injury to the kidney with a single type of stem cell. The
native BM is known for its many types and subtypes of stem cells,
which are defined by their reactivity to various antibodies. The
BM also contains many progenitor cells (i.e. monocytes) that can
further differentiate, for example to macrophages. Macrophages
have been shown recently to have a crucial role in the scarring
process post-MI. Thus LLLT may induce concomitantly in the BM
various types of cells that will increase in number in the blood
circulation following their enhanced proliferation in the BM.
These cells will probably, eventually, and to a certain extent and
under certain circumstances, home in on the ischemic zone in the
ischemic organ (heart, kidney etc.). In this study [8] it was found
that when LLLT was applied in vivo to the BM, and MSCs were
isolated from that BM 3 and 6 weeks later and grown in vitro, they
grew at a higher rate of proliferation relative to MSCs isolated
from non-laser-treated BM. This indicated that the MSCs when
in the BM, following LLLT application in vivo can be induced to
proliferate to a higher rate than non-treated MSCs. Furthermore,
laser application (Diode laser 808nm at power density of 10mW/
cm2 for 100 sec comprising 1J/cm2 energy density) to the BM (at
about 20 min post-MI) caused a marked and significant decrease
(79%) in infarct size 3 weeks post-MI. This extent of infarct size
reduction was even more effective in reducing scarring than that
of laser application directly to the infarcted heart, as also found
in previous studies with infarcted rat and dog hearts [10]. Even
when laser was applied 4 hours post-MI to the BM of infarcted
rats, a marked and significant reduction in the infarcted area
was observed in the laser-treated rats compared to control.
We also found a significantly higher density of c-kit+ (a marker
of MSCs) cells in the myocardium of laser-treated rats relative
to non-treated rat’s post-MI. Moreover, it was demonstrated in
this study that c-kit+ cells post-laser application to the BM of MIinduced
rats, homed specifically in on the infarcted heart and
not on uninjured organs (i.e. liver, kidney) in the same rat [16].
It can be hypothesized that the increased number of c-kit+ cells
found in the myocardium came from proliferating MSCs in the
BM that had migrated to the circulating blood and homed onto
the infarcted heart. Another finding of this study was that of
the preferred homing of the recruited or endogenous c-kit+ cells
in on the infarcted area, rather than their random deposition
throughout the left ventricle in the heart. Indeed, at 3-weeks
post-MI the density of c-kit+ cells in the infarcted area was 27-
fold higher in the rats whose BM had been treated with LLLT as
compared to control rats. Similarly, Hatzistergos et al. [6] found
that endogenous c-kit+ cardiac stem cells increased by 20-fold
in the porcine infarcted heart as compared to control following













